Abstract
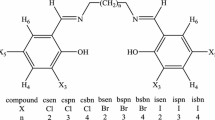
By the reaction of 5-(4-aminophenyl)-1,3,4-oxadiazolin-2-thione with alkyl halides 2-alkylthio-5-(4-aminophenyl)-1,3,4-oxadiazoles, 2–4 were obtained. By the reaction of compounds 2–4 with aromatic aldehydes, new Schiff bases 5a–d and 6a–d were synthesized. The structures of synthesized compounds are confirmed by the IR, UV, 1H NMR for all compounds, 13C NMR for compounds 3, 4, 5a–5d, 6a, 6d and X-ray for compound 3. All substances were tested in vitro for their antibacterial and antifungal activity. The results showed that Compound 2 exhibited remarkable activity against Candida albicans. Compounds 5а–d were found to exhibit a weak selective activity against Gram-positive bacteria of Bacillus subtilis and compounds 6а–d against Gram-negative bacteria of Escherichia coli.




Similar content being viewed by others
References
R.A. Rane, P. Bangalore, S.D. Borhade, P.K. Khandare, Eur. J. Med. Chem. 70, 49 (2013)
R. Kharb, R. Kaur, A.K. Sharma, Eur. J. Biomed. Pharm. Sci. 1, 401 (2014)
R. Sharma, N. Kumar, R. Yaday, Res. Rev. J. Chem. 4, 1 (2015)
P. Dholaria, K. Parikh, J. Deepkumar, Int. J. ChemTech Appl. 4, 1 (2015)
S. Rasool, M.A. Aziz-ur-Rehman, S.Z. Abbasi, S.A. Siddiqui, A. Shah, BJPS 52, 471 (2016)
A. Siddiqa, et al., Pak. J. Pharm. Sci. 29, 27 (2016)
D.S. Ismailova, A.A. Ziyaev, B.Z. Elmuradov, T.T. Toshmurodov, Kh.M. Bobakulov, R.P. Zakirova, J. Basic Appl. Res. 2, 476 (2016)
A.A. Ziyaev, I.F. Tozhiev, S.A. Sasmakov, S.S. Azimova, B.Z. Elmuradov, D.S. Ismailova, Pharm. Chem. J. 4, 29 (2017)
G.G. Galust’yan, A.A. Ziyaev, Chem. Heterocycl. Compd. 38, 1104 (2002)
A.A. Ziyaev, I.F. Tozhiev, Kh.M. Shakhidoyatov, Chem. Heterocycl. Compd. 48, 488 (2012)
Clinical and Laboratory Standards Institute (CLSI) Performance Standards for Antimicrobial Disk Diffusion Susceptibility Tests, approved standard, 19th edn. (CLSI Document M100-S19, Wayne, 2009)
T. Lindl, J. Bauer, Zell und Gewebekultur (Gustav-Fischer-Verlag Jena, Berlin, 1989)
DIN Taschenbuch 222, Medizinische Mikrobiologie und Immunologie (Beuth-Verlag, Berlin, 2004)
CrysAlisPro, Oxford Diffraction. (Oxford Diffraction Ltd, Abingdon, 2007)
G.M. Sheldrick, Acta Crystallogr. A 64, 112 (2008)
E.S. Al-Abdullah, S.S.H.R. Sebastian, R.I. Al-Wabli, A.A. El-Emam, C.Y. Panicker, C. Van Alsenoy, Spectrochim. Acta A. 133, 605 (2014)
S. Vijayaraghavan, R.R. Somani, P.Y. Shirodkar, V.J. Kadam, Int. J. PharmTech. Res. 1, 811 (2009)
C. Estarellas, D. Escudero, A. Frontera, D. Quinonero, P. Deya, Theor. Chem. Acc. 122, 325 (2009)
Acknowledgements
Authors are thankful to the Academy of Sciences of the Republic of Uzbekistan for supporting this study (Grant VA-FA-F7-006 and FA-F6-T198).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest associated with the authors of this paper.
Rights and permissions
About this article
Cite this article
Ismailova, D.S., Ziyaev, A.A., Bobakulov, K.M. et al. The new Schiff bases of 2-alkylthio-5-(4-aminophenyl)-1,3,4-oxadiazoles and their antimicrobial activity. J IRAN CHEM SOC 16, 545–551 (2019). https://doi.org/10.1007/s13738-018-1530-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-018-1530-9