Abstract
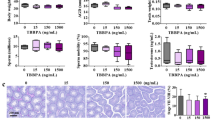
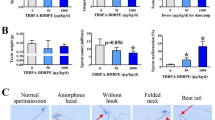
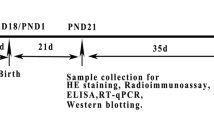
Despite growing concern about adverse effects of bisphenol AF (BPAF) due to its endocrine disrupting properties, there is a lack of toxicity data from low-dose studies and direct evidence linking its adverse effects to endocrine disrupting properties. Here, we investigated the effects of gestational and postnatal exposure to BPAF through drinking water (0.15–15 μg/mL, equivalent to the daily intake of ~ 50 and 5 mg/kg/day) on testis development in mice. We found that like mestranol, 5 mg/kg/day BPAF resulted in remarkable decreases in multiple male reproductive parameters in adulthood, such as the sperm number and serum testosterone level. Notably, 50 μg/kg/day BPAF also caused significant decreases in anogenital distance (AGD), the luteinizing hormone level and spermatocyte number, along with declining trends in sperm number and the serum levels of testosterone and follicle-stimulating hormone. In line with the adverse outcomes observed in adulthood, on postnatal day (PND) 9, we also observed BPAF-caused dose-dependent alterations, including reduced AGD, seminiferous tubule area and numbers of total germ cells, spermatocytes and Leydig cells, coupled with down-regulated expression of male-biased genes in testes. Even when exposure to 5 mg/kg/day BPAF as well as MES was initiated from PND 0, similar alterations in male reproductive parameters were also found on PND 9, along with a decrease in the GnRH content in the hypothalamus; moreover, testicular alterations and the reduction in AGD were partly antagonized by the estrogen receptor (ER) antagonist ICI 182,780, but the reduction of GnRH production was not done, showing that the effects of BPAF on testis development may be partially mediated by ER signaling. In conclusion, all the findings demonstrate that low-dose BPAF can partly disrupt mammal testis development and cause adverse testicular outcomes in adulthood, indicating a potential reproductive risk to mammals including humans. Importantly, our finding that developmental alterations elicited by BPAF have been detectable on PND 9 provides important motivation for the development of effective methods for early detection of adverse effects of estrogenic chemicals on testis development.






Similar content being viewed by others
References
Cao LY, Ren XM, Li CH et al (2017) Bisphenol AF and bisphenol B exert higher estrogenic effects than bisphenol A via G protein-coupled estrogen receptor pathway. Environ Sci Technol 51(19):11423–11430. https://doi.org/10.1021/acs.est.7b03336
Chen D, Kannan K, Tan H et al (2016) Bisphenol analogues other than BPA: environmental occurrence, human exposure, and toxicity-a review. Environ Sci Technol 50(11):5438–5453. https://doi.org/10.1021/acs.est.5b05387
Chimento A, Sirianni R, Casaburi I, Pezzi V (2014) Role of estrogen receptors and G protein-coupled estrogen receptor in regulation of hypothalamus-pituitary-testis axis and spermatogenesis. Front Endocrinol 5:1. https://doi.org/10.3389/fendo.2014.00001
Dean A, Sharpe RM (2013) Clinical review: anogenital distance or digit length ratio as measures of fetal androgen exposure: relationship to male reproductive development and its disorders. J Clin Endocrinol Metab 98(6):2230–2238. https://doi.org/10.1210/jc.2012-4057
D’Souza R, Gill-Sharma MK, Pathak S, Kedia N, Kumar R, Balasinor N (2005) Effect of high intratesticular estrogen on the seminiferous epithelium in adult male rats. Mol Cell Endocrinol 241(1–2):41–48. https://doi.org/10.1016/j.mce.2005.04.011
ECHA (2020) Mass of bisphenol AF and benzyl(diethylamino)diphenylphosphonium. https://www.echaeuropaeu/documents/10162/2842450/clh_rep_React+mass+of+bisphenol+AF+and+benzyl%28diethylamino%29diphenylphosphonium_15689_enpdf/23d8e2cc-5fbd-9d83-3d36-cde6cdeed223. Accessed 25 Dec 2021
Escriva L, Zilliacus J, Hessel E, Beronius A (2021) Assessment of the endocrine disrupting properties of bisphenol AF: a case study applying the European regulatory criteria and guidance. Environ Health Glob Acc Sci Sour 20(1):48. https://doi.org/10.1186/s12940-021-00731-0
Feng Y, Yin J, Jiao Z, Shi J, Li M, Shao B (2012) Bisphenol AF may cause testosterone reduction by directly affecting testis function in adult male rats. Toxicol Lett 211(2):201–209. https://doi.org/10.1016/j.toxlet.2012.03.802
Guyot R, Odet F, Leduque P, Forest MG, Le Magueresse-Battistoni B (2004) Diethylstilbestrol inhibits the expression of the steroidogenic acute regulatory protein in mouse fetal testis. Mol Cell Endocrinol 220(1–2):67–75. https://doi.org/10.1016/j.mce.2004.03.008
Ikeda Y, Tanaka H, Esaki M (2008) Effects of gestational diethylstilbestrol treatment on male and female gonads during early embryonic development. Endocrinology 149(8):3970–3979. https://doi.org/10.1210/en.2007-1599
Kitamura S, Suzuki T, Sanoh S et al (2005) Comparative study of the endocrine-disrupting activity of bisphenol A and 19 related compounds. Toxicol Sci off J Soc Toxicol 84(2):249–259. https://doi.org/10.1093/toxsci/kfi074
Lee PC (1998) Disruption of male reproductive tract development by administration of the xenoestrogen, nonylphenol, to male newborn rats. Endocrine 9(1):105–111. https://doi.org/10.1385/endo:9:1:105
Li J, Sheng N, Cui R et al (2016) Gestational and lactational exposure to bisphenol AF in maternal rats increases testosterone levels in 23-day-old male offspring. Chemosphere 163:552–561. https://doi.org/10.1016/j.chemosphere.2016.08.059
Li A, Zhuang T, Shi W et al (2020) Serum concentration of bisphenol analogues in pregnant women in China. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2019.136100
Li Y, Dong M, **ong Y et al (2022) Effects of postnatal exposure to tetrabromobisphenol A on testis development in mice and early key events. Arch Toxicol. https://doi.org/10.1007/s00204-022-03259-5
Liu Y, Zhang S, Song N et al (2017) Occurrence, distribution and sources of bisphenol analogues in a shallow Chinese freshwater lake (Taihu Lake): Implications for ecological and human health risk. Sci Total Environ 599–600:1090–1098. https://doi.org/10.1016/j.scitotenv.2017.05.069
Liu J, Zhang L, Lu G, Jiang R, Yan Z, Li Y (2021a) Occurrence, toxicity and ecological risk of Bisphenol A analogues in aquatic environment: a review. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2020.111481
Liu X, Xue Q, Zhang H, Fu J, Zhang A (2021b) Structural basis for molecular recognition of G protein-coupled estrogen receptor by selected bisphenols. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2021.148558
Martin OV, Shialis T, Lester JN, Scrimshaw MD, Boobis AR, Voulvoulis N (2008) Testicular dysgenesis syndrome and the estrogen hypothesis: a quantitative meta-analysis. Environ Health Perspect 116(2):149–157. https://doi.org/10.1289/ehp.10545
Matsushima A, Liu X, Okada H, Shimohigashi M, Shimohigashi Y (2010) Bisphenol AF is a full agonist for the estrogen receptor ERalpha but a highly specific antagonist for ERbeta. Environ Health Perspect 118(9):1267–1272. https://doi.org/10.1289/ehp.0901819
NTP (2008) Chemical information profile for bisphenol AF [CAS No. 1478–61–1]. http://www.ntpniehsnihgov/. Accessed 21 Dec 2021
Pan Y, Deng M, Li J et al (2020) Occurrence and maternal transfer of multiple bisphenols, including an emerging derivative with unexpectedly high concentrations, in the human maternal-fetal-placental unit. Environ Sci Technol 54(6):3476–3486. https://doi.org/10.1021/acs.est.0c00206
Qin F, Wang X, Liu S et al (2014) Gene expression profiling of key genes in hypothalamus-pituitary-gonad axis of rare minnow Gobiocypris rarus in response to EE2. Gene 552(1):8–17. https://doi.org/10.1016/j.gene.2014.09.006
Schwartz CL, Christiansen S, Vinggaard AM, Axelstad M, Hass U, Svingen T (2019) Anogenital distance as a toxicological or clinical marker for fetal androgen action and risk for reproductive disorders. Arch Toxicol 93(2):253–272. https://doi.org/10.1007/s00204-018-2350-5
Shamhari AA, Abd Hamid Z, Budin SB, Shamsudin NJ, Taib IS (2021) Bisphenol A and its analogues deteriorate the hormones physiological function of the male reproductive system: a mini-review. Biomedicines. https://doi.org/10.3390/biomedicines9111744
Sharpe RM, Atanassova N, McKinnell C et al (1998) Abnormalities in functional development of the Sertoli cells in rats treated neonatally with diethylstilbestrol: a possible role for estrogens in Sertoli cell development. Biol Reprod 59(5):1084–1094. https://doi.org/10.1095/biolreprod59.5.1084
Song S, Ruan T, Wang T, Liu R, Jiang G (2012) Distribution and preliminary exposure assessment of bisphenol AF (BPAF) in various environmental matrices around a manufacturing plant in China. Environ Sci Technol 46(24):13136–13143. https://doi.org/10.1021/es303960k
Teng C, Goodwin B, Shockley K et al (2013) Bisphenol A affects androgen receptor function via multiple mechanisms. Chem Biol Interact 203(3):556–564. https://doi.org/10.1016/j.cbi.2013.03.013
Toppari J, Larsen JC, Christiansen P et al (1996) Male reproductive health and environmental xenoestrogens. Environ Health Perspect 104(Suppl 4):741–803. https://doi.org/10.1289/ehp.96104s4741
Umano T, Tanaka R, Yamasaki K (2012) Endocrine-mediated effects of 4,4’-(hexafluoroisopropylidene)diphenol in SD rats, based on a subacute oral toxicity study. Arch Toxicol 86(1):151–157. https://doi.org/10.1007/s00204-011-0731-0
Waidyanatha S, Black SR, Aillon K et al (2019) Toxicokinetics and bioavailability of bisphenol AF following oral administration in rodents: a dose, species, and sex comparison. Toxicol Appl Pharmacol 373:39–47. https://doi.org/10.1016/j.taap.2019.04.015
Waidyanatha S, Collins BJ, Cunny H et al (2021) An investigation of systemic exposure to bisphenol AF during critical periods of development in the rat. Toxicol Appl Pharmacol. https://doi.org/10.1016/j.taap.2020.115369
Wu D, Huang CJ, Jiao XF et al (2019) Bisphenol AF compromises blood-testis barrier integrity and sperm quality in mice. Chemosphere. https://doi.org/10.1016/j.chemosphere.2019.124410
Yamasaki K, Takeyoshi M, Sawaki M, Imatanaka N, Shinoda K, Takatsuki M (2003) Immature rat uterotrophic assay of 18 chemicals and Hershberger assay of 30 chemicals. Toxicology 183(1–3):93–115. https://doi.org/10.1016/s0300-483x(02)00445-6
Yang Y, Guan J, Yin J, Shao B, Li H (2014a) Urinary levels of bisphenol analogues in residents living near a manufacturing plant in south China. Chemosphere 112:481–486. https://doi.org/10.1016/j.chemosphere.2014.05.004
Yang Y, Lu L, Zhang J, Yang Y, Wu Y, Shao B (2014b) Simultaneous determination of seven bisphenols in environmental water and solid samples by liquid chromatography-electrospray tandem mass spectrometry. J Chromatogr A 1328:26–34. https://doi.org/10.1016/j.chroma.2013.12.074
Yang S, Cheng W, Li X et al (2020) Use of embryonic stem cell-derived cardiomyocytes to study cardiotoxicity of bisphenol AF via the GPER/CAM/eNOS pathway. Toxicology. https://doi.org/10.1016/j.tox.2020.152380
Yu Y, **n X, Ma F et al (2022) Bisphenol AF blocks Leydig cell regeneration from stem cells in male rats. Environ Pollut. https://doi.org/10.1016/j.envpol.2022.118825
Zhang B, He Y, Zhu H et al (2020) Concentrations of bisphenol A and its alternatives in paired maternal-fetal urine, serum and amniotic fluid from an e-waste dismantling area in China. Environ Int. https://doi.org/10.1016/j.envint.2019.105407
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2018YFA0901103) and National Natural Science Foundation of China (22076211). The authors would like to thank Professor Wei Li and Dr. Chao Liu (State Key Laboratory of Stem Cell and Reproductive Biology, Institute of Zoology, Chinese Academy of Sciences) for some assistance with sperm analysis. Figure 6 was created in BioRender.com (https://biorender.com/).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
204_2022_3377_MOESM1_ESM.docx
Supplementary file1 Experimental section including RNA extraction and RT-qPCR, IF assay, quantitative analysis of seminiferous tubules. A table listing primer sequences of genes. (DOCX 23 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., **ong, Y., Lv, L. et al. Effects of low-dose bisphenol AF on mammal testis development via complex mechanisms: alterations are detectable in both infancy and adulthood. Arch Toxicol 96, 3373–3383 (2022). https://doi.org/10.1007/s00204-022-03377-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-022-03377-0