Abstract
Background
Immunotherapy is currently being examined as a treatment modality for glioblastoma. Maintaining an optimal total lymphocyte count (TLC) after radiotherapy (RT) and using temozolomide may be beneficial in optimizing immunotherapy. However, conventional temozolomide-based chemoradiation is known to induce immunosuppressive effects, including lymphopenia. Therefore, this study aimed to identify potential clinical predictors of acute severe lymphopenia (ASL) in patients receiving chemoradiation for glioblastoma.
Methods
We identified patients with glioblastoma treated with RT plus temozolomide from 2006 to 2017. ASL was defined as a TLC of < 500/μL within 3 months after initiating RT. Independent predictors of ASL were determined using logistic regression.
Results
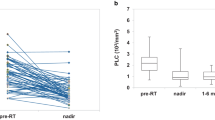
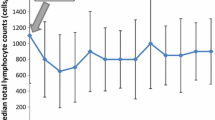
A total of 336 patients were evaluated. Three-dimensional conformal RT (3D-CRT) and intensity-modulated RT (IMRT) were used in 186 (55.4%) and 150 patients (44.6%), respectively. TLC decreased during RT and remained persistently low during the 1-year follow-up, whereas the levels of other blood cell types recovered. In total, 118 patients (35.1%) developed ASL. During a median follow-up of 19.3 months, patients with ASL showed significantly worse overall survival than did those without ASL (median, 18.2 vs. 22.0 months; P = .028). Multivariable analysis revealed that increased planning target volume (PTV) was independently associated with increased ASL incidence (hazard ratio [HR], 1.02; 95% confidence interval [CI], 1.00–1.03; P = .042), while IMRT was independently associated with decreased ASL incidence (HR, 0.48; 95% CI, 0.27–0.87; P = .015). A propensity-matched comparison showed that the incidence of ASL was lower with IMRT than with 3D-CRT (20% vs. 37%; P = .005).
Conclusions
IMRT and low PTV were significantly associated with decreased ASL incidence after RT plus temozolomide for glioblastoma. An IMRT-based strategy is necessary to enhance treatment outcomes in the immune-oncology era.
Similar content being viewed by others
Background
Radiotherapy (RT)-induced lymphopenia (i.e., a reduction in the total lymphocyte count [TLC]) has been reported in various types of tumors, such as glioblastomas, pancreatic cancer, and lung cancer [1,2,3,4,5,6,7]. Although radiation has local effects, RT to peripheral organs can result in irradiation of a substantial proportion of circulating lymphocytes during multifraction treatments [8]. Recently, several studies have demonstrated that partial brain RT can contribute to systemic lymphopenia [5,6,7,8]. RT-induced lymphopenia is associated with poor survival in patients with high-grade gliomas who underwent standard therapy with RT and temozolomide [5, 6].
Despite multimodal treatment involving surgery, RT, and temozolomide, glioblastoma has a poor prognosis and almost all patients with glioblastoma eventually experience disease relapse [9]. Although repeat surgery, re-irradiation, and pharmacological treatment have been performed in the recurrent setting, evidence that any therapeutic intervention has a major effect on survival is lacking [10, 11]. Accordingly, different immunotherapy modalities for glioblastoma are being actively investigated, spurred on by advances in immuno-oncology for other tumor types [ This study revealed that although a large PTV can increase the risk of ASL, IMRT can effectively lower the risk of ASL after the initiation of RT plus temozolomide for treating glioblastoma. Our findings add to the growing evidence on the association between RT and treatment-induced lymphopenia in patients with glioblastoma. Particularly, in cases with a large tumor size or surgical cavity, IMRT-based therapeutic strategies should be actively considered to preserve the TLC. Such strategies could potentially improve treatment outcomes in the immuno-oncology era, and would thus need further study.Conclusions
Abbreviations
- 3D-CRT:
-
Three-dimensional conformal radiotherapy
- ASL:
-
Acute severe lymphopenia
- CI:
-
Confidence interval
- CTV:
-
Clinical target volume
- GTV:
-
Gross tumor volume
- HR:
-
Hazard ratio
- IMRT:
-
Intensity-modulated radiotherapy
- MRI:
-
Magnetic resonance imaging
- OS:
-
Overall survival
- PTV:
-
Planning target volume
- RT:
-
Radiotherapy
- TLC:
-
Total lymphocyte count
References
Grossman SA, Ellsworth S, Campian J, et al. Survival in patients with severe lymphopenia following treatment with radiation and chemotherapy for newly diagnosed solid tumors. J Natl Compr Cancer Netw. 2015;13(10):1225–31.
Petrini B, Wasserman J, Blomgren H, et al. Blood lymphocyte subpopulations in breast cancer patients following post-operative adjuvant chemotherapy or radiotherapy. Clin Exp Immunol. 1979;38(2):361–5.
Wild AT, Ye X, Ellsworth SG, et al. The association between Chemoradiation-related lymphopenia and clinical outcomes in patients with locally advanced pancreatic adenocarcinoma. Am J Clin Oncol. 2015;38(3):259–65.
Wild AT, Herman JM, Dholakia AS, et al. Lymphocyte-sparing effect of stereotactic body radiation therapy in patients with Unresectable pancreatic Cancer. Int J Radiat Oncol Biol Phys. 2016;94(3):571–9.
Grossman SA, Ye X, Lesser G, et al. Immunosuppression in patients with high-grade gliomas treated with radiation and temozolomide. Clin Cancer Res. 2011;17(16):5473–80.
Rudra S, Hui C, Rao YJ, et al. Effect of radiation treatment volume reduction on lymphopenia in patients receiving Chemoradiotherapy for glioblastoma. Int J Radiat Oncol Biol Phys. 2018;101(1):217–25.
Huang J, DeWees TA, Badiyan SN, et al. Clinical and Dosimetric predictors of acute severe lymphopenia during radiation therapy and concurrent Temozolomide for high-grade glioma. Int J Radiat Oncol Biol Phys. 2015;92(5):1000–7.
Yovino S, Kleinberg L, Grossman SA, et al. The etiology of treatment-related lymphopenia in patients with malignant gliomas: modeling radiation dose to circulating lymphocytes explains clinical observations and suggests methods of modifying the impact of radiation on immune cells. Cancer Investig. 2013;31(2):140–4.
Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–96.
Weller M, van den Bent M, Tonn JC, et al. European Association for Neuro-Oncology (EANO) guideline on the diagnosis and treatment of adult astrocytic and oligodendroglial gliomas. Lancet Oncol. 2017;18(6):e315–e29.
Lee J, Ahn SS, Chang JH, et al. Hypofractionated re-irradiation after maximal surgical resection for recurrent glioblastoma: therapeutic adequacy and its prognosticators of survival. Yonsei Med J. 2018;59(2):194–201.
Lim M, **a Y, Bettegowda C, et al. Current state of immunotherapy for glioblastoma. Nat Rev Clin Oncol. 2018;15(7):422–42.
Gupta T, Mohanty S, Moiyadi A, et al. Factors predicting temozolomide induced clinically significant acute hematologic toxicity in patients with high-grade gliomas: a clinical audit. Clin Neurol Neurosurg. 2013;115(9):1814–9.
Choi SH, Kim JW, Chang JS, et al. Impact of including Peritumoral edema in radiotherapy target volume on patterns of failure in glioblastoma following Temozolomide-based Chemoradiotherapy. Sci Rep. 2017;7:42148.
Lim DA, Cha S, Mayo MC, et al. Relationship of glioblastoma multiforme to neural stem cell regions predicts invasive and multifocal tumor phenotype. Neuro-Oncology. 2007;9(4):424–9.
Carr D, ported by Nicholas Lewin-Koh, Martin Maechler and contains copies of latticefunction written by Deepayan Sarkar: Hexbin: hexagonal binning routines. Package version 1.27.0. http://CRAN.R-project.org/package=hexbin. (2014).
Ho KI D, King G, Stuart EA. MatchIt: nonparametric preprocessing for parametric causal inference. J Stat Softw. 2011;42(8):1–28.
Gramatzki D, Dehler S, Rushing EJ, et al. Glioblastoma in the Canton of Zurich, Switzerland revisited: 2005 to 2009. Cancer. 2016;122(14):2206–15.
Weller M, Cloughesy T, Perry JR, et al. Standards of care for treatment of recurrent glioblastoma--are we there yet? Neuro-Oncology. 2013;15(1):4–27.
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420(6917):860–7.
Tran C, Knowles SR, Liu BA, et al. Gender differences in adverse drug reactions. J Clin Pharmacol. 1998;38(11):1003–9.
Nakamura N, Kusunoki Y, Akiyama M. Radiosensitivity of CD4 or CD8 positive human T-lymphocytes by an in vitro colony formation assay. Radiat Res. 1990;123(2):224–7.
Narayana A, Yamada J, Berry S, et al. Intensity-modulated radiotherapy in high-grade gliomas: clinical and dosimetric results. Int J Radiat Oncol Biol Phys. 2006;64(3):892–7.
Chen AB. Comparative effectiveness research in radiation oncology: assessing technology. Semin Radiat Oncol. 2014;24(1):25–34.
Nutting CM, Morden JP, Harrington KJ, et al. Parotid-sparing intensity modulated versus conventional radiotherapy in head and neck cancer (PARSPORT): a phase 3 multicentre randomised controlled trial. Lancet Oncol. 2011;12(2):127–36.
Liu LT, Chen QY, Tang LQ, et al. The prognostic value of treatment-related lymphopenia in nasopharyngeal carcinoma patients. Cancer Res Treat. 2018;50(1):19–29.
Lohr J, Ratliff T, Huppertz A, et al. Effector T-cell infiltration positively impacts survival of glioblastoma patients and is impaired by tumor-derived TGF-beta. Clin Cancer Res. 2011;17(13):4296–308.
Gooden MJ, de Bock GH, Leffers N, et al. The prognostic influence of tumour-infiltrating lymphocytes in cancer: a systematic review with meta-analysis. Br J Cancer. 2011;105(1):93–103.
Campian JL, Sarai G, Ye X, et al. Association between severe treatment-related lymphopenia and progression-free survival in patients with newly diagnosed squamous cell head and neck cancer. Head Neck. 2014;36(12):1747–53.
Venkatesulu BP, Mallick S, Lin SH, et al. A systematic review of the influence of radiation-induced lymphopenia on survival outcomes in solid tumors. Crit Rev Oncol Hematol. 2018;123:42–51.
Jafri NF, Clarke JL, Weinberg V, et al. Relationship of glioblastoma multiforme to the subventricular zone is associated with survival. Neuro-Oncology. 2013;15(1):91–6.
Yan H, Parsons DW, ** G, et al. IDH1 and IDH2 mutations in gliomas. N Engl J Med. 2009;360(8):765–73.
Hegi ME, Diserens AC, Gorlia T, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005;352(10):997–1003.
Acknowledgements
None.
Funding
This work was supported by the Ministry of Science, Korea, through the research and development program of the National Research Foundation of Korea (NRF-2017R1C1B2010379).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Contributions
HKB and NK collected the clinical data. HKB and HIY were involved in the study design, performed the statistical analysis, interpreted the results, and drafted the manuscript. HIY, SGK, SHK, JC, JHC, and COS cared for the patients. JGB performed the treatment planning. All of the authors have read and approved the final draft.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional file
Additional file 1:
Table S1. Univariate and multivariate Cox regression analyses for overall survival in the 3D-CRT group. Table S2. Univariate and multivariate Cox regression analyses for overall survival in the IMRT group. Table S3. Univariate and multivariate Cox regression analyses for overall survival in all patients. Table S4. Covariates included in the propensity score matching. Table S5. The dosimetric parameters for brain. (DOCX 47 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Byun, H.K., Kim, N., Yoon, H.I. et al. Clinical predictors of radiation-induced lymphopenia in patients receiving chemoradiation for glioblastoma: clinical usefulness of intensity-modulated radiotherapy in the immuno-oncology era. Radiat Oncol 14, 51 (2019). https://doi.org/10.1186/s13014-019-1256-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13014-019-1256-6