Abstract
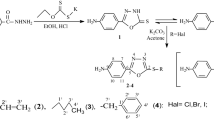
The basic nucleus 4-amino-5-[4-(methylsulfonyl)benzyl]-2,4-dihydro-3H-[1,2,4]-triazole-3-thione was prepared by cyclisation of potassium dithiocarbazinate with hydrazine hydrate using ethanol as solvent under reflux condition. The compound which has been synthesized successfully was subjected to addition reaction with different aldehydes to synthesize Schiff bases. The compounds were confirmed by spectroscopic methods (IR, NMR, and Mass). In order to ascertain the pharmaceutical application, the selective pharmacological screening of the derivatives was carried out according to the standard procedures. The compounds were screened for their cytotoxic activity and antimicrobial activity.

Similar content being viewed by others
References
Banachiewicz BM, Banachiewicz J, Chodkowska A, Wojtowicz EJ, Mazur L (2004) Synthesis and biological activity of new derivatives of 3-(3,4-diaryl-[1,2,4]-triazole-5-yl)propenoic acid. Eur J Med Chem 39:873–887. doi:10.1016/j.ejmech.2004.07.002
Bhandari SV, Bothara KG, Raut MK, Patil AA, Sarkate AP, Mokale VJ (2008) Design, synthesis and evaluation of anti-inflammatory, analgesic and ulcerogenicity studies of novel s-substituted phenacyl-[1,3,4]-oxadiazole-2-thiol and Schiff bases of diclofenac acid as nonulcerogenic derivatives. Bioorg Med Chem 16:1822–1831
Hearn MJ, Cynamon MH (2004) Design and synthesis of antituberculars: preparation and evaluation against Mycobacterium tuberculosis of an isoniazid Schiff base. J Antimicrob Chemother 53:185–191. doi:10.1093/jac/dkh041
Holla BS, Veerendra B, Shivananda MK, Poojary B (2003) Synthesis characterization and anticancer activity studies on some Mannich bases derived from [1,2,4]-triazoles. Eur J Med Chem 38:759–767. doi:10.1016/S0223-5234(03)00128-4
Jarrahpour A, Khalili D, De Clercq E, Salmi C, Brunel JM (2007) Synthesis, antibacterial, antifungal and antiviral activity evaluation of some new bis-Schiff bases of isatin and their derivatives. Molecules 12(8):1720–1730. doi:10.3390/12081720
Kaplan JP, Raizon BM, Desarmenien M, Feltz P, Headley PM, Worms P, Lloyd KG, Bartholini G (1980) New anticonvulsants: Schiff bases of gamma-aminobutyric acid and gamma-aminobutyramide. J Med Chem 23(6):702–704. doi:10.1021/jm00180a029
Kuzmin VE, Artemenko AG, Lozytska RN, Fedtchouk AS, Lozitsky VP, Muratov EN, Mescheriakov AK (2005) Investigation of anticancer activity of macrocyclic Schiff bases by means of 4D-QSAR based on simplex representation of molecular structure. SAR QSAR Environ Res 16(3):219–230
Mackie, McCartney (1989) In: Collee JG, Duguid JP, Fraser AG, Marmion BP (eds) Practical medicinal microbiology, 13th edn. Churchill Livingstone, London, p 87
Mathew V, Keshavayya J, Vaidya VP, Giles D (2007) Studies on synthesis and pharmacological activities of 3,6-disubstituted-[1,2,4]-triazolo-[3,4-b]-[1,3,4]thiadiazoles and their dihydro analogues. Eur J Med Chem 42:823–840. doi:10.1016/j.ejmech.2006.12.010
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Meth 65:55–63
Patole J, Shingnapurkar D, Padhye S, Ratledge C (2006) Schiff base conjugates of p-aminosalicylic acid as antimycobacterial agents. Bioorg Med Chem Lett 16:1514–1517. doi:10.1016/j.bmcl.2005.12.035
Ragenovic KC, Dimova V, Kakurinov V, Molnar DG, Buzarovaska A (2001) Synthesis, antibacterial and antifungal activity of 4-substituted-5-aryl-[1,2,4]-triazoles. Molecules 6:815–824. doi:10.3390/61000815
Ren S, Wang R, Komatsu K, Bonaz-Krause P, Zyrianov Y, McKenna CE, Csipke C, Tokes ZA, Lien EJ (2002) Synthesis, biological evaluation and quantitative structure activity relationship analysis of new Schiff bases of hydroxysemicarbazide as potential antitumor agents. J Med Chem 45:410–419
Ronad P, Dharbamalla S, Hunshal R, Maddi V (2008) Synthesis of novel substituted 7-(benzylideneamino)-4-methyl-2H-chromen-2-one derivatives as anti-inflammatory and analgesic agents. Arch Pharm 341(11):696–700
Shi L, Ge HM, Tan SH, Li HQ, Song YC, Zhu HL, Tan RX (2007) Synthesis and antimicrobial activities of Schiff bases derived from 5-chloro-salicylaldehyde. Eur J Med Chem 42(4):558–564. doi:10.1016/j.ejmech.2006.11.010
Sridhar SK, Pandeya SN, Stables JP, Ramesh A (2002) Anticonvulsant activity of hydrazones, Schiff and Mannich bases of isatin derivatives. Eur J Pharm Sci 16(3):129–132. doi:10.1016/S0928-0987(02)00077-5
Verma M, Pandeya SN, Singh KN, Stables JP (2004) Anticonvulsant activity of Schiff bases of isatin derivatives. Acta Pharm 54(1):49–56
Wu J, Liu X, Cheng X, Cao Y, Wang D, Li Z (2007) Synthesis of novel derivatives of 4-amino-3-(2-furyl)-5-mercapto-[1,2,4]-triazole as potential HIV-1 NNRTIs. Molecules 12:2003–2016. doi:10.3390/12082003
Acknowledgments
We are grateful to the Management, SeQuent Scientific Ltd., New Mangalore. The authors are also thankful to Prof. A. Srikrishna, Department of Organic Chemistry, IISc, Bangalore for providing 1H NMR and 13C NMR spectral facilities. The authors also thank the Department of Pharmacology, Manipal College of Pharma science, Manipal University for the analysis of cytotoxicity.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sumangala, V., Poojary, B., Chidananda, N. et al. Synthesis and biological evaluation of some Schiff bases of 4-amino-5-(4-methylsulfonyl)benzyl-2,4-dihydro-3H-[1,2,4]-triazole-3-thione. Med Chem Res 22, 2921–2928 (2013). https://doi.org/10.1007/s00044-012-0294-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-012-0294-5