Abstract
The development of inhaled drugs for respiratory diseases is frequently impacted by lung pathology in non-clinical safety studies. To enable design of novel candidate drugs with the right safety profile, predictive in vitro lung toxicity assays are required that can be applied during drug discovery for early hazard identification and mitigation. Here, we describe a novel high-content imaging-based screening assay that allows for quantification of the tight junction protein occludin in A549 cells, as a model for lung epithelial barrier integrity. We assessed a set of compounds with a known lung safety profile, defined by clinical safety or non-clinical in vivo toxicology data, and were able to correctly identify 9 of 10 compounds with a respiratory safety risk and 9 of 9 compounds without a respiratory safety risk (90% sensitivity, 100% specificity). The assay was sensitive at relevant compound concentrations to influence medicinal chemistry optimization programs and, with an accessible cell model in a 96-well plate format, short protocol and application of automated imaging analysis algorithms, this assay can be readily integrated in routine discovery safety screening to identify and mitigate respiratory toxicity early during drug discovery. Interestingly, when we applied physiologically-based pharmacokinetic (PBPK) modelling to predict epithelial lining fluid exposures of the respiratory tract after inhalation, we found a robust correlation between in vitro occludin assay data and lung pathology in vivo, suggesting the assay can inform translational risk assessment for inhaled small molecules.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Inhaled drug delivery is an effective treatment paradigm for respiratory diseases, such as asthma and chronic obstructive pulmonary disease (COPD), due to direct access to the intended target organ and the potential to minimize systemic exposure and consequent side effects (Ruge et al. 2013). The development of innovative inhaled medicines is often impacted by toxicity in the respiratory tract in non-clinical species, leading to attrition or dose limitations in clinic, typically after the selection of novel drug candidates (David et al. 2014). As part of the industry shift from observational to predictive toxicology, a proactive discovery safety strategy aims to identify and mitigate these safety risks early during drug discovery, and thereby enable the design and selection of drug candidates with the right safety profile (Hornberg et al. 2014a; Hornberg and Mow 2014; Morgan et al. 2018). This relies heavily on in vitro methods that are predictive for in vivo toxicity, clinical safety and compatible with the design-make-test-analyze (DMTA) cycle during lead optimization campaigns (Hornberg et al. 2014b; Johansson et al. 2019). A plethora of high-content screening (HCS) methodologies have been developed and deployed to successfully identify for example cardio-, hepato-, neuro-, and nephrotoxicity (Li and ** the modelled ELF concentrations onto the dose response curve. Interestingly, we found that by map** the modelled ELF concentrations onto the occludin dose–response curve, we could show that pathological changes in vivo were only observed in areas of the respiratory tract that were exposed to drug concentrations (14 µM) similar to that of the in vitro IC50 (27 µM) (Fig. 5c). Equally, no pathological changes were observed in areas of the respiratory tract that were exposed to concentrations (3.2 & 0.02 µM Cmax) that did not elicit an effect in the occludin assay. This suggests that a combination of PBPK modelling and this novel in vitro assay can inform translational and quantitative risk assessment for inhaled small molecules.
Discussion
Pharmaceutical R&D productivity requires a proactive discovery safety strategy to identify and mitigate safety risks early to bring forward candidate drugs with the Right Safety profile (Hornberg and Mow 2014; Morgan et al. 2018). To inform decision-making during drug discovery, robust in vitro assays are required that are compatible with integration in the DMTA cycle namely being cost-effective, have sufficient throughput, and with readouts that are predictive for (non)clinical safety (Blass 2021; Hornberg et al. 2014b; Johansson et al. 2019; McKim 2010; Sanders et al. 2017; Szymanski et al. 2012). Currently, a range of organ-specific assays exist, which use a single cell type rather than attempting to fully recapitulate the complexity of the organ they represent, several of which are applicable to early drug screening, for example those aimed towards the cardiovascular system, liver and kidney (Gustafsson et al. 2014; Persson et al. 2013; Pognan et al. 2023; Pointon et al. 2017; Sjogren et al. 2018). Options to screen inhaled drugs for lung toxicity however have lagged behind. Some bronchial cell-based assays have shown some promise when grown in ALI conditions, such as our predictive TEER-based ALI assay for the detection of lung toxicants (Balogh Sivars et al. 2018) and an imaging-based assay correlating junctional protein staining with TEER measurements as a primary readout of lung barrier integrity (Pell et al. 2021). Recently published breathing lung-on-chip models have also shown significant promise as an in vitro tool to study inhalation toxicity (Sengupta et al. 2022). However, these assays come with significant limitations in terms of throughput, cost, and their ability to stratify compounds within a chemical series. Relevant comparative assay details are listed in Table 2. A549 cells have been widely used as a cell model to study toxicological responses, such as cytotoxicity, oxidative stress and inflammatory responses (Barosova et al. 2021). However, TEER values from these cells when elevated to ALI conditions are far lower than alternative alveolar cell lines such as the lentivirus immortalized hAELVi cells. Indeed hAELVi cells have also been shown to possess a more type I phenotype than the type II of the A549 cells and thus representing a larger proportion of the alveolar population (Kuehn et al. 2016). In an effort to develop an assay which can be rapidly deployed for screening within a DMTA cycle, we focused on measuring a single endpoint from a widely studied cell line which, whilst not recapitulating the full complexity of the lung alveolar space allowed for robust measurement of a physiologically relevant parameter such as junctional protein morphology. Here, we showed that quantification of occludin staining in A549 cells in a simple 2D format can detect lung toxicants/irritants with 90% sensitivity and 100% specificity after 24 h compound administration. This is likely driven by the central role played by junctional proteins in maintaining an effective epithelial barrier in the lung. Occludin is a classic tight junction protein which functions to link cells to adjacent cells and to the internal cytoskeletal structures (Kojima et al. 2013). It has been shown to have a clear link to barrier maintenance, ATP production and gene regulation (Castro et al. 2018), whilst studies with occludin knock-out mice have also shown chronic inflammation amongst other pathologies, indicating not only a wide role for occludin within cellular homeostasis but also a pivotal link to inflammation and pathological processes (Saitou et al. 2000; Sugita and Kabashima 2020).
To develop an imaging-based assay measuring localization of a tight junction protein in a simple 2D culture, growth conditions of the chosen cell line were found to be of utmost importance. Specifically, in the development of this assay, it was found that the morphological characteristics of the bronchial cell lines 16HBE and Calu3 were incompatible with the requirements of the imaging algorithm. In order for robust measurement of the occludin staining pattern, it was necessary to use a whole cell stain to allow for determination of cell edges independent of the junctional marker to narrow the region of measurement to those edges. In the case of Calu3, these cells displayed a highly overlap** growth pattern, tending to form multilayered colonies rather than a uniform monolayer of cells. 16HBE, on the other hand, grew in a highly consistent monolayer fashion but displayed an undulating pattern of cell-to-cell contact rather than a liner line. These inherent conditions posed a challenge when it came to determining the degree of overlap of the junctional marker with the defined cell edge. Alternative seeding densities and culture time points were explored (data not shown). However, A549 cells were consistently found to provide the most robust basis to enable screening assay development.
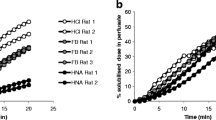
In order to apply in vitro toxicology assays to quantitative risk assessment, it is essential to incorporate drug exposure information and establish how in vitro concentrations relate to clinical exposure (Bell et al. 2018). While there are challenges to scale compound concentrations and elicited effects in in vitro assays to plasma drug exposure and adverse events in the clinic, it has been possible to use in vitro assay data for quantitative risk assessment of some organ toxicities (Albrecht et al. 2019; Archer et al. 2018; Hengstler et al. 2020; Kappenberg et al. 2020; O'Brien et al. 2006; Persson et al. 2013; Sjogren et al. 2018). Quantitative risk assessment has been more challenging for inhaled drugs because, unlike for orally administered drugs, the systemic plasma exposure at a given inhaled dose does not accurately reflect the relevant exposure in the lung (Frohlich 2019; Kassinos et al. 2021). To address this gap, we applied an existing PBPK modelling approach, which mathematically describes respiratory tract physiology and the fate of deposited drug dose, and accurately predicts PK, target site exposure, and interestingly, spatial heterogeneity in target site concentrations within the lung (Boger et al. 2016; Boger and Friden 2019). This approach allowed us to directly correlate actual compound concentrations from the in vitro assay with calculated compound exposure in the ELF in a toxicity study and demonstrate that the pathological changes consistent with airway irritation were restricted to areas of the respiratory tract that reached concentrations that aligned with the IC50 of the imaging assay. While recent studies have shown the applicability of modelling to in vitro to in vivo extrapolation (IVIVE) for toxic vapours from agrichemicals (Moreau et al. 2022) and environmental contaminants such as endocrine disrupters (**e et al. 2023), to our knowledge, this is the first-time in vitro toxicity data have been quantitatively linked to exposure levels in the lung for compound risk assessment in drug development.
The propensity of a compound to cause to toxicity may depend on primary target pharmacology, off-target secondary pharmacology and chemical reactivity, which originate from its chemical structure and physicochemical properties, and can manifest in many different downstream effects in the cell, including direct damage to organelles or DNA, membrane permeability, impaired cellular signalling or metabolism. In general, assessing cell health parameters has proven an effective trade-off between, on the one hand, ensuring high sensitivity (compared to more generic cell survival or cytotoxicity assessment) and, on the other hand, providing robust, accessible screening options with throughput (compared to more specific but therefore less-sensitive molecular endpoints). Here, the validation compound set encompassed diverse physicochemical properties, therapeutic targets, off-target pharmacology, and a range of pathologies across multiple pre-clinical tox species, including epithelial erosion, atrophy, oedema, hyperplasia and inflammation. In summary, we present a novel imaging-based assay that can be applied to identify and mitigate the risk for lung toxicity of inhaled compounds during early drug discovery, and for quantitative risk assessment in drug development. This will enable acceleration of the identification of inhaled drug candidates with the right safety profile without the need for animal studies.
References
Aghapour M, Raee P, Moghaddam SJ, Hiemstra PS, Hei**k IH (2018) Airway epithelial barrier dysfunction in chronic obstructive pulmonary disease: role of cigarette smoke exposure. Am J Respir Cell Mol Biol 58(2):157–169. https://doi.org/10.1165/rcmb.2017-0200TR
Albrecht W, Kappenberg F, Brecklinghaus T et al (2019) Prediction of human drug-induced liver injury (DILI) in relation to oral doses and blood concentrations. Arch Toxicol 93(6):1609–1637. https://doi.org/10.1007/s00204-019-02492-9
Archer CR, Sargeant R, Basak J, Pilling J, Barnes JR, Pointon A (2018) Characterization and validation of a human 3D cardiac microtissue for the assessment of changes in cardiac pathology. Sci Rep 8(1):10160. https://doi.org/10.1038/s41598-018-28393-y
Balogh Sivars K, Sivars U, Hornberg E et al (2018) A 3D human airway model enables prediction of respiratory toxicity of inhaled drugs in vitro. Toxicol Sci 162(1):301–308. https://doi.org/10.1093/toxsci/kfx255
Barosova H, Meldrum K, Karakocak BB et al (2021) Inter-laboratory variability of A549 epithelial cells grown under submerged and air-liquid interface conditions. Toxicol in Vitro. https://doi.org/10.1016/j.tiv.2021.105178
Bell SM, Chang X, Wambaugh JF et al (2018) In vitro to in vivo extrapolation for high throughput prioritization and decision making. Toxicol in Vitro 47:213–227. https://doi.org/10.1016/j.tiv.2017.11.016
Berube K, Prytherch Z, Job C, Hughes T (2010) Human primary bronchial lung cell constructs: the new respiratory models. Toxicology 278(3):311–318. https://doi.org/10.1016/j.tox.2010.04.004
Blass BE (2021) Chapter 4: in vitro screening systems. In: Blass BE (ed) Basic principles of drug discovery and development (Second Edition). Academic Press, pp 185–256
Boger E, Friden M (2019) Physiologically based pharmacokinetic/pharmacodynamic modeling accurately predicts the better bronchodilatory effect of inhaled versus oral salbutamol dosage forms. J Aerosol Med Pulm Drug Deliv 32(1):1–12. https://doi.org/10.1089/jamp.2017.1436
Boger E, Evans N, Chappell M et al (2016) Systems pharmacology approach for prediction of pulmonary and systemic pharmacokinetics and receptor occupancy of inhaled drugs. CPT Pharmacometrics Syst Pharmacol 5(4):201–210. https://doi.org/10.1002/psp4.12074
Castro V, Skowronska M, Lombardi J et al (2018) Occludin regulates glucose uptake and ATP production in pericytes by influencing AMP-activated protein kinase activity. J Cereb Blood Flow Metab 38(2):317–332. https://doi.org/10.1177/0271678X17720816
Cooper JA 2nd, Saracci R, Cole P (1979) Describing the validity of carcinogen screening tests. Br J Cancer 39(1):87–89. https://doi.org/10.1038/bjc.1979.10
David C, Dearg B, Robert A et al (2014) Lessons learned from the fate of AstraZeneca’s drug pipeline: a five-dimensional framework. Nat Rev Drug Discovery 13(6):419. https://doi.org/10.1038/nrd4309
Frohlich E (2019) Biological obstacles for identifying in vitro-in vivo correlations of orally inhaled formulations. Pharmaceutics 11:7. https://doi.org/10.3390/pharmaceutics11070316
Gon Y, Hashimoto S (2018) Role of airway epithelial barrier dysfunction in pathogenesis of asthma. Allergol Int 67(1):12–17. https://doi.org/10.1016/j.alit.2017.08.011
Gustafsson F, Foster AJ, Sarda S, Bridgland-Taylor MH, Kenna JG (2014) A correlation between the in vitro drug toxicity of drugs to cell lines that express human P450s and their propensity to cause liver injury in humans. Toxicol Sci 137(1):189–211. https://doi.org/10.1093/toxsci/kft223
Hashimoto Y (1862) Campbell M (2020) Tight junction modulation at the blood-brain barrier: current and future perspectives. Biochim Biophys Acta BBA Biomembranes. https://doi.org/10.1016/j.bbamem.2020.183298
Hengstler JG, Sjogren AK, Zink D, Hornberg JJ (2020) In vitro prediction of organ toxicity: the challenges of scaling and secondary mechanisms of toxicity. Arch Toxicol 94(2):353–356. https://doi.org/10.1007/s00204-020-02669-7
Hiemstra PS, Grootaers G, van der Does AM, Krul CAM, Kooter IM (2018) Human lung epithelial cell cultures for analysis of inhaled toxicants: lessons learned and future directions. Toxicol in Vitro 47:137–146. https://doi.org/10.1016/j.tiv.2017.11.005
Hornberg JJ, Mow T (2014) How can we discover safer drugs? Future Med Chem 6(5):481–483. https://doi.org/10.4155/fmc.14.15
Hornberg JJ, Laursen M, Brenden N et al (2014a) Exploratory toxicology as an integrated part of drug discovery. Part I: why and how. Drug Discov Today 19(8):1131–1136. https://doi.org/10.1016/j.drudis.2013.12.008
Hornberg JJ, Laursen M, Brenden N et al (2014b) Exploratory toxicology as an integrated part of drug discovery. Part II: Screening strategies. Drug Discov Today 19(8):1137–1144. https://doi.org/10.1016/j.drudis.2013.12.009
Johansson J, Larsson MH, Hornberg JJ (2019) Predictive in vitro toxicology screening to guide chemical design in drug discovery. Curr Opin Toxicol 15:99–108. https://doi.org/10.1016/j.cotox.2019.08.005
Kappenberg F, Brecklinghaus T, Albrecht W et al (2020) Handling deviating control values in concentration-response curves. Arch Toxicol 94(11):3787–3798. https://doi.org/10.1007/s00204-020-02913-0
Kassinos S, Bäckman P, Conway J, Hickey AJ, Hickey AJJ (2021) Inhaled Medicines : Optimizing Development Through Integration of in Silico, in Vitro and in Vivo Approaches. Academic Press 2021
Knudsen L, Ochs M (2018) The micromechanics of lung alveoli: structure and function of surfactant and tissue components. Histochem Cell Biol 150(6):661–676. https://doi.org/10.1007/s00418-018-1747-9
Kojima T, Go M, Takano K et al (2013) Regulation of tight junctions in upper airway epithelium. Biomed Res Int. https://doi.org/10.1155/2013/947072
Kuehn A, Kletting S, de Souza C-W et al (2016) Human alveolar epithelial cells expressing tight junctions to model the air-blood barrier. Altex 33(3):251–260. https://doi.org/10.14573/altex.1511131
Lee D, Wexler AS (2011) Particle deposition in juvenile rat lungs: a model study. J Aerosol Sci 42(9):567–579. https://doi.org/10.1016/j.jaerosci.2011.06.004
Li S, **a M (2019) Review of high-content screening applications in toxicology. Arch Toxicol 93(12):3387–3396. https://doi.org/10.1007/s00204-019-02593-5
McKim JM Jr (2010) Building a tiered approach to in vitro predictive toxicity screening: a focus on assays with in vivo relevance. Comb Chem High Throughput Screen 13(2):188–206. https://doi.org/10.2174/138620710790596736
Moreau M, Fisher J, Andersen ME et al (2022) NAM-based prediction of point-of-contact toxicity in the lung: a case example with 1,3-dichloropropene. Toxicology. https://doi.org/10.1016/j.tox.2022.153340
Morgan P, Brown DG, Lennard S et al (2018) Impact of a five-dimensional framework on R and D productivity at AstraZeneca. Nat Rev Drug Discov 17(3):167–181. https://doi.org/10.1038/nrd.2017.244
Nernst W (1904) Theorie der Reaktionsgeschwindigkeit in heterogenen Systemen. Zeitschrift für Physikalische Chemie 47U(1):52–55. https://doi.org/10.1515/zpch-1904-4704
Noyes AA, Whitney WR (1897) The rate of solution of solid substances in their own solutions. J Am Chem Soc 19(12):930–934. https://doi.org/10.1021/ja02086a003
O’Brien PJ, Irwin W, Diaz D et al (2006) High concordance of drug-induced human hepatotoxicity with in vitro cytotoxicity measured in a novel cell-based model using high content screening. Arch Toxicol 80(9):580–604. https://doi.org/10.1007/s00204-006-0091-3
Overgaard CE, Mitchell LA, Koval M (2012) Roles for claudins in alveolar epithelial barrier function. Ann N Y Acad Sci 1257:167–174. https://doi.org/10.1111/j.1749-6632.2012.06545.x
Pell TJ, Gray MB, Hopkins SJ et al (2021) Epithelial barrier integrity profiling: combined approach using cellular junctional complex imaging and transepithelial electrical resistance. SLAS Discov 26(7):909–921. https://doi.org/10.1177/24725552211013077
Persson M, Hornberg JJ (2016) Advances in predictive toxicology for discovery safety through high content screening. Chem Res Toxicol 29(12):1998–2007. https://doi.org/10.1021/acs.chemrestox.6b00248
Persson M, Loye AF, Mow T, Hornberg JJ (2013) A high content screening assay to predict human drug-induced liver injury during drug discovery. J Pharmacol Toxicol Methods 68(3):302–313. https://doi.org/10.1016/j.vascn.2013.08.001
Pognan F, Beilmann M, Boonen HCM et al (2023) The evolving role of investigative toxicology in the pharmaceutical industry. Nat Rev Drug Discov 22(4):317–335. https://doi.org/10.1038/s41573-022-00633-x
Pointon A, Pilling J, Dorval T, Wang Y, Archer C, Pollard C (2017) From the cover: high-throughput imaging of cardiac microtissues for the assessment of cardiac contraction during drug discovery. Toxicol Sci 155(2):444–457. https://doi.org/10.1093/toxsci/kfw227
Rouaud F, Sluysmans S, Flinois A, Shah J, Vasileva E (2020) Scaffolding proteins of vertebrate apical junctions: structure, functions and biophysics. Biochim Biophys Acta Biomembr. https://doi.org/10.1016/j.bbamem.2020.183399
Ruge CA, Kirch J, Lehr C-M (2013) Pulmonary drug delivery: from generating aerosols to overcoming biological barriers—therapeutic possibilities and technological challenges. Lancet Respir Med 1(5):402–413. https://doi.org/10.1016/S2213-2600(13)70072-9
Saitou M, Furuse M, Sasaki H et al (2000) Complex phenotype of mice lacking occludin, a component of tight junction strands. Mol Biol Cell 11(12):4131–4142. https://doi.org/10.1091/mbc.11.12.4131
Sanders JM, Beshore DC, Culberson JC et al (2017) Informing the selection of screening hit series with in silico absorption, distribution, metabolism, excretion, and toxicity profiles. J Med Chem 60(16):6771–6780. https://doi.org/10.1021/acs.jmedchem.6b01577
Schilpp C, Lochbaum R, Braubach P et al (2021) TGF-beta1 increases permeability of ciliated airway epithelia via redistribution of claudin 3 from tight junction into cell nuclei. Pflugers Arch 473(2):287–311. https://doi.org/10.1007/s00424-020-02501-2
Sengupta A, Roldan N, Kiener M et al (2022) A new immortalized human alveolar epithelial cell model to study lung injury and toxicity on a breathing lung-on-chip system. Front Toxicol. https://doi.org/10.3389/ftox.2022.840606
Sjogren AK, Breitholtz K, Ahlberg E et al (2018) A novel multi-parametric high content screening assay in ciPTEC-OAT1 to predict drug-induced nephrotoxicity during drug discovery. Arch Toxicol 92(10):3175–3190. https://doi.org/10.1007/s00204-018-2284-y
Slifer ZM, Blikslager AT (2020) The integral role of tight junction proteins in the repair of injured intestinal epithelium. Int J Mol Sci 21:3. https://doi.org/10.3390/ijms21030972
Sugita K, Kabashima K (2020) Tight junctions in the development of asthma, chronic rhinosinusitis, atopic dermatitis, eosinophilic esophagitis, and inflammatory bowel diseases. J Leukoc Biol 107(5):749–762. https://doi.org/10.1002/JLB.5MR0120-230R
Szymanski P, Markowicz M, Mikiciuk-Olasik E (2012) Adaptation of high-throughput screening in drug discovery-toxicological screening tests. Int J Mol Sci 13(1):427–452. https://doi.org/10.3390/ijms13010427
Upadhyay S, Palmberg L (2018) Air-liquid interface: relevant in vitro models for investigating air pollutant-induced pulmonary toxicity. Toxicol Sci 164(1):21–30. https://doi.org/10.1093/toxsci/kfy053
Vielle NJ, Garcia-Nicolas O, Oliveira Esteves BI, Brugger M, Summerfield A, Alves MP (2019) The human upper respiratory tract epithelium is susceptible to flaviviruses. Front Microbiol 10:811. https://doi.org/10.3389/fmicb.2019.00811
**e R, Wang X, Xu Y, Zhang L, Ma M, Wang Z (2023) In vitro to in vivo extrapolation for predicting human equivalent dose of phenolic endocrine disrupting chemicals: PBTK model development, biological pathways, outcomes and performance. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2023.165271
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors were employees of AstraZeneca at the time of study completion and were compensated as such.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fitzpatrick, P.A., Johansson, J., Maglennon, G. et al. A novel in vitro high-content imaging assay for the prediction of drug-induced lung toxicity. Arch Toxicol (2024). https://doi.org/10.1007/s00204-024-03800-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00204-024-03800-8