Abstract
Bioethanol is recognized as a valuable substitute for renewable energy sources to meet the fuel and energy demand of the nation, considered an environmentally friendly resource obtained from agricultural residues such as sugarcane bagasse, rice straw, husk, wheat straw and corn stover. The energy demand is sustained using lignocellulosic biomass to produce bioethanol. Lignocellulosic biomass (LCBs) is the point of attention in replacing the dependence on fossil fuels. The recalcitrant structure of the lignocellulosic biomass is disrupted using effective pretreatment techniques that separate complex interlinked structures among cellulose, hemicellulose, and lignin. Pretreatment of biomass involves various physical, chemical, biological, and physiochemical protocols which are of importance, dependent upon their individual or combined dissolution effect. Physical pretreatment involves a reduction in the size of the biomass using mechanical, extrusion, irradiation, and sonification methods while chemical pretreatment involves the breaking of various bonds present in the LCB structure. This can be obtained by using an acidic, alkaline, ionic liquid, and organosolvent methods. Biological pretreatment is considered an environment-friendly and safe process involving various bacterial and fungal microorganisms. Distinct pretreatment methods, when combined and utilized in synchronization lead to more effective disruption of LCB, making biomass more accessible for further processing. These could be utilized in terms of their effectiveness for a particular type of cellulosic fiber and are namely steam explosion, liquid hot water, ammonia fibre explosion, CO2 explosion, and wet air oxidation methods. The present review encircles various distinct and integrated pretreatment processes developed till now and their advancement according to the current trend and future aspects to make lignocellulosic biomass available for further hydrolysis and fermentation.
Similar content being viewed by others
Introduction
Agricultural waste is one of the most abundant lignocellulosic biomasses available in India and an attractive alternative to renewable energy generation. Renewable energy generated from agricultural biomasses has the possibility of substituting fossil fuel generation [1]. But due to a lack of awareness, these are burned and dumped in the open environment leading to high greenhouse gas (GHG) emissions. Many countries have planned to reduce GHG emissions by switching to cleantech sources, i.e., ethanol. Ethanol is produced from lignocellulosic waste, the most abandoned renewable biomass, derived from agricultural feedstock such as wheat husk, rice straw, sugarcane bagasse, and corn stover. This organic waste is derived from biological sources, primarily plant biomass, being the most readily available global source of renewable materials, with an estimated annual worldwide production of 1010 MT. On evaluating the total production of various agricultural residues globally, the sugarcane bagasse (SCB) biomass is considered one of the abundant agricultural residues that hold the key to solving the global energy problem and environmental concern [2]. According to recent research, the potential for lignocellulose biomass contributed from SCB worldwide annually is 243 million tonnes, which translates to producing 4.3 EJ of energy, which covers 6.8% of the present global supply of bioenergy [3]. Similarly, corn stover production in 2021–22 was approximately 120 MT globally and has the potential to produce 23–53 billion tons of bioethanol in the United States alone [4]. Approximately 512.8 MT of rice is produced globally every year [5] and according to IRRI, the typical rice grain to straw production ratio is 0.7:1.4 [6]. Thus, it is estimated to produce 1025.6 MT of straw that is burned by the local farmers which if utilized rationally could add up to the global bioethanol production [7]. Among these agricultural waste obtained from food crops, peanut shell is considered bulky waste, producing about 230–300 gm/kg of peanuts with 50.34 MMT of peanut produced worldwide in the 2021–2022 season which can also add up to the cause [8]. It could be easily comprehended that utilizing agricultural residue for ethanol production could be one of the most promising sustainable energy processes due to unending supplies of available lignocellulosic biomass wastes. Global biofuel production relative to different countries in the year 2021 is illustrated in Fig. 1, taking reference from Statista report on world biofuel production by various countries [9] and bioethanol production influence with both positive and negative impact is illustrated in Fig. 2.
Lignocellulosic biomass has complex biochemical and highly heterogeneous structures, characterized by using both chemical and physical structural properties. The structural properties of biomass which include chemical composition, fiber characterization, and cell proposition show a significant effect on the further saccharification process [10]. It is concluded that feedstock with higher cellulose, hemicellulose, lower lignin, and silica content is suitable for bioethanol production. It is estimated that the biochemical structure with the compositional analysis of cellulose (32–47%), hemicellulose (19–27%), and lignin (5–24%) are suitable feedstock for bioethanol production. Since most agricultural wastes contain ≥ 50% fermentable sugars but due to their recalcitrant structure, it is not used further for any chemical and biological process to ferment sugar [11]. However, to make it suitable for further process of hydrolysis and fermentation, a prerequisite step, i.e., pretreatment is required. An ideal pretreatment step dwindles the connective link between lignocellulosic recalcitrant structure and makes feedstock available for further process, i.e., enzymatic accessibility and saccharification process with less inhibitor formation and increase in the recovery rate of cellulose and hemicellulose [12]. The process can be cost-effective by deploying advanced techniques of pretreatment. According to various reports, effective pretreatment reduces the size of the biomass, minimizes sugar loss, and maximizes lignin removal along with a reduction in the formation of inhibitors, thereby making the process economical.
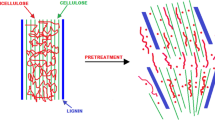
Pretreatment is required to disintegrate the lignin structure and to make the cellulosic complex more accessible for hydrolysis by enhancing enzyme accessibility. Pretreatment is used to reinforce the accessibility of biomass for the conversion of cellulose to glucose, thus making it more accessible to the enzymatic action by hydrolysis of hemicellulosic content and by solubilization of lignin content in the biomass [13]. Figure 3 depicts the diagrammatic representation of the production of bioethanol through various processes along with the cellulase effect on lignocellulosic biomass.
This review paper covers various pretreatment techniques with an integrated approach toward the degradation of the recalcitrant structure of biomass. Various distinct pretreatment methods are examined in this review along with integrated pretreatment approaches with emphasis on the effect of pretreatment on numerous lignocellulosic biomass. There is a huge need to produce bioethanol in a cost-efficient manner and make it available for commercial purposes. It has been earlier formulated that pretreatment and hydrolysis are relatively costlier processes. The main concern remains regarding the strategy that should be adopted to make biomass affordable for further processing towards ethanol production, which can act as a replacement for petroleum-based fuel that will further solve issues related to dependency on petroleum fuels and provide flexibility in the operation.
Lignocellulosic biomass structure
Lignocellulosic biomass is a complex structure consisting of fermentable and non-fermentable sugar. Cellulose is the most abundant LCB (lignocellulosic biomass) with compositional analysis of 33–47% that is utilized for further process of hydrolysis [14]. Another copious compound in the LCB is hemicellulose (19–27%) in composition. Non-fermentable part of biomass are lignin (5–24%) and silica (18.3%) component which forms lignin–carbohydrates complex and hinders the further process of hydrolysis by binding with cellulose, reducing the exposed surface area for enzymatic action [15] as well as forms a hindrance against external encroachment and prevents degradation. Both hemicellulose and lignin form cover over a cellulosic portion of biomass and reduce the efficiency of enzymatic hydrolysis and fermentation which ultimately lowers the product yield. It is a prerequisite to have the region-wise analysis of biomass as LCB (lignocellulosic biomass), as a versatile resource not only used for biofuel production, but also turned out to account for the production of varied profit-based industrial products. With its high economic value, it is required to estimate the economic viability of the biofuel industry [16].
Cellulose
The largest carbohydrate constituent of LCB (lignocellulosic biomass) is a polymer of anhydrous-D-glucose with a lengthy structural chain constituent of β-glucose monomers having an affinity with β-(1,4)-glycosidic bond and gathered together into microfibril bundle [17]. The linear cellulosic chain is associated together with inter- and intramolecular H-bonds presenting a different degree of polymerization. This H-bond forms a highly ordered crystalline region that makes it accessible for the activity of the hydrolytic enzyme [18]. Some regions in the cellulosic structure are less crystalline–amorphous regions that make it resistant to biodegradation and the enzyme can easily bind to cellulose in these regions to start the hydrolysis process. It has been visualized that feedstock with more cellulosic content is accessible for bioethanol production.
Hemicellulose
Hemicellulose is the group of polysaccharides consisting of a short-branched chain of sugars such as arabino-glucouronoxylan, arabino-4-O-methyl-glucuronic- xylan, glucurono-xylan, arabino-xylan, and galactic-arabino-glucurono-xylan. In another word, it is the polymer edifice of both hexose sugars (D-glucose, D-mannose, and D-galactose) and pentose sugars (D-xylose and L-arabinose) and acetylated sugars [17]. It is a random structure containing five or six-carbon sugar. It is the second most abandoned polymer located in the secondary cell wall of plants.
The main hemicellulose in the plant cell wall is in the form of xylan, which gets converted into its by-product xylose in the hydrolysis process utilized for strain development in biomass [19]. Thus, acetylation frequently takes place during the biosynthesis of galactose residue and another by-product such as acetic acid formed by hydrolysis of hemicellulose which inhibits microbial growth and ethanol fermentation [15]. Thus, to inhibit the formation of by-products, required to maintain the temperature and retention time of hemicellulose degradation. Due to its branched-chain structure with a short lateral chain and low molecular weight, hemicellulose can easily be hydrolyzed [20].
Lignin
It is a complex and large molecular structure, mainly formed by three types of monomers such as p-coumarin, sinapyl alcohols, and coniferyl, which are combined to form integrated and highly interlinked structure, has a high ambivalence, which is responsible for the hardness of structure [21]. It is barren of a sugar-based edifice having a 3D structure that possesses an alkyl-aryl bond among cellulose and hemicellulose moiety embedded in it and acts like an adhesive between them. Typically, the presence of lignin reduces the efficiency of enzymatic hydrolysis. Through electrostatic, hydrophobic, and H-bond interactions, lignin may bind enzymes, and the discharge of chemicals that are soluble lignin-derived may serve as harmful enzyme inhibitors [22].
Pretreatment processes often break down the hemicellulose polymer that links the cellulose molecules into fibres. A portion of the cellulose fibres may also be broken by pretreatment, especially in the amorphous areas. In the ensuing hydrolysis processes, the elimination of the lignin and hemicellulose improves the access of the hydrolytic reagents to the cellulose molecules. However, several physical, biological, chemical, and physiochemical pretreatment are enacted to loosen the strong interactions among these LCB (lignocellulosic biomass) and remove lignin for increasing accessibility of carbohydrates for further process of ethanol production [23]. Figure 4 depicts the widely varying composition of commonly available lignocellulosic sources such as rice straw, wheat straw, and SCB. The first step towards the utilization of LCBs is the disruption of the natural boundaries to extract the cellulose and hemicellulose, which become the substrate for the further process of saccharification. At present, this approach is to break the barrier of LCB degradation through pretreatment that can eliminate lignin and hemicellulose along with rupturing linkage with cellulose to destruct its crystalline structure and degree of polymerization [24]. It was shown that using 2% NaOH (sodium hydroxide) at 121 °C for 1 h removed the lignin content with a slight effect on cellulose and hemicellulose as compared with increasing concentration of H2SO4 in which cellulose and hemicellulose content increased while reversed with lignin content. Thus, using acid pretreatment, hemicellulose can easily be hydrolyzed [25], and further, it is required to evaluate the correct compositional analysis of lignocellulosic biomass for maximum conversion yield and to determine the economic process of bioethanol conversion. There are some methods for compositional analysis of LCBs, these are sulfuric acid hydrolysis method, kinetic analysis methods, and near-infrared spectroscopy methods [23].
The pretreatment methods show the following effect on the lignocellulosic biomass by comparing its pretreatment efficiency both before and after the pretreatment process. The pretreatment is considered to disrupt the compositional analysis of the biomass and enhances the adaptation towards available biomass with the main emphasis on particle size, and degradation of lignin, hemicellulose, and cellulose for subsequent processing. This will enhance the formation of reducing sugar and compatibility towards fermentation, further morphological analysis using XRD, TGA, FESEM, and FTIR spectra show the variation in the structural composition of biomass both before and after the pretreatment process. The efficient pretreatment has minimum sugar degradation with a slight formation of toxic compounds. The pretreatment is the pre-requisite step towards bioethanol production and its effect on feedstock are size reduction, and cellulose disruption along with hemicellulose and lignin depolarization are illustrated in the supplementary file (Additional file 1: Figure S1).
It is quite impossible to follow the strict criteria of each pretreatment, some of the compromises can be made by associating various merits of unusual pretreatment and employing hybrid pretreatment techniques with a maximum yield of desired products. While combining these processes has increased the production cost as well as the complexity of the methods. Nevertheless, to overcome these effects, some pretreatment methods with their mechanism along with some of the hybrid forms of pretreatment methods have been discussed. The main available pretreatment techniques performed on lignocellulosic biomass, a path towards the conversion of affordable biomass available for further processing of saccharification and fermentation is depicted in Fig. 5.
Physical pretreatment processes
Physical pretreatment mainly centres on energy and strength by disrupting the lignin barrier in the lignocellulosic complex and making sugar available for conversion to biofuels [26]. This method creates variations in temperature and pressure simultaneously requiring high energy consumption and power that results in high production costs. This pretreatment involves microwave radiation, milling, extrusion, pyrolysis, and mechanical process [27]. It is carried out through mechanized size reduction, surface area, and crystallinity index of biomass which improves further downstream processing which is an energy and cost-intensive process.
Mechanical process
The mechanical process is the most vital step in the pretreatment process as it is meant for size reduction of the biomass along with disrupting biomass surface configuration by breaking the physical structure of feedstock. The cellulose crystallinity of agricultural waste is reduced through milling methods, which may be in the form of ball milling, wet disk milling, roll milling, grinding, and chip** processes [28]. This process enables the complete conversion of cellulose into its amorphous form and makes it available for hydrolysis so that it can easily be attacked by the hydrolytic enzyme. Ball milling pretreatment (BMP) decreases the crystallinity and size of biomass, which slackens the interior structure of biomass. It increases the internal energy of the pretreated rice straw and decreases the stability of the hydrogen bond between lignocellulosic biomasses. One of the significant importance of ball milling is that no weight loss and no inhibitor formation during the fermentation process [29]. On the other hand, high energy consumption and low effectiveness of the process have hindered its further application over other processes. Moreover, different mechanical methods have a comparable effect on biomass based on its impact, compression, friction, and shear force. It is estimated that the vibratory milling process is more effective in reducing the size of biomass as well as the crystallinity of cellulose obtained from LCBs [30]. Water absorption at 400% (w/w) during ball milling at 80 °C for 30 min on corn stover biomass enhances the glucose yield up to 66.69% than at 100 °C along with ball milling process, thus, water intake during the milling process has increased its sustainability and made the process efficient at a commercial scale [31]. Using suitable conditions for the pretreatment process by chemo-mechanical method reduces energy consumption by up to 20–80%. Thus, combining the milling process with alkaline pretreatment, i.e., NaOH proves to be the best alternative for combined process by enhancing glucose yield up to 300 mg/g of SCB biomass, the highest among various alkaline and acidic pretreatment methods. By rupturing the ether linkages in lignin/phenolics carbohydrates complexes, alkaline pretreatment can effectively remove hemicelluloses without dissolving lignin [32]. Even little dosages of dilute alkali of 4% (w/w) NaOH assisted with ball milling and then hydrothermal pretreatment at 100 ℃ for 40 min, yielded 40.75% of reducing sugar with 20.08% of xylose from pretreated SCB biomass. In this instance, NaOH had a more significant role in the deacetylation process than that of the alkaline reagent, which led to the creation of enzyme inhibitors [33]. Similarly, the combined pretreatment method of wet disk milling and liquid hot water compression at 160 ℃ for 30 min along with autoclaving at 135 ℃ for 60 min of residence time have led to 90% of glucose yield while 79% of xylose yield. The significance of using liquid hot water compression is to dissolve a portion of hemicellulose and make a portion of rice straw available for the respective action of cellulase on the surface of biomass [34]. The critical drawback of using a mechanical process for lignocellulosic conversion is that it is an energy-intensive process to break the LCB structure [35]. Due to its high energy usage in large-scale manufacturing, LCB milling increases biofuel yield but is not cost-effective. A recent study found that mechanical pretreatment improved the disintegration of structural parts in two distinct stages by decreasing the trailing duration throughout anaerobic digestion (AD) thus increasing the biofuel output by up to 22%. So, to surmount this limitation, it is required to combine both chemical and mechanical pretreatment that has been proven to lower the energy consumption of milling and ultimately increase the efficiency of glucose yield.
Extrusion process
Nowadays, the combined process of pretreatment has increased attention. One such process is the extrusion process, which mainly includes a combination of mechanical, thermal, and chemical techniques simultaneously, leading to structural alteration due to force generated by high-power rotation. The rotation process generates shearing force among different components associated with the process, i.e., biomass, screw, and barrel, that lead to an increase in temperature and pressure of the barrel [24]. This rotation reduces biomass, efficient heat transfer, and ultimately leads to high sugar recovery from the biomass. The pretreatment process is performed radically at three reaction zone, namely conveying, reverse, and kneading. The conveying zone automatically squeezes the biomass and dispatches it to the kneading zone, where the catalyst gets mixed and forwards it to the next zone, i.e., the reverse, conveying zone where the reactor maintains the pressure required for the process. There is the continuous movement of biomass from the kneading zone to conveying zone for the pretreatment process [36]. It may be either single-gear or twin-gear extrusion-mediated pretreatment required for effective sugar recovery from various types of biomasses available. The process such as a twin-gear extruder is a promising way of pre-treating rice straw with high solid lignocellulose as it removes the amorphous region, leading to a rise in crystallinity index (CrI) up to 50% [37]. After this process, cellulose peaks become sharper, revealing an increase in glucan content ranging from 40.83% to 63.16% as well as the removal of lignin, i.e., 64.51%. Twin gear extrusion is a viable pretreatment method for lignocellulose with a high solid content in particular biomass [30]. The major optimum condition for the extrusion process is the material diameter (60 mesh), extrusion temperature (143 ℃), screw speed of 350 rpm, the reaction time of around 1.5 h; around 77.5% of cellulose and hemicellulose conversion. The findings suggest that cellulose and hemicellulose both were broken down as a result of extrusion, with hemicellulose losing more of its structural integrity than cellulose. This further suggests that cellulose is more challenging to disintegrate than hemicellulose. Additionally, it was discovered that extrusion pretreatment can result in a notable reduction in dietary fiber that is insoluble. The mechanical interruption of cell wall assembly caused by extrusion was likely caused by the combined impact of heat and shearing forces, which resulted in the breakdown of the lignocellulosic biomass structure [24]. The advantage of using the extrusion process is high continuous output, and economic feasibility, the product is obtained with no sugar degradation, under enhanced monitoring and control process with higher yield in a cost-effective manner. The aforementioned studies show that when the extrusion process combines with other pretreatment methods, it has a significant effect on the breakdown of cellulose and hemicellulose structure and enhances the total output of reducing sugars.
Irradiation
Microwave irradiation alters the complex structure of cellulose as well as degrades hemicellulose and lignin in LCBs (lignocellulosic biomass) material and increases the enzymatic vulnerability of biomass for ethanol production. It persuades the breakdown of LCB through molecular collisions such as blending and stretching by dielectric polarization on covalent bonds between cellulose and hemicellulose. This dielectric polar movement leads to rapid heating with an elevated frequency of approximately up to a million times per second which depresses the operation time. Next, the electron beam irradiation process proved to be significant for enzyme digestibility and increases the crystalline portion of available feedstock. It is estimated that glucose yield is theoretically 52.1% higher than the untreated rice straw obtained after 132 h of hydrolysis. Due to the bombardment of electron irradiation during EBI, the interior surface of the biomass was more exposed to enzymatic hydrolysis [29]. Similarly, the researcher comprehensively studied the consequence of microwaves on chemically pretreated feedstock. This type of radiation implies 1% (w/v) NaOH pretreated rice straw yielding 31.3% ethanol in a limited time as contrasted with the traditional alkali pretreatment method [29]. The chemical treatment anticipated before microwave pretreatment disrupts the crystalline cellulose and lignin solubilization. The maximum reducing sugar yield obtained is 246.34 mg/g, cellulose content is 17.53% when pretreated with 1% (w/v) NaOH at a frequency of around 2450 MHz for 5 min at 850 W and around 150 ℃ temperature. This showed the stretching of aromatic rings (C = O bond) corresponding to acetyl groups of hemicellulose that lead to the reduction of hemicellulose content in pretreated rice straw [15]. It has been found that yield from the irradiation process increases with the decrease in the size of biomass. Similarly, the application of microwave irradiation is considered the alternative to conventional barometers as it provides a shorter heating duration with better performance, along with its immediate stop-and-start application over the feedstock. Lower dosages were probably insufficient to significantly alter glucose production. The constituents of the lignocellulose are likely to break down with the increase in irradiation doses, leading to lower glucose production [29]. The optimum condition for performing microwave irradiation is 300 W of constant power generation for 5, 10, and 15 min at 372 kPa pressure. This produces 75% cellulose hydrolysis from rye and wheat stillages. It also shows that intensive microwave pretreatment, i.e., at 372 kPa for 10 min, increases the dehydration of reducing sugar produced as well as inhibits the fermentation process, and subsequently leads to a decrease in ethanol production [38]. Thus, it is necessary to scale up the biorefinery method for the economically efficient synthesis of ethanol from microwave–alkali–acid pretreated biomass.
Another effective ϒ-irradiation of 891 kGy was appropriate for the conversion of microcrystalline cellulose with the highest degree of crack and swelling in the biomass. This is an effective pretreatment technique as the increase in the doses of irradiation has increased the glucose from 0.01% to 0.65% as well as oligosaccharide from 0.04% to 26.78%. It showed that the cellulose underwent gamma irradiation, producing carbonyl and carboxy groups at the locations of bond breaking [39]. Thus, during the microwave irradiation process, thermal "hot spots" are created that lead microwave radiation to penetrate deep into the biomass, accelerating the interaction of ions with nearby molecules of LCBs. The silicified waxy surface was disrupted and the lignin–hemicellulose matrix was broken down as a result of the dipole molecules' rapid rotation, which generated a sharp rise in temperature [33].
Sonification
Ultrasound (sonification) is the advanced technique of pretreatment of LCBs (lignocellulosic biomass). Ultrasound-assisted pretreatment alters both the physical and chemical properties of biomass. This pretreatment process recognizes itself as an efficient and eco-friendly technique. This helps in the formation of subsequent bubbles that lead to the disruption of cellulose and hemicellulose recalcitrant structure along with an increase in the porosity of cellulosic content leading to its breakage into simple reducing sugar [15]. The deformation process of ultrasonic pretreatment reduces the lignin percentage, disrupts the biomass functional groups, and increases the crystallinity index along with an increase in the surface porosity and area of biomass. The sonification process was performed mainly at a frequency around 24 kHz and at an operating power of 400 W. Ultrasonic-assisted alkali pretreatment had been used desirably to boost the efficiency of alkali-treated biomass that can increase the lignin removal up to 80–85% and is performed with 0.5% (w/v) NaOH alkaline solution. This would consequently release more carbohydrates with fewer fermentation-inhibitory residues through the creation of Ultrasound induced cavitation [40]. Likewise, ultrasonic-assisted acid pretreatment has made its way adjacent to bioethanol production from waste potato mass. Further, it was assumed that the increase in sonification time from 5 to 10 min consequently, increases bioethanol yield up to 65.8 mg/l. Higher ultrasonic time, i.e., > 10 min disintegrates starch particles and releases the lignin in the cell wall that forbids the saccharification process and disfavors bioethanol output at a subsequent stage [41]. This ultrasonic wave of 20 kHz creates a disturbance, cavitation, or agitation in the chemistry of the lignocellulosic biomass structure. Cavitation in the biomass structure increases the mass transfer and reduction in the particle size of the exposed biomass. This process can be applied even at mild concentrations and showed regular cleavage of rice straw structure resembling rough and irregular surfaces. The microporous structure of interior biomass enables easy penetration of enzymes [42].
Chemical pretreatment processes
This pretreatment is mainly based on the use of chemicals in the procedure which transforms the crystalline structure of lignocellulosic mass into an amorphous form in need of energy requirement. For chemical pretreatment, it is required to maintain ambient temperature during the process which subsequently enhances the glucose yield for further process. Various chemicals have different abilities to break down the compact structure of lignocellulosic biomass. These are meant to disturb and break the hydrogen bond along with the covalent bond between cellulose, hemicellulose, and lignin. Thus, it is required to understand the mechanism of the process along with the advantages and disadvantages of the particular chemical pretreatment method (Table 1).
Alkaline pretreatment
Among chemical pretreatment methods, alkaline pretreatment was widely accepted as being a simple process and having a strong pretreatment effect over some time. This method selectively removes lignin from its carbohydrate counterparts and expands the surface area as well as the porosity of the biomass, decrease in the polymerization degree and crystallinity, resulting in enhancing enzymatic hydrolysis [21]. The chemicals used under alkaline pretreatment are non-corrosive and non-pollutants such as sodium hydroxide (NaOH), ammonia and lime (calcium hydroxide, Ca(OH)2), sulfite, and ammonium hydroxide [43]. The common effect regarding alkaline pretreatment is that it raises the digestibility of lignocellulosic biomass, which can be obtained by transforming the intricate lignin–hemicellulose network enhancing lignin removal. With rupturing cellulose's H-bonds and enabling amorphous cellulose more soluble at higher temperatures or longer residence times, high-severity conditions cause the thermochemical alterations [44]. Experimental analysis reveals that NaOH is the most effective pretreatment which breaks the intercellular bond between cellulose and another component (lignin and hemicellulose). NaOH pre-treated biomass causes the lignin to break down by solubilizing the lignin carbohydrate bond and increases the surface area of cellulose structure while minimizing the degree of crystallinity and polymerization, carried out at low temperature and pressure [45]. In alkaline conditions, lignin's alkyl-aryl bonds are easily disrupted for enzymatic activity.
In a comparison of various alkaline pretreatment processes, alkaline peroxide is best as it increases the fermentation yield by solubilizing lignin effectively and increases the digestibility of feedstock required for further process. Alkaline peroxide used for the process was 5% H2O2 solid concentration at 50 ℃ for 1 h performed on rape seed straw which results in higher enzyme digestibility [46]. This alkaline peroxide treatment before alkaline pretreatment is performed at mild conditions effectively destructing the structure of biomass while facilitating isolation of lignin from the complex recalcitrant structure of the macromolecules. The peroxide loading carried out at 80 mg/g of pretreated wheat straw results in 59.9% lignin removal due to the degradation of lignin during the process [47].
Alkaline peroxide pretreatment is combined with other two oxidizing reagents namely NaOH and HCL (37%) with H2O2 (33%) employing different thermo and thermochemical reactions by autoclaving both reagents used in the process. This shows around 74% hemicellulose solubilization with the release of 2.6% of glucose as compared with pretreatment methods performed alone. Since NaOH shows strong lignin removal and hemicellulose solubilization by breaking ester linkages, it is frequently used for lignocellulose pretreatment. This increases biomass porosity and deteriorates the polysaccharide chain and cellulosic content [48]. Similarly, pretreatment with 1% NaOH assisted with 1% of H2O2 treatment for 24 h results in total sugar of 0.17 g/g of dry biomass while reducing sugar is about 0.024 g/g of dry biomass after 48 h at ambient temperature. Furfural and other inhibitors of hemicellulose breakdown were not taken into consideration with most alkaline solutions perturbing and disintegrating the association of LCBs structure but do not destroy hemicellulose like acid pretreatment. Following alkaline pretreatment, the diversity of sugars found in the liquid fraction shows that NaOH can absorb a wide range of soluble compounds under moderate circumstances [49]. Likewise, NaOH catalyzed organosolvent pretreatment with 10% loading of NaOH along with 150 ml of ethanol/water at 60/40 (%v/v) ratio at 180 ℃ for 30 min resulted in the solid recovery of 81.2% along with lignin removal of 40.7% from SCB biomass. However, when the alkali concentration rises, more cellulose can be converted into oligosaccharides and subunits, reducing the amount of solid recovered and the amount of sugar produced [129].
Bacterial pretreatment
Biological pretreatment assisted with bacterial treatment showed higher lignin degradation than fungal pretreatment due to its easier genetic manipulation and its tolerance towards environmental conditions. Various bacteria such as Sphingobium sp. SYK-6, Rhodococcus sp., Ceriporiopsis sp., Pandoraea sp., galactomyces sp., and Mycobacterium sp. used for bacterial pretreatment show efficient lignin and polycyclic aromatic hydrocarbon degradation prior to cellulose. The bacterial strain Mycobacterium smegmatis L2-K2 grows in the medium containing glucose as the carbon source required during the process [ Second-generation (lignocellulosic) bioethanol production appears to be the most promising renewable feedstock for meeting Sustainable Development Goals. Several feedstock pretreatments have revealed process challenges in terms of yield and product inhibitors. However, the pretreatment of lignocellulosic biomass is a crucial step towards bioethanol production from available biomass due to the recalcitrant structure of LCBs. It is required for the delignification of biomass, i.e., the removal of lignin to make the availability of cellulose and hemicellulose for further processes of saccharification. Till now, the known pretreatment methods, i.e., physical, chemical, biological, and physiochemical approaches are enacted. Further advancement in these processes is required to develop the combined pretreatment for economically feasible processes. The main focus is to develop an efficient pretreatment to remove the non-fermentable part of lignocellulosic biomass to get fermentable sugar. Subsequently, the combined process of pretreatment will lessen the incubation time for the process with more efficient desired outcomes. Thus, this will shorten the pretreatment time as well as it will help in develo** various new combined pretreatment processes at the required temperature, pH and retention time. This review article is based on current and future aspects of various combined pretreatment processes performed on available feedstocks. This will help the researcher in further planning, selection and development of an effective pretreatment process that will disintegrate the recalcitrant structure of lignocellulosic biomass.Conclusion
Availability of data and materials
All data analysed are included within this article.
Abbreviations
- LCBs:
-
Lignocellulosic biomass
- MT:
-
Million tonnes
- GHG:
-
Greenhouse gases
- BMP:
-
Ball milling pretreatment
- FTIR:
-
Fourier transform infrared spectroscopy
- CrI:
-
Crystallinity index
- KU:
-
KOH/urea
- SCB:
-
Sugarcane bagasse
- PEG:
-
Polyethylene glycerol
- IL:
-
Ionic liquid
- AFEX:
-
Ammonium fibre explosion
- SAA:
-
Soaking in aqueous ammonia
- EA:
-
Extractive ammonia
- LHW:
-
Liquid hot water
- WAO:
-
Wet air oxidation
- SEKOH:
-
Steam explosion assisted with potassium hydroxide
- NaOH:
-
Sodium hydroxide
- H2SO4 :
-
Sulphuric acid
- HCl:
-
Hydrochloric acid
- CO2 :
-
Carbon dioxide
- HMF:
-
Hydroxymethyl furfural
References
Kaur J, Chugh P, Soni R, Kumar S. Bioresource Technology Reports A low-cost approach for the generation of enhanced sugars and ethanol from rice straw using in-house produced cellulase-hemicellulase consortium from. Bioresour Technol Rep. 2020;11:100469. https://doi.org/10.1016/j.biteb.2020.100469.
Ajala EO, Ighalo JO, Ajala MA, Adeniyi AG, Ayanshola AM. Sugarcane bagasse : a biomass sufficiently applied for improving global energy, environment and economic sustainability. Bioresour Bioprocess. 2021. https://doi.org/10.1186/s40643-021-00440-z.
Pan S, Zabed HM, Wei Y, Qi X. Technoeconomic and environmental perspectives of biofuel production from sugarcane bagasse: current status, challenges and future outlook. Ind Crops Prod. 2022;188:115684.
Ruan Z, Wang X, Liu Y, Liao W. Corn. Integr Process Technol Food Agric. 2019. https://doi.org/10.1016/B978-0-12-814138-0.00003-4.
FAO Cereal Supply and Demand Brief | World Food Situation | Food and Agriculture Organization of the United Nations. [cited 2023 Jan 14]. https://www.fao.org/worldfoodsituation/csdb/en/
Rice Straw Management | International Rice Research Institute [Internet]. [cited 2023 Jan 12]. https://www.irri.org/rice-straw-management
Molaverdi M, Karimi K, Mirmohamadsadeghi S. Improvement of dry simultaneous saccharification and fermentation of rice straw to high concentration ethanol by sodium carbonate pretreatment. Energy. 2019;167:654–60. https://doi.org/10.1016/j.energy.2018.11.017.
Dukhnytskyi B. World agricultural production. Ekon APK. 2019. p. 59–65.
Fuel ethanol production in major countries 2021 | Statista [Internet]. [cited 2023 Jan 19]. https://www.statista.com/statistics/281606/ethanol-production-in-selected-countries/
Bhattacharyya P, Bhaduri D, Adak T, Munda S, Satapathy BS, Dash PK, et al. Characterization of rice straw from major cultivars for best alternative industrial uses to cutoff the menace of straw burning. Ind Crops Prod. 2020;143:111919. https://doi.org/10.1016/j.indcrop.2019.111919.
Shukla A, Arora R. Cost-effective production of environment sustainable bioethanol from paddy straw by various pretreatment methods at large scale. J emerg innov res. 2019;6:602–14.
Kumar A, Jain KK, Singh B. Process optimization for chemical pretreatment of rice straw for bioethanol production. Renew Energy. 2020. https://doi.org/10.1016/j.renene.2020.04.052.
Jamil F, Aslam M, Al-Muhtaseb AH, Bokhari A, Rafiq S, Khan Z, et al. Greener and sustainable production of bioethylene from bioethanol: Current status, opportunities and perspectives. Rev Chem Eng. 2022;38:185–207.
Singh R, Srivastava M, Shukla A. Environmental sustainability of bioethanol production from rice straw in India: A review. Renew Sustain Energy Rev. 2016;54:202–16. https://doi.org/10.1016/j.rser.2015.10.005.
Akhtar N, Goyal D, Goyal A. Characterization of microwave-alkali-acid pre-treated rice straw for optimization of ethanol production via simultaneous saccharification and fermentation (SSF). Energy Convers Manag. 2017;141:133–44. https://doi.org/10.1016/j.enconman.2016.06.081.
Naresh Kumar M, Ravikumar R, Thenmozhi S, Ranjith Kumar M, Kirupa Shankar M. Choice of pretreatment technology for sustainable production of bioethanol from lignocellulosic biomass: bottle necks and recommendations. Waste Biomass Valoriz. 2019;10:1693–709. https://doi.org/10.1007/s12649-017-0177-6.
Sebayang AH, Masjuki HH, Ong HC, Dharma S, Silitonga AS, Mahlia TMI, et al. A perspective on bioethanol production from biomass as alternative fuel for spark ignition engine. RSC Adv. 2016;6:14964–92.
Raj T, Kapoor M, Gaur R, Christopher J, Lamba B, Tuli DK, et al. Physical and chemical characterization of various Indian agriculture residues for biofuels production. Energy Fuels. 2015;29:3111–8.
Rocha-Meneses L, Ferreira JA, Mushtaq M, Karimi S, Orupõld K, Kikas T. Genetic modification of cereal plants: a strategy to enhance bioethanol yields from agricultural waste. Ind Crops Prod. 2020;150:112408. https://doi.org/10.1016/j.indcrop.2020.112408.
Singh J, Suhag M, Dhaka A. Augmented digestion of lignocellulose by steam explosion, acid and alkaline pretreatment methods: a review. Carbohydr Polym. 2015;117:624–31. https://doi.org/10.1016/j.carbpol.2014.10.012.
Rezania S, Oryani B, Cho J, Talaiekhozani A, Sabbagh F, Hashemi B, et al. Different pretreatment technologies of lignocellulosic biomass for bioethanol production: an overview. Energy. 2020;199:117457. https://doi.org/10.1016/j.energy.2020.117457.
Liu Y, Zheng X, Tao S, Hu L, Zhang X, Lin X. Process optimization for deep eutectic solvent pretreatment and enzymatic hydrolysis of sugar cane bagasse for cellulosic ethanol fermentation. Renew Energy. 2021;177:259–67. https://doi.org/10.1016/j.renene.2021.05.131.
Cai J, He Y, Yu X, Banks SW, Yang Y, Zhang X, et al. Review of physicochemical properties and analytical characterization of lignocellulosic biomass. Renew Sustain Energy Rev. 2017;76:309–22.
Zhang Y, Li T, Shen Y, Wang L, Zhang H, Qian H, et al. Extrusion followed by ultrasound as a chemical-free pretreatment method to enhance enzymatic hydrolysis of rice hull for fermentable sugars production. Ind Crops Prod. 2020;149:112356. https://doi.org/10.1016/j.indcrop.2020.112356.
** X, Song J, Liu GQ. Bioethanol production from rice straw through an enzymatic route mediated by enzymes developed in-house from Aspergillus fumigatus. Energy. 2020;190:116395. https://doi.org/10.1016/j.energy.2019.116395.
da Silva ARG, Errico M, Rong BG. Systematic procedure and framework for synthesis and evaluation of bioethanol production processes from lignocellulosic biomass. Bioresour Technol Rep. 2018;4:29–39. https://doi.org/10.1016/j.biteb.2018.08.015.
Carrillo-Nieves D, Rostro Alanís MJ, de la Cruz QR, Ruiz HA, Iqbal HMN, Parra-Saldívar R. Current status and future trends of bioethanol production from agro-industrial wastes in Mexico. Renew Sustain Energy Rev. 2019;102:63–74.
Yuan L, Chen Z, Zhu Y, Liu X, Liao H, Chen D. One step conversion of wheat straw to sugars by simultaneous ball milling, mild acid, and fungus Penicillium simplicissimum treatment. Appl Biochem Biotechnol. 2012;167:39–51.
Bak JS, Ko JK, Han YH, Lee BC, Choi IG, Kim KH. Improved enzymatic hydrolysis yield of rice straw using electron beam irradiation pretreatment. Bioresour Technol. 2009;100:1285–90. https://doi.org/10.1016/j.biortech.2008.09.010.
Zhang H, Zhang P, Ye J, Wu Y, Liu J, Fang W, et al. Comparison of various pretreatments for ethanol production enhancement from solid residue after rumen fluid digestion of rice straw. Bioresour Technol. 2018;247:147–56. https://doi.org/10.1016/j.biortech.2017.09.065.
Gu YM, Kim H, Sang B-I, Lee JH. Effects of water content on ball milling pretreatment and the enzymatic digestibility of corn stover. Water-Energy Nexus. 2018;1:61–5. https://doi.org/10.1016/j.wen.2018.07.002.
Sambusiti C, Licari A, Solhy A, Aboulkas A, Cacciaguerra T, Barakat A. One-Pot dry chemo-mechanical deconstruction for bioethanol production from sugarcane bagasse. Bioresour Technol. 2015;181:200–6. https://doi.org/10.1016/j.biortech.2015.01.058.
Huang J, Zhu Y, Liu T, Sun S, Ren J, Wu A, et al. A novel wet-mechanochemical pretreatment for the efficient enzymatic saccharification of lignocelluloses: Small dosage dilute alkali assisted ball milling. Energy Convers Manag. 2019;194:46–54. https://doi.org/10.1016/j.enconman.2019.04.078.
Hideno A, Inoue H, Yanagida T, Tsukahara K, Endo T, Sawayama S. Combination of hot compressed water treatment and wet disk milling for high sugar recovery yield in enzymatic hydrolysis of rice straw. Bioresour Technol. 2012;104:743–8. https://doi.org/10.1016/j.biortech.2011.11.014.
Dahunsi SO. Mechanical pretreatment of lignocelluloses for enhanced biogas production: methane yield prediction from biomass structural components. Bioresour Technol. 2019;280:18–26. https://doi.org/10.1016/j.biortech.2019.02.006.
Cha YL, Yang J, Seo SI, An GH, Moon YH, You GD, et al. Alkaline twin-screw extrusion pretreatment of Miscanthus with recycled black liquor at the pilot scale. Fuel. 2016;164:322–8. https://doi.org/10.1016/j.fuel.2015.10.006.
Ahmed MA, Rehman MSU, Terán-Hilares R, Khalid S, Han JI. Optimization of twin gear-based pretreatment of rice straw for bioethanol production. Energy Convers Manag. 2017;141:120–5. https://doi.org/10.1016/j.enconman.2016.06.022.
Mikulski D, Kłosowski G. Microwave-assisted dilute acid pretreatment in bioethanol production from wheat and rye stillages. Biomass Bioenergy. 2020;136:105528.
Liu Y, Zhou H, Wang S, Wang K, Su X. Comparison of c -irradiation with other pretreatments followed with simultaneous saccharification and fermentation on bioconversion of microcrystalline cellulose for bioethanol production. Bioresour Technol. 2015;182:289–95. https://doi.org/10.1016/j.biortech.2015.02.009.
Muthuvelu KS, Rajarathinam R, Kanagaraj LP, Ranganathan RV, Dhanasekaran K, Manickam NK. Evaluation and characterization of novel sources of sustainable lignocellulosic residues for bioethanol production using ultrasound-assisted alkaline pre-treatment. Waste Manag. 2019;87:368–74. https://doi.org/10.1016/j.wasman.2019.02.015.
Suresh T, Sivarajasekar N, Balasubramani K, Ahamad T, Alam M, Naushad M. Process intensification and comparison of bioethanol production from food industry waste (potatoes) by ultrasonic assisted acid hydrolysis and enzymatic hydrolysis: Statistical modelling and optimization. Biomass Bioenergy. 2020;142:105752. https://doi.org/10.1016/j.biombioe.2020.105752.
Rehman MSU, Kim I, Kim KH. Optimization of sono-assisted dilute sulfuric acid process for simultaneous pretreatment and saccharification of rice straw. Int J Environ Sci. 2013;11(2):543–50.
Kim JS, Lee YY, Kim TH. A review on alkaline pretreatment technology for bioconversion of lignocellulosic biomass. Bioresour Technol. 2016;199:42–8. https://doi.org/10.1016/j.biortech.2015.08.085.
Bali G, Meng X, Deneff JI, Sun Q, Ragauskas AJ. The effect of alkaline pretreatment methods on cellulose structure and accessibility. Chemsuschem. 2015;8:275–9.
Zhao C, Zou Z, Li J, Jia H, Liesche J, Chen S, et al. Efficient bioethanol production from sodium hydroxide pretreated corn stover and rice straw in the context of on-site cellulase production. Renew Energy. 2018;118:14–24. https://doi.org/10.1016/j.renene.2017.11.001.
Karagöz P, Rocha IV, Özkan M, Angelidaki I. Alkaline peroxide pretreatment of rapeseed straw for enhancing bioethanol production by Same Vessel Saccharification and Co-Fermentation. Bioresour Technol. 2012;104:349–57.
Yuan Z, Wen Y, Li G. Production of bioethanol and value added compounds from wheat straw through combined alkaline/alkaline-peroxide pretreatment. Bioresour Technol. 2018;259:228–36. https://doi.org/10.1016/j.biortech.2018.03.044.
Toquero C, Bolado S. Effect of four pretreatments on enzymatic hydrolysis and ethanol fermentation of wheat straw. Influence of inhibitors and washing. Bioresour Technol. 2014;157:68–76. https://doi.org/10.1016/j.biortech.2014.01.090.
Vu PT, Unpaprom Y, Ramaraj R. Impact and significance of alkaline-oxidant pretreatment on the enzymatic digestibility of Sphenoclea zeylanica for bioethanol production. Bioresour Technol. 2017. https://doi.org/10.1016/j.biortech.2017.09.012.
Zhang H, Zhang J, **e J, Qin Y. Effects of NaOH-catalyzed organosolv pretreatment and surfactant on the sugar production from sugarcane bagasse. Bioresour Technol. 2020;312:123601. https://doi.org/10.1016/j.biortech.2020.123601.
** Y, Shi Z, Xu G, Yang H, Yang J. A stepwise pretreatment of sugarcane bagasse by alkaline and hydroxymethyl reagent for bioethanol production. Ind Crops Prod. 2020;145:112136. https://doi.org/10.1016/j.indcrop.2020.112136.
**e X, Feng X, Chi S, Zhang Y, Yu G, Liu C, et al. A sustainable and effective potassium hydroxide pretreatment of wheat straw for the production of fermentable sugars. Bioresour Technol Reports. 2018;3:169–76. https://doi.org/10.1016/j.biteb.2018.07.014.
Wang W, Tan X, Imtiaz M, Wang Q, Miao C, Yuan Z, et al. Rice straw pretreatment with KOH / urea for enhancing sugar yield and ethanol production at low temperature. Ind Crop Prod. 2021;170:113776. https://doi.org/10.1016/j.indcrop.2021.113776.
Wang W, Tan X, Guo Y, Zhang B, Chen X, et al. Mild Urea/KOH pretreatment to enhance enzymatic hydrolysis of corn stover with liquid waste recovery for plant growth. J Clean Prod. 2021;284:125392. https://doi.org/10.1016/j.jclepro.2020.125392.
Chong G, Di J, Qian J, Wang C, He Y, Huo X, et al. Efficient pretreatment of sugarcane bagasse via dilute mixed alkali salts (K2CO3/K2SO3) soaking for enhancing its enzymatic saccharification. Process Biochem. 2018;68:121–30. https://doi.org/10.1016/j.procbio.2018.02.022.
Du S, Zhu X, Wang H, Zhou D, Yang W, Xu H. High pressure assist-alkali pretreatment of cotton stalk and. Bioresour Technol. 2013. https://doi.org/10.1016/j.biortech.2013.09.020.
Bernier-Oviedo DJ, Rincón-Moreno JA, Solanilla-Duqué JF, Muñoz-Hernández JA, Váquiro-Herrera HA. Comparison of two pretreatments methods to produce second-generation bioethanol resulting from sugarcane bagasse. Ind Crops Prod. 2018;122:414–21. https://doi.org/10.1016/j.indcrop.2018.06.012.
Zhang J, Zhou H, Liu D, Zhao X. Pretreatment of lignocellulosic biomass for efficient enzymatic saccharification of cellulose. Lignocellul Biomass Liq Biofuels. 2019. https://doi.org/10.1016/B978-0-12-815936-1.00002-2.
Liu Z, Wan X, Wang Q, Tian D, Hu J, Huang M, et al. Performances of a multi-product strategy for bioethanol, lignin, and ultra-high surface area carbon from lignocellulose by PHP (phosphoric acid plus hydrogen peroxide) pretreatment platform. Renew Sustain Energy Rev. 2021;150:111503. https://doi.org/10.1016/j.rser.2021.111503.
Yao F, Tian D, Shen F, Hu J, Zeng Y, Yang G, et al. Recycling solvent system in phosphoric acid plus hydrogen peroxide pretreatment towards a more sustainable lignocellulose biorefinery for bioethanol. Bioresour Technol. 2019;275:19–26. https://doi.org/10.1016/j.biortech.2018.12.040.
Yang Q, Huo D, Han X, Gu C, Hou Q, Zhang F, et al. Improvement of fermentable sugar recovery and bioethanol production from eucalyptus wood chips with the combined pretreatment of NH4Cl impregnation and refining. Ind Crops Prod. 2021;167:113503. https://doi.org/10.1016/j.indcrop.2021.113503.
Iram A, Cekmecelioglu D, Demirci A. Optimization of dilute sulfuric acid, aqueous ammonia, and steam explosion as the pretreatments steps for distillers ’ dried grains with solubles as a potential fermentation feedstock. Bioresour Technol. 2019;282:475–81. https://doi.org/10.1016/j.biortech.2019.03.009.
Mikulski D, Kłosowski G. Hydrotropic pretreatment on distillery stillage for efficient cellulosic ethanol production. Bioresour Technol. 2020;300:122661.
Cobalt(II) chloride hydrate | Cl2CoH2O—PubChem [Internet]. [cited 2023 Jan 16]. https://pubchem.ncbi.nlm.nih.gov/compound/Cobalt_II_-chloride-hydrate
Kumar B, Bhardwaj N, Verma P. Microwave assisted transition metal salt and orthophosphoric acid pretreatment systems: generation of bioethanol and xylo-oligosaccharides. Renew Energy. 2020;158:574–84. https://doi.org/10.1016/j.renene.2020.05.006.
Zhao X, Wen J, Chen H, Liu D. The fate of lignin during atmospheric acetic acid pretreatment of sugarcane bagasse and the impacts on cellulose enzymatic hydrolyzability for bioethanol production. Renew Energy. 2018;128:200–9. https://doi.org/10.1016/j.renene.2018.05.071.
Sorn V, Chang KL, Phitsuwan P, Ratanakhanokchai K, Di DC. Effect of microwave-assisted ionic liquid/acidic ionic liquid pretreatment on the morphology, structure, and enhanced delignification of rice straw. Bioresour Technol. 2019;293:121929. https://doi.org/10.1016/j.biortech.2019.121929.
Abdolmaleki A, Nabavizadeh SS, Badbedast M. 1-(Carboxymethyl)pyridinium chloride as an acidic ionic liquid for rice straw effective pretreatment. Renew Energy. 2021;177:544–53. https://doi.org/10.1016/j.renene.2021.05.158.
Husson E, Buchoux S, Avondo C, Cailleu D, Djellab K, Gosselin I, et al. Enzymatic hydrolysis of ionic liquid-pretreated celluloses: contribution of CP-MAS 13C NMR and SEM. Bioresour Technol. 2011;102:7335–42. https://doi.org/10.1016/j.biortech.2011.04.097.
Auxenfans T, Husson E, Sarazin C. Simultaneous pretreatment and enzymatic saccharification of (ligno) celluloses in aqueous-ionic liquid media: a compromise. Biochem Eng J. 2017;117:77–86. https://doi.org/10.1016/j.bej.2016.10.004.
Azizan A, Azmi IS, Darim RA, Jusri NAA, Jalil R, Abdul Rahman MF, et al. Lignocellulosic ionic liquid pretreated biomaterial/biomass. Mater Today Proc. 2020;46:1688–92. https://doi.org/10.1016/j.matpr.2020.07.368.
Poornejad N, Karimi K, Behzad T. Ionic liquid pretreatment of rice straw to enhance saccharification and bioethanol production. J Biomass Biofuel. 2014. https://doi.org/10.11159/jbb.2014.002.
Amoah J, Ogura K, Schmetz Q, Kondo A, Ogino C. Co-fermentation of xylose and glucose from ionic liquid pretreated sugar cane bagasse for bioethanol production using engineered xylose assimilating yeast. Biomass Bioenergy. 2019;128:105283. https://doi.org/10.1016/j.biombioe.2019.105283.
Nasirpour N, Mousavi SM. RSM based optimization of PEG assisted ionic liquid pretreatment of sugarcane bagasse for enhanced bioethanol production: Effect of process parameters. Biomass Bioenergy. 2018;116:89–98. https://doi.org/10.1016/j.biombioe.2018.06.008.
Jawad H, Madhab D, Murthy GS. A comparative account of glucose yields and bioethanol production from separate and simultaneous sacchari fi cation and fermentation processes at high solids loading with variable PEG concentration. Bioresour Technol. 2019;283:67–75. https://doi.org/10.1016/j.biortech.2019.03.060.
Koo BW, Kim HY, Park N, Lee SM, Yeo H, Choi IG. Organosolv pretreatment of Liriodendron tulipifera and simultaneous saccharification and fermentation for bioethanol production. Biomass Bioenergy. 2011;35:1833–40. https://doi.org/10.1016/j.biombioe.2011.01.014.
Joy SP, Kumar AA, Gorthy S, Jaganathan J, Kunappareddy A, Gaddameedi A, et al. Variations in structure and saccharification efficiency of biomass of different sorghum varieties subjected to aqueous ammonia and glycerol pretreatments. Ind Crops Prod. 2021;159:113072. https://doi.org/10.1016/j.indcrop.2020.113072.
Li X, Zheng Y. Lignin-enzyme interaction: Mechanism, mitigation approach, modeling, and research prospects. Biotechnol Adv. 2017;35:466–89. https://doi.org/10.1016/j.biotechadv.2017.03.010.
Ebrahimi M, Villaflores OB, Ordono EE, Caparanga AR. Effects of acidified aqueous glycerol and glycerol carbonate pretreatment of rice husk on the enzymatic digestibility, structural characteristics, and bioethanol production. Bioresour Technol. 2017;228:264–71. https://doi.org/10.1016/j.biortech.2016.12.106.
Chin DW, Lim S, Chin W, Fournier J, Le O, Rahman A. Investigation of organosolv pretreatment to natural microbial- investigation of international organosolv pretreatment natural degraded empty fruit bunch for sugar based substrate recovery degraded empty fruit bunc. Energy Proc. 2019;158:1065–71. https://doi.org/10.1016/j.egypro.2019.01.258.
Zhao Z, Chen X, Ali MF, Abdeltawab AA, Yakout SM, Yu G. Pretreatment of wheat straw using basic ethanolamine-based deep eutectic solvents for improving enzymatic hydrolysis. Bioresour Technol. 2018. https://doi.org/10.1016/j.biortech.2018.05.016.
Sharma V, Nargotra P, Sharma S, Bajaj BK. Efficacy and functional mechanisms of a novel combinatorial pretreatment approach based on deep eutectic solvent and ultrasonic waves for bioconversion of sugarcane bagasse. Renew Energy. 2021;163:1910–22. https://doi.org/10.1016/j.renene.2020.10.101.
Wu M, Gong L, Ma C, He YC. Enhanced enzymatic saccharification of sorghum straw by effective delignification via combined pretreatment with alkali extraction and deep eutectic solvent soaking. Bioresour Technol. 2021;340:125695. https://doi.org/10.1016/j.biortech.2021.125695.
Bjelic A, Hočevar B, Grilc M, Novak U, Likozar B. A review of sustainable lignocellulose biorefining applying (natural) deep eutectic solvents (DESs) for separations, catalysis and enzymatic biotransformation processes. Rev Chem Eng. 2022;38:243–72.
Ballesteros I, Negro MJ, Oliva JM, Cabañas A, Manzanares P, Ballesteros M. Ethanol production from steam-explosion pretreated wheat straw. Appl Biochem Biotechnol. 2006;129:496–508.
John I, Yaragarla P, Muthaiah P, Ponnusamy K, Appusamy A. Statistical optimization of acid catalyzed steam pretreatment of citrus peel waste for bioethanol production. Resour Technol. 2017;3:429–33.
Sharma S, Kumar R, Gaur R, Agrawal R, Gupta RP, Tuli DK, et al. Pilot scale study on steam explosion and mass balance for higher sugar recovery from rice straw. Bioresour Technol. 2015;175:350–7. https://doi.org/10.1016/j.biortech.2014.10.112.
Fan X, Cheng G, Zhang H, Li M, Wang S, Yuan Q. Effects of acid impregnated steam explosion process on xylose recovery and enzymatic conversion of cellulose in corncob. Carbohydr Polym. 2014;114:21–6.
Gao H, Wang Y, Yang Q, Peng H, Li Y, Zhan D, et al. Combined steam explosion and optimized green-liquor pretreatments are effective for complete saccharification to maximize bioethanol production by reducing lignocellulose recalcitrance in one-year-old bamboo. Renew Energy. 2021;175:1069–79. https://doi.org/10.1016/j.renene.2021.05.016.
Bon F, Cagno M, Rey F, Torres M, Böthig S, Menéndez P, et al. Pretreatment of switchgrass by steam explosion in a semi-continuous pre-pilot reactor. Biomass Bioenergy. 2019;121:41–7.
Chu Q, Song K, Bu Q, Hu J, Li F, Wang J, et al. Two-stage pretreatment with alkaline sulphonation and steam treatment of Eucalyptus woody biomass to enhance its enzymatic digestibility for bioethanol production. Energy Convers Manag. 2018;175:236–45. https://doi.org/10.1016/j.enconman.2018.08.100.
Banoth C, Sunkar B, Tondamanati PR, Bhukya B. Improved physicochemical pretreatment and enzymatic hydrolysis of rice straw for bioethanol production by yeast fermentation. 3 Biotech. 2017. https://doi.org/10.1007/s13205-017-0980-6.
Lu J, Song F, Liu H, Chang C, Cheng Y, Wang H. Production of high concentration bioethanol from reed by combined liquid hot water and sodium carbonate-oxygen pretreatment. Energy. 2020. https://doi.org/10.1016/j.energy.2020.119332.
Wang Z, Dien BS, Rausch KD, Tumbleson ME, Singh V. Fermentation of undetoxified sugarcane bagasse hydrolyzates using a two stage hydrothermal and mechanical refining pretreatment. Bioresour Technol. 2018;261:313–21. https://doi.org/10.1016/j.biortech.2018.04.018.
Tian D, Shen F, Yang G, Deng S, Long L, He J, et al. Liquid hot water extraction followed by mechanical extrusion as a chemical- free pretreatment approach for cellulosic ethanol production from rigid hardwood. Fuel. 2019;252:589–97. https://doi.org/10.1016/j.fuel.2019.04.155.
Lu J, Liu H, Song F, **a F, Huang X, Zhang Z, et al. Combining hydrothermal-alkaline/oxygen pretreatment of reed with PEG 6,000-assisted enzyme hydrolysis promote bioethanol fermentation and reduce enzyme loading. Ind Crops Prod. 2020;153:112615. https://doi.org/10.1016/j.indcrop.2020.112615.
Chen X, Li H, Sun S, Cao X, Sun R. Co-production of oligosaccharides and fermentable sugar from wheat straw by hydrothermal pretreatment combined with alkaline ethanol extraction. Ind Crop Prod. 2018;111:78–85. https://doi.org/10.1016/j.indcrop.2017.10.014.
Song K, Chu Q, Hu J, Bu Q, Li F, Chen X, et al. Two-stage alkali-oxygen pretreatment capable of improving biomass saccharification for bioethanol production and enabling lignin valorization via adsorbents for heavy metal ions under the biorefinery concept. Bioresour Technol. 2019;276:161–9. https://doi.org/10.1016/j.biortech.2018.12.107.
Li S, Li M, Bian J, Sun S, Peng F, Xue Z. Biphasic 2-methyltetrahydrofuran/oxalic acid/water pretreatment to enhance cellulose enzymatic hydrolysis and lignin valorization. Bioresour Technol. 2017. https://doi.org/10.1016/j.biortech.2017.07.075.
Imman S, Laosiripojana N, Champreda V. Effects of liquid hot water pretreatment on enzymatic hydrolysis and physicochemical changes of corncobs. Appl Biochem Biotechnol. 2017. https://doi.org/10.1007/s12010-017-2541-1.
Zhao C, Qiao X, Cao Y, Shao Q. Application of hydrogen peroxide presoaking prior to ammonia fiber expansion pretreatment of energy crops. Fuel. 2017;205:184–91. https://doi.org/10.1016/j.fuel.2017.05.073.
Zhao C, Shao Q, Chundawat SPS. Recent advances on ammonia-based pretreatments of lignocellulosic biomass. Bioresour Technol. 2020;298:122446. https://doi.org/10.1016/j.biortech.2019.122446.
Yuan H, Guan R, Wachemo AC, Zhang Y, Zuo X, Li X. Chinese Journal of Chemical Engineering Improving physicochemical characteristics and anaerobic digestion performance of rice straw via ammonia pretreatment at varying concentrations and moisture levels. Chin J Chem Eng. 2019. https://doi.org/10.1016/j.cjche.2019.07.009.
Cha YL, Yang J, Ahn JW, Moon YH, Yoon YM, Yu GD, et al. The optimized CO2-added ammonia explosion pretreatment for bioethanol production from rice straw. Bioprocess Biosyst Eng. 2014;37:1907–15.
Phitsuwan P, Permsriburasuk C, Waeonukul R, Pason P, Tachaapaikoon C, Ratanakhanokchai K. Evaluation of fuel ethanol production from aqueous ammonia-treated rice straw via simultaneous saccharification and fermentation. Biomass Bioenergy. 2016;93:150–7. https://doi.org/10.1016/j.biombioe.2016.07.012.
Ebrahimi M, Caparanga AR, Ordono EE, Villaflores OB, Pouriman M. Effect of ammonium carbonate pretreatment on the enzymatic digestibility, structural characteristics of rice husk and bioethanol production via simultaneous saccharification and fermentation process with Saccharomyces cerevisiae Hansen 2055. Ind Crops Prod. 2017;101:84–91. https://doi.org/10.1016/j.indcrop.2017.03.006.
Arumugham T, AlYammahi J, Rambabu K, Hassan SW, Banat F. Supercritical CO2 pretreatment of date fruit biomass for enhanced recovery of fruit sugars. Sustain Energy Technol Assess. 2022;52:102231.
Sun J, Ding R, Yin J. Pretreatment corn ingredient biomass with high pressure CO2 for conversion to fermentable sugars via enzymatic hydrolysis of cellulose. Ind Crops Prod. 2022;177:114518.
Ge S, Wu Y, Peng W, **a C, Mei C, Cai L, et al. High-pressure CO2 hydrothermal pretreatment of peanut shells for enzymatic hydrolysis conversion into glucose. Chem Eng J. 2020;385:123949. https://doi.org/10.1016/j.cej.2019.123949.
Arvaniti E, Bjerre AB, Schmidt JE. Wet oxidation pretreatment of rape straw for ethanol production. Biomass Bioenergy. 2012;39:94–105. https://doi.org/10.1016/j.biombioe.2011.12.040.
Schultz-Jensen N, Thygesen A, Leipold F, Thomsen ST, Roslander C, Lilholt H, et al. Pretreatment of the macroalgae Chaetomorpha linum for the production of bioethanol—comparison of five pretreatment technologies. Bioresour Technol. 2013;140:36–42. https://doi.org/10.1016/j.biortech.2013.04.060.
Banerjee S, Sen R, Pandey RA, Chakrabarti T, Satpute D, Giri BS, et al. Evaluation of wet air oxidation as a pretreatment strategy for bioethanol production from rice husk and process optimization. Biomass Bioenergy. 2009;33:1680–6. https://doi.org/10.1016/j.biombioe.2009.09.001.
Banerjee S, Sen R, Morone A, Chakrabarti T, Pandey R, Mudliar S. Improved wet air oxidation pretreatment for enhanced enzymatic hydrolysis of rice husk for bioethanol production. 2012;1–3. http://www.globalsciencebooks.info/Online/GSBOnline/images/2012/DBPBMB_6(SI2)/DBPBMB_6(SI2)43-45o.pdf
Martín C, Thomsen MH, Hauggaard-Nielsen H, BelindaThomsen A. Wet oxidation pretreatment, enzymatic hydrolysis and simultaneous saccharification and fermentation of clover-ryegrass mixtures. Bioresour Technol. 2008;99:8777–82.
Kumar AK, Sharma S. Recent updates on different methods of pretreatment of lignocellulosic feedstocks: a review. Bioresour Bioprocess. 2017;4:1–19.
Naresh Kumar M, Ravikumar R, Thenmozhi S, Kirupa Sankar M. Development of natural cellulase inhibitor mediated intensified biological pretreatment technology using Pleurotus florida for maximum recovery of cellulose from paddy straw under solid state condition. Bioresour Technol. 2017;244:353–61. https://doi.org/10.1016/j.biortech.2017.07.105.
Sharma HK, Xu C, Qin W. Biological pretreatment of lignocellulosic biomass for biofuels and bioproducts: an overview. Waste Biomass Valoriz. 2019;10:235–51.
**e C, Gong W, Yang Q, Zhu Z, Yan L, Hu Z, et al. White-rot fungi pretreatment combined with alkaline/oxidative pretreatment to improve enzymatic saccharification of industrial hemp. Bioresour Technol. 2017;243:188–95. https://doi.org/10.1016/j.biortech.2017.06.077.
Arora A, Priya S, Sharma P, Sharma S, Nain L. Evaluating biological pretreatment as a feasible methodology for ethanol production from paddy straw. Biocatal Agric Biotechnol. 2016;8:66–72. https://doi.org/10.1016/j.bcab.2016.08.006.
Shen F, Zhong B, Wang Y, **a X, Zhai Z, Zhang Q. Cellulolytic microflora pretreatment increases the efficiency of anaerobic co-digestion of rice straw and pig manure. Bioenergy Res BioEnergy Research. 2019;12:703–13.
Kumar P, Kumar V, Kumar S, Singh J, Kumar P. Bioethanol production from sesame (Sesamum indicum L.) plant residue by combined physical, microbial and chemical pretreatments. Bioresour Technol. 2020. https://doi.org/10.1016/j.biortech.2019.122484.
Ummalyma SB, Supriya RD, Sindhu R, Binod P, Nair RB, Pandey A, et al. Biological pretreatment of lignocellulosic biomass-current trends and future perspectives. Second Third Gener Feed Evol Biofuels. 2019. https://doi.org/10.1016/B978-0-12-815162-4.00007-0.
Zhang Q, He J, Tian M, Mao Z, Tang L, Zhang J, et al. Enhancement of methane production from cassava residues by biological pretreatment using a constructed microbial consortium. Bioresour Technol. 2011;102:8899–906. https://doi.org/10.1016/j.biortech.2011.06.061.
Mohanram S, Rajan K, Julie D, Nain L, Arora A. Insights into biological delignification of rice straw by Trametes hirsuta and Myrothecium roridum and comparison of saccharification yields with dilute acid pretreatment. Biomass Bioenergy. 2015;76:54–60. https://doi.org/10.1016/j.biombioe.2015.02.031.
Mustafa AM, Poulsen TG, Sheng K. Fungal pretreatment of rice straw with Pleurotus ostreatus and Trichoderma reesei to enhance methane production under solid-state anaerobic digestion. Appl Energy. 2016;180:661–71. https://doi.org/10.1016/j.apenergy.2016.07.135.
García-Torreiro M, López-Abelairas M, Lu-Chau TA, Lema JM. Fungal pretreatment of agricultural residues for bioethanol production. Ind Crops Prod. 2016;89:486–92. https://doi.org/10.1016/j.indcrop.2016.05.036.
Mushlihah S, Husain DR, Langford A, Tassakka ACMAR. Fungal pretreatment as a sustainable and low cost option for bioethanol production from marine algae. J Clean Prod. 2020;265:121763. https://doi.org/10.1016/j.jclepro.2020.121763.
Zhang Q, Wei Y, Han H, Weng C. Enhancing bioethanol production from water hyacinth by new combined pretreatment methods. Bioresour Technol. 2018;251:358–63. https://doi.org/10.1016/j.biortech.2017.12.085.
Gupta K, Chundawat TS. Zinc oxide nanoparticles synthesized using Fusarium oxysporum to enhance bioethanol production from rice-straw. Biomass Bioenergy. 2020;143:105840. https://doi.org/10.1016/j.biombioe.2020.105840.
Zhang K, Xu R, Abomohra AE, **e S, Yu Z, Guo Q. A sustainable approach for efficient conversion of lignin into biodiesel accompanied by biological pretreatment of corn straw. Energy Convers Manag. 2019;199:111928. https://doi.org/10.1016/j.enconman.2019.111928.
Barati B, Zafar FF, Rupani PF, Wang S. Bacterial pretreatment of microalgae and the potential of novel nature hydrolytic sources. Environ Technol Innov. 2021;21:101362. https://doi.org/10.1016/j.eti.2021.101362.
Baramee S, Siriatcharanon AK, Ketbot P, Teeravivattanakit T, Waeonukul R, Pason P, et al. Biological pretreatment of rice straw with cellulase-free xylanolytic enzyme-producing Bacillus firmus K-1: Structural modification and biomass digestibility. Renew Energy. 2020;160:555–63. https://doi.org/10.1016/j.renene.2020.06.061.
Dai Y, Zhang HS, Huan B, He Y. Enhancing the enzymatic saccharification of bamboo shoot shell by sequential biological pretreatment with Galactomyces sp. CCZU11–1 and deep eutectic solvent extraction. Bioprocess Biosyst Eng. 2017;40:1427–36.
Zhuo S, Yan X, Liu D, Si M, Zhang K, Liu M, et al. Use of bacteria for improving the lignocellulose biorefinery process: importance of pre-erosion. Biotechnol Biofuels. 2018;11:1–13. https://doi.org/10.1186/s13068-018-1146-4.
Zhang D, Luo Y, Chu S, Zhi Y, Wang B, Zhou P. Biological pretreatment of rice straw with streptomyces griseorubens JSD-1 and its optimized production of cellulase and xylanase for improved enzymatic saccharification efficiency. Prep Biochem Biotechnol. 2015;46:575–85.
Tsegaye B, Balomajumder C, Roy P. Biodegradation of wheat straw by Ochrobactrum oryzae BMP03 and Bacillus sp. BMP01 bacteria to enhance biofuel production by increasing total reducing sugars yield. Environ Sci Pollut Res. 2018;25:30585–96.
Tian J, Pourcher A, Bize A, Wazeri A, Peu P. Impact of wet aerobic pretreatments on cellulose accessibility and bacterial communities in rape straw. Bioresour Technol. 2017. https://doi.org/10.1016/j.biortech.2017.03.142.
Meenakshisundaram S, Fayeulle A, Leonard E, Ceballos C, Pauss A. Fiber degradation and carbohydrate production by combined biological and chemical/physicochemical pretreatment methods of lignocellulosic biomass—a review. Bioresour Technol. 2021;331:125053.
Nasir A, Chen HZ, Wang L. Novel single-step pretreatment of steam explosion and choline chloride to de-lignify corn stover for enhancing enzymatic edibility. Process Biochem. 2020;94:273–81. https://doi.org/10.1016/j.procbio.2020.04.036.
Shukla A, Kumar D, Girdhar M, Sharma A, Mohan A. Steam explosion pretreatment with different concentrations of hydrogen peroxide along with citric acid : a former step towards bioethanol production. Int J Energy Res 2023;2023. https://www.hindawi.com/journals/ijer/2023/2492528/
Mosier N, Wyman C, Dale B, Elander R, Lee YY, Holtzapple M, et al. Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour Technol. 2005;96:673–86.
Rajesh Banu J, Poornima Devi T, Yukesh Kannah R, Kavitha S, Kim SH, Muñoz R, et al. A review on energy and cost effective phase separated pretreatment of biosolids. Water Res. 2021;198:117169. https://doi.org/10.1016/j.watres.2021.117169.
Kavitha S, Gondi R, Kannah RY, Kumar G, Rajesh BJ. A review on current advances in the energy and cost effective pretreatments of algal biomass: enhancement in liquefaction and biofuel recovery. Bioresour Technol. 2023;369:128383. https://doi.org/10.1016/j.biortech.2022.128383.
Gandam PK, Chinta ML, Pabbathi NPP, Baadhe RR, Sharma M, Thakur VK, et al. Second-generation bioethanol production from corncob—a comprehensive review on pretreatment and bioconversion strategies, including techno-economic and lifecycle perspective. Ind Crops Prod. 2022;186:115245. https://doi.org/10.1016/j.indcrop.2022.115245.
Malik K, Sharma P, Yang Y, Zhang P, Zhang L, **ng X, et al. Lignocellulosic biomass for bioethanol: Insight into the advanced pretreatment and fermentation approaches. Ind Crops Prod. 2022;188:115569. https://doi.org/10.1016/j.indcrop.2022.115569.
Liu LY, Chandra RP, Tang Y, Huang XY, Bai FW, Liu CG. Instant catapult steam explosion: an efficient preprocessing step for the robust and cost-effective chemical pretreatment of lignocellulosic biomass. Ind Crops Prod. 2022;188:115664. https://doi.org/10.1016/j.indcrop.2022.115664.
Basak B, Kumar R, Bharadwaj AVSLS, Kim TH, Kim JR, Jang M, et al. Advances in physicochemical pretreatment strategies for lignocellulose biomass and their effectiveness in bioconversion for biofuel production. Bioresour Technol. 2023;369:128413.
Srivastava N, Singh R, Srivastava M, Mohammad A, Harakeh S, Pratap Singh R, et al. Impact of nanomaterials on sustainable pretreatment of lignocellulosic biomass for biofuels production: an advanced approach. Bioresour Technol. 2023;369:128471.
Mumtaz M, Baqar Z, Hussain N, Bilal M, Azam HMH, et al. Application of nanomaterials for enhanced production of biodiesel, biooil, biogas, bioethanol, and biohydrogen via lignocellulosic biomass transformation. Fuel. 2022;315:122840.
Acknowledgements
All authors are thankful to Lovely Professional University for providing labs to perform work.
Funding
No source of funding is applicable.
Author information
Authors and Affiliations
Contributions
AS has contributed substantially to writing the draft and preparing the manuscript; AM and MG have contributed to the conceptualization, manuscript editing, and drawing of the hypothesis for the study; AG has contributed to the making of tables and figures for the manuscript; AK has contributed critically revising the work; DK and TM has contributed to study design and editing. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1.
Illustrate mechanism of pretreatment techniques towards bioethanol production and its effect on feedstock with main emphasis on the size reduction, cellulose disruption along with hemicellulose and lignin depolarization [14, 18, 22]. Figure S2. Depicts diagrammatic representation showing effect of acid pretreatment on feedstock [55, 56]. Figure S3. Depicts the diagrammatical representation of combined pretreatment using chemical along with steam explosion process [32, 34].
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Shukla, A., Kumar, D., Girdhar, M. et al. Strategies of pretreatment of feedstocks for optimized bioethanol production: distinct and integrated approaches. Biotechnol Biofuels 16, 44 (2023). https://doi.org/10.1186/s13068-023-02295-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13068-023-02295-2