Abstract
Purpose
Lymphopenia in patients with glioblastoma (GBM) is related to treatment as well as disease progression. This retrospective study investigated the prevalence, influencing factors, recoverability, and clinical significance of lymphopenia in GBM patients treated with concomitant chemoradiotherapy (CCRT).
Patients and methods
A total of 219 patients with newly diagnosed GBM who had received at least 3 cycles of adjuvant temozolomide (TMZ) followed by CCRT with TMZ were enrolled. Serial data on complete blood cell counts, including differential cell counts, were collected just before a new phase and before every treatment cycle of the regimen. Relationships between white blood cell (WBC) variable changes and treatment modalities as well as survival were analyzed. Lymphopenia was classified using the definition of the Common Terminology Criteria for Adverse Events version 5.0.
Results
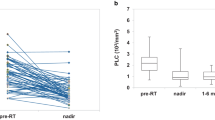
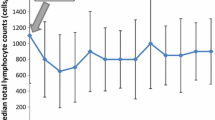
A total of 92 patients (42.0%) showed decreased levels of lymphocytes (< 1500/µL) at baseline. The WBC count, absolute neutrophil count, lymphocyte count, and neutrophil-to-lymphocyte ratio were all significantly decreased after RT/TMZ treatment and did not recover during the adjuvant TMZ period. However, these metrics all began to recover 3 months after the last TMZ cycle, except for the lymphocyte count. The proportion of lymphopenia patients (< 1500 lymphocytes/µL) increased to 74.8% after RT/TMZ and remained steady at approximately 71.5% (range 63.7–75.3%) throughout the management period. Moreover, the number of patients with grade 3 lymphopenia (< 500 lymphocytes/µL) also increased significantly after treatment to reach 2.9% (from 0.9% at baseline). Statistically, 75.7% of lymphopenia patients were predicted to recover in a median time of 240.3 days (95% confidence interval ± 104.7 days) after TMZ withdrawal. There were no dose-dependent relationships between RT or TMZ and lymphopenia. Grade 3 (< 500 lymphocytes/µL) lymphopenia measured at 1 month after RT/TMZ predicted significantly reduced survival (13.0 months vs. 19.5 months, p = 0.011).
Conclusion
Lymphopenia is a frequent event during GBM disease progression and treatment. Treatment-related lymphopenia is profound and prolonged and can be used as a prognostic factor for GBM patients.




Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Mendez JS, Govindan A, Leong J et al (2016) Association between treatment-related lymphopenia and overall survival in elderly patients with newly diagnosed glioblastoma. J Neuro-Oncol 127:329–335
Bae SH, Park MJ, Lee MM et al (2014) Toxicity profile of temozolomide in the treatment of 300 malignant glioma patients in Korea. J Korean Med Sci 29:980–984
Lim M, **a Y, Bettegowda C et al (2018) Current state of immunotherapy for glioblastoma. Nat Rev Clin Oncol 15:422–442
Campian JL, Piotrowski AF, Ye X et al (2017) Serial changes in lymphocyte subsets in patients with newly diagnosed high grade astrocytomas treated with standard radiation and temozolomide. J Neuro-Oncol 135:343–351
Ellsworth S, Balmanoukian A, Kos F et al (2014) Sustained CD4(+) T cell-driven lymphopenia without a compensatory IL-7/IL-15 response among high-grade glioma patients treated with radiation and temozolomide. Oncoimmunology 3:e27357
Yovino S, Kleinberg L, Grossman SA et al (2013) The etiology of treatment-related lymphopenia in patients with malignant gliomas: modeling radiation dose to circulating lymphocytes explains clinical observations and suggests methods of modifying the impact of radiation on immune cells. Cancer Investig 31:140–144
Sampson JH, Aldape KD, Archer GE et al (2011) Greater chemotherapy-induced lymphopenia enhances tumor-specific immune responses that eliminate EGFRvIII-expressing tumor cells in patients with glioblastoma. Neuro-oncology 13:324–333
Grossman SA, Ye X, Lesser G et al (2011) Immunosuppression in patients with high-grade gliomas treated with radiation and temozolomide. Clin Cancer Res 17:5473–5480
Lee JS, Kim NY, Na SH et al (2018) Reference values of neutrophil-lymphocyte ratio, lymphocyte-monocyte ratio, platelet-lymphocyte ratio, and mean platelet volume in healthy adults in South Korea. Medicine 97:e11138
Mahaley MS Jr, Brooks WH, Roszman TL et al (1977) Immunobiology of primary intracranial tumors. Part 1: studies of the cellular and humoral general immune competence of brain-tumor patients. J Neurosurg 46:467–476
Dix AR, Brooks WH, Roszman TL et al (1999) Immune defects observed in patients with primary malignant brain tumors. J Neuroimmunol 100:216–232
Dunn GP, Fecci PE, Curry WT (2012) Cancer immunoediting in malignant glioma. Neurosurgery 71:201–222 (discussion 222–203)
Fecci PE, Heimberger AB, Sampson JH (2014) Immunotherapy for primary brain tumors: no longer a matter of privilege. Clin Cancer Res 20:5620–5629
Woroniecka KI, Rhodin KE, Chongsathidkiet P et al (2018) T-cell dysfunction in Glioblastoma: applying a new framework. Clin Cancer Res 24:3792–3802
Chongsathidkiet P, Jackson C, Koyama S et al (2018) Sequestration of T cells in bone marrow in the setting of glioblastoma and other intracranial tumors. Nat Med 24:1459–1468
Yovino S, Grossman SA (2012) Severity, etiology and possible consequences of treatment-related lymphopenia in patients with newly diagnosed high-grade gliomas. CNS Oncol 1:149–154
Meyer KK (1970) Radiation-induced lymphocyte-immune deficiency. A factor in the increased visceral metastases and decreased hormonal responsiveness of breast cancer. Arch Surg 101:114–121
Yuan C, Wang Q (2018) Comparative analysis of the effect of different radiotherapy regimes on lymphocyte and its subpopulations in breast cancer patients. Clin Transl Oncol 20:1219–1225
Eric A, Juranic Z, Tisma N et al (2009) Radiotherapy-induced changes of peripheral blood lymphocyte subpopulations in cervical cancer patients: relationship to clinical response. J BUON 14:79–83
Stein R, Sharkey RM, Goldenberg DM (1992) Haematological effects of radioimmunotherapy in cancer patients. Br J Haematol 80:69–76
Raben M, Walach N, Galili U et al (1976) The effect of radiation therapy on lymphocyte subpopulations in cancer patients. Cancer 37:1417–1421
Heier HE, Christensen I, Froland SS et al (1975) Early and late effects of irradiation for seminoma testis on the number of blood lymphocytes and their B and T subpopulations. Lymphology 8:69–74
MacLennan IC, Kay HE (1978) Analysis of treatment in childhood leukemia. IV. The critical association between dose fractionation and immunosuppression induced by cranial irradiation. Cancer 41:108–111
Su YB, Sohn S, Krown SE et al (2004) Selective CD4 + lymphopenia in melanoma patients treated with temozolomide: a toxicity with therapeutic implications. J Clin Oncol 22:610–616
Dolan RD, Laird BJA, Horgan PG et al (2018) The prognostic value of the systemic inflammatory response in randomised clinical trials in cancer: a systematic review. Crit Rev Oncol/Hematol 132:130–137
Bambury RM, Teo MY, Power DG et al (2013) The association of pre-treatment neutrophil to lymphocyte ratio with overall survival in patients with glioblastoma multiforme. J Neuro-Oncol 114:149–154
Lopes M, Carvalho B, Vaz R et al (2018) Influence of neutrophil-lymphocyte ratio in prognosis of glioblastoma multiforme. J Neuro-Oncol 136:173–180
Schernberg A, Nivet A, Dhermain F et al (2018) Neutrophilia as a biomarker for overall survival in newly diagnosed high-grade glioma patients undergoing chemoradiation. Clin Transl Radiat Oncol 10:47–52
Kanemura Y, Sumida M, Okita Y et al (2017) Systemic intravenous adoptive transfer of autologous lymphokine-activated alphabeta T-cells improves temozolomide-induced lymphopenia in patients with glioma. Anticancer Res 37:3921–3932
Suryadevara CM, Desai R, Abel ML et al (2018) Temozolomide lymphodepletion enhances CAR abundance and correlates with antitumor efficacy against established glioblastoma. Oncoimmunology 7:e1434464
Acknowledgements
This research was supported by the Bio & Medical Technology Development Program of the NRF funded by the Korean government, MSIP (NRF-2015M3A9A7029740), and by a Grant from the Seoul National University Hospital (330-2018-0010).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None of the authors have any conflicts of interest to disclose.
Ethical approval
Ethical approval was provided by the Institutional Review Board of Seoul National University Hospital (H-1712-100-908).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, W.J., Dho, YS., Ock, CY. et al. Clinical observation of lymphopenia in patients with newly diagnosed glioblastoma. J Neurooncol 143, 321–328 (2019). https://doi.org/10.1007/s11060-019-03167-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-019-03167-2