Abstract
Background
To investigate the association between the Homeostasis Model Assessment of Insulin Resistance (HOMA-IR) and Homeostasis Model Assessment of Beta-cell function (HOMA-B) with the incidence of diabetes and pre-diabetes subtypes.
Methods
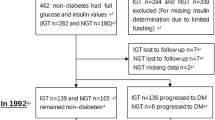
A total of 3101 normoglycemic people aged 20–70 years were included in the 6-year follow-up study. Multinomial logistic regression was used to calculate the incidence possibility of isolated Impaired Fasting Glucose (iIFG), isolated Impaired Glucose Tolerance (iIGT), Combined impaired fasting glucose & impaired glucose tolerance (CGI), and Diabetes Mellitus (DM) per standard deviation (SD) increment in HOMA-IR and HOMA-B in the crude and multivariable model.
Results
In the multivariate model, an increase in one SD change in HOMA-IR was associated with a 43, 42, 75, and 92% increased risk of iIFG, iIGT, CGI, and DM, respectively. There was a positive correlation between the increase in HOMA-B and the incidence of iIGT; however, after adjusting the results for metabolic syndrome components, it was inversely correlated with the incidence of iIFG [Odds Ratio = 0.86(0.75–0.99)].
Conclusions
HOMA-IR is positively correlated with diabetes and pre-diabetes subtypes’ incidence, and HOMA-B is inversely correlated with the incidence of iIFG but positively correlated with iIGT incidence. However, none of these alone is a good criterion for predicting diabetes and pre-diabetes.
Similar content being viewed by others
Introduction
Type 2 diabetes is a growing disease worldwide, imposing a severe burden on society along with its complications [1–4]. In Iran, the incidence rate of type 2 diabetes in the urban population over 20 years is more than 1 % annually [5]. The incidence rate of pre-diabetes is significantly higher and estimated to be more than 4 % each year, indicating a very high prevalence of diabetes soon [6].
Pre-diabetes is associated with a high risk of micro and macrovascular complications [7–10]. As a result, early detection of people susceptible to pre-diabetes, lifestyle modifications, and effective medications is necessary to prevent develo** pre-diabetes and its complications in these people [4, 11]. 30-year results of the Da Qing Diabetes Prevention Outcome Study showed that lifestyle intervention in people with pre-diabetes could reduce 40% development of diabetes and delay its onset by 3·96 years [12]. Insulin resistance and dysfunction of pancreatic beta cells are important factors in the pathophysiology of diabetes and pre-diabetes, contributing to different degrees among pre-diabetic subgroups according to different races and ethnicities [13–27]; thus, the effects of HOMA-B changes on the incidence of pre-diabetes can be affected by the time interval until the desired outcome in the future.
Also, in a study by Derakhshan et al., beta-cell dysfunction or low HOMA-B (HOMA-B < 25 percentile) was associated with increased risk of iIFG in a multivariate model [H.R. = 1.37(1.03–1.81) in men, and HR = 1.36(1.02–1.80) in women] which is in line with our findings. This study further showed that increased HOMA-IR was associated with increased HR in all four groups, which is in line with the results of our study, too [28]. A 2009 prospective study in Denmark to determine the association between insulin sensitivity and insulin secretion with the development of normoglycemic individuals to IFG or IGT reported that liver insulin resistance and subsequent dysfunction of beta cells in the secretion of insulin are the main factors in the progression of normal blood glucose to iIFG during 5 years, while the reduction of whole-body sensitivity and consequently insulin secretion dysfunction was reported as the major factors leading to iIGT [21]. It should be noted that, in this study, OGTT was performed, HOMA-S and ISI (Insulin Sensitivity Index) were used to assess insulin sensitivity, and EPIR (Early Phase Insulin Release) and DI (Disposition Index) were used to measure insulin secretion [29].
Too many studies investigated the association of HOMA-IR and HOMA-B with IFG and IGT. However, most of these were cross-sectional studies and evaluated this association concurrently with the occurrence of phenotypes, making it impossible to compare their results with the findings of this study. In this study, increasing HOMA-B per SD resulted in a reduced incidence of iIFG and an increased incidence of iIGT, CGI, and DM; Thus, AUC was not an appropriate criterion for predicting the development of pre-diabetes. On the other hand, HOMA-IR does not produce an acceptable AUC and cut-off point, so HOMA-IR is not a good index for predicting pre-diabetes. None of the ROC diagrams of HOMA-IR and HOMA-B, which were prepared separately for iIFG and iIGT, resulted in appropriate AUCs. In a study by Dr. Ghassemi et al., the HOMA-IR cut-off point for diagnosis of diabetes was 2.17 (50% sensitivity and 76.7% specificity) in women and 1.85% (75.9% sensitivity and 58.3% specificity) in men [19]. Compared with the results of Dr. Ghassemi’s study for diabetes, the cut-off point of our study seems to be inappropriate for predicting the incidence of pre-diabetes because the HOMA-IR cut-off point for pre-diabetes is expected to be lower than that for diabetes. Also, in a cross-sectional study by Baek et al., the reported cut-off point of HOMA-IR for detecting dysglycemia based on FBS and HbA1C (equal to 1.6) was lower than that obtained in our study [30]. Given that the HOMA-IR formula only includes fasting glucose and insulin, mainly related to insulin resistance in the liver, it seems complicated to obtain a single HOMA-IR cut-off point for all pre-diabetic subjects.
Since insulin levels and glucose concentrations are the only measurements needed to calculate HOMA-IR and HOMA-B, they have become the most widely used surrogate indicators, providing valuable insights into insulin resistance, b-cell function, and glucose metabolism [26, 31, 32]. However, in our findings, these were not good criteria for predicting the incidence of diabetes/pre-diabetes, emphasizing that HOMA should not be considered exclusively in the framework of clinical practice. Using other clinical features of subjects or indices such as triglyceride-glucose index [33], which has been shown that are important in predicting metabolic syndrome alongside HOMA-B/HOMA-IR, could help to decide more precisely for individuals concerning the prediction of pre-diabetes/diabetes in the future. Nonetheless, significant differences in insulin levels have been reported between different populations and ethnicities [34], and further research may be needed to re-evaluate our findings in other populations.
This study has some limitations and benefits. The main limitation of this study is the inability to use more precise methods to measure insulin resistance and its secretion (such as the clamp technique). Other limitations include the lack of information on HbA1C and missing participants during the study. Although the technique used in this study for measuring insulin (Electrochemical Luminescence) is currently one of the most precise methods, since there is no reference method for measuring insulin and the results of insulin measurement by different methods can vary up to 2 times, the method of insulin measurement should be considered while using the results [35].
The advantages of this study are as follows: It is the only study that evaluated the HOMA-IR cut-off point to predict the incidence of pre-diabetes. This study was performed in a high sample size and is the only study that prospectively investigates the association between changes in HOMA-B and HOMA-IR with the incidence of diabetes and pre-diabetes subtypes. In this study, the data analysis was performed using a multinomial logistic regression method, suitable for investigating the concurrent relationship between HOMA-B and HOMA-IR with several outcomes (iIFG, iIGT, CGI, and DM). This method seems to be more appropriate for investigating this relationship than the COX method used in the previous study.
In conclusion, this study showed that increased HOMA-IR in normoglycemic individuals is associated with increased incidence of pre-diabetes in both iIFG and iIGT subtypes, while increased HOMA-B leads to a higher risk of develo** iIGT and lower risk of iIFG. There was no significant difference between the incidence of iIGT and iIFG with increased HOMA-IR. Based on the results of this study, it seems that cut-off points of HOMA-IR and HOMA-B are not suitable criteria for predicting the incidence of pre-diabetes.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Yip WC, Sequeira IR, Plank LD, Poppitt SD. Prevalence of pre-diabetes across ethnicities: a review of impaired fasting glucose (IFG) and impaired glucose tolerance (IGT) for classification of dysglycaemia. Nutrients. 2017;9(11):1273.
Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14(2):88–98.
Chen L, Magliano DJ, Zimmet PZ. The worldwide epidemiology of type 2 diabetes mellitus—present and future perspectives. Nat Rev Endocrinol. 2012;8(4):228–36.
Perreault L, Færch K. Approaching pre-diabetes. J Diabetes Complications. 2014;28(2):226–33.
Harati H, Hadaegh F, Saadat N, Azizi F. Population-based incidence of type 2 diabetes and its associated risk factors: results from a six-year cohort study in Iran. BMC Public Health. 2009;9(1):1–8.
Hadaegh F, Derakhshan A, Zafari N, Khalili D, Mirbolouk M, Saadat N, et al. Pre-diabetes tsunami: incidence rates and risk factors of pre-diabetes and its different phenotypes over 9 years of follow-up. Diabet Med. 2017;34(1):69–78.
Saydah SH, Loria CM, Eberhardt MS, Brancati FL. Subclinical states of glucose intolerance and risk of death in the US. Diabetes Care. 2001;24(3):447–53.
Brannick B, Dagogo-Jack S. Prediabetes and cardiovascular disease: pathophysiology and interventions for prevention and risk reduction. Endocrinol Metab Clin. 2018;47(1):33–50.
Brannick B, Wynn A, Dagogo-Jack S. Prediabetes as a toxic environment for the initiation of microvascular and macrovascular complications. Exp Biol Med. 2016;241(12):1323–31.
Saydah SH, Miret M, Sung J, Varas C, Gause D, Brancati FL. Postchallenge hyperglycemia and mortality in a national sample of US adults. Diabetes Care. 2001;24(8):1397–402.
Rhee SY, Woo J-T. The prediabetic period: review of clinical aspects. Diabetes Metab J. 2011;35(2):107–16.
Gong Q, Zhang P, Wang J, Ma J, An Y, Chen Y, et al. Morbidity and mortality after lifestyle intervention for people with impaired glucose tolerance: 30-year results of the Da Qing diabetes prevention outcome study. Lancet Diabetes Endocrinol. 2019;7(6):452–61.
Kodama K, Tojjar D, Yamada S, Toda K, Patel CJ, Butte AJ. Ethnic differences in the relationship between insulin sensitivity and insulin response: a systematic review and meta-analysis. Diabetes Care. 2013;36(6):1789–96.
Bando Y, Ushiogi Y, Okafuji K, Toya D, Tanaka N, Fujisawa M. The relationship of fasting plasma glucose values and other variables to 2-h postload plasma glucose in Japanese subjects. Diabetes Care. 2001;24(7):1156–60.
Suzuki H, Fukushima M, Usami M, Ikeda M, Taniguchi A, Nakai Y, et al. Factors responsible for development from normal glucose tolerance to isolated postchallenge hyperglycemia. Diabetes Care. 2003;26(4):1211–5.
Aoyama-Sasabe S, Fukushima M, **n X, Taniguchi A, Nakai Y, Mitsui R, et al. Insulin secretory defect and insulin resistance in isolated impaired fasting glucose and isolated impaired glucose tolerance. J Diabetes Res. 2016;2016(1298601):1–8.
Bloomgarden ZT. Measures of insulin sensitivity. Clin Lab Med. 2006;26(3):611–33.
Tm W, Levy J, Matthews D. Use and abuse of HOMA modelling. Diabetes Care. 2004;27(6):1487–95.
Ghasemi A, Tohidi M, Derakhshan A, Hasheminia M, Azizi F, Hadaegh F. Cut-off points of homeostasis model assessment of insulin resistance, beta-cell function, and fasting serum insulin to identify future type 2 diabetes: Tehran Lipid and Glucose Study. Acta Diabetol. 2015;52(5):905–15.
Salami M, Hosseinpanah F, Azizi F. Correlation of insulin resistance and impaired glucose metabolism in Tehranian adults: Tehran Lipid and Glucose Study. Iran J Endocrinol Metab. 2006;8(3):259–68.
Unwin N, Shaw J, Zimmet P, Alberti K. Impaired glucose tolerance and impaired fasting glycaemia: the current status on definition and intervention. Diabet Med. 2002;19(9):708–23.
Association AD. Introduction: standards of medical care in diabetes—2020. Am Diabetes Assoc. 2020:S1–2.
Azizi F, Hadaegh F, Khalili D, Esteghamati A, Hossein PF, Delavari A, et al. Appropriate definition of metabolic syndrome among Iranian adults: report of the Iranian National Committee of Obesity. 2010.
Muniyappa R, Lee S, Chen H, Quon MJ. Current approaches for assessing insulin sensitivity and resistance in vivo: advantages, limitations, and appropriate usage. Am J Physiol-Endocrinol Metab. 2008;294(1):E15–26.
Meyer C, Pimenta W, Woerle HJ, Van Haeften T, Szoke E, Mitrakou A, et al. Different mechanisms for impaired fasting glucose and impaired postprandial glucose tolerance in humans. Diabetes Care. 2006;29(8):1909–14.
Song Y, Manson JE, Tinker L, Howard BV, Kuller LH, Nathan L, et al. Insulin sensitivity and insulin secretion determined by homeostasis model assessment and risk of diabetes in a multiethnic cohort of women: the Women’s Health Initiative Observational Study. Diabetes Care. 2007;30(7):1747–52.
Tabák AG, Jokela M, Akbaraly TN, Brunner EJ, Kivimäki M, Witte DR. Trajectories of glycaemia, insulin sensitivity, and insulin secretion before diagnosis of type 2 diabetes: an analysis from the Whitehall II study. Lancet. 2009;373(9682):2215–21.
Derakhshan A, Tohidi M, Arshi B, Khalili D, Azizi F, Hadaegh F. Relationship of hyperinsulinaemia, insulin resistance and β-cell dysfunction with incident diabetes and pre-diabetes: the Tehran Lipid And Glucose Study. Diabet Med. 2015;32(1):24–32.
Færch K, Vaag A, Holst JJ, Hansen T, Jørgensen T, Borch-Johnsen K. Natural history of insulin sensitivity and insulin secretion in the progression from normal glucose tolerance to impaired fasting glycemia and impaired glucose tolerance: the Inter99 study. Diabetes Care. 2009;32(3):439–44.
Baek JH, Kim H, Kim KY, Jung J. Insulin resistance and the risk of diabetes and dysglycemia in Korean general adult population. Diabetes Metab J. 2018;42(4):296–307.
Abbasi F, Okeke Q, Reaven GM. Evaluation of fasting plasma insulin concentration as an estimate of insulin action in nondiabetic individuals: comparison with the homeostasis model assessment of insulin resistance (HOMA-IR). Acta Diabetol. 2014;51(2):193–7.
Borai A, Livingstone C, Kaddam I, Ferns G. Selection of the appropriate method for the assessment of insulin resistance. BMC Med Res Methodol. 2011;11(1):1–10.
Son D-H, Lee HS, Lee Y-J, Lee J-H, Han J-H. Comparison of triglyceride-glucose index and HOMA-IR for predicting prevalence and incidence of metabolic syndrome. Nutr Metab Cardiovasc Dis. 2022;32(3):596–604.
Timóteo AT, Miranda F, Carmo MM, Ferreira RC. Optimal cut-off value for homeostasis model assessment (HOMA) index of insulin-resistance in a population of patients admitted electively in a Portuguese cardiology ward. Acta Med Port. 2014;27(4):473–9.
Tohidi M, Arbab P, Ghasemi A. Assay-dependent variability of serum insulin concentrations: a comparison of eight assays. Scand J Clin Lab Invest. 2017;77(2):122–9.
Acknowledgments
We express our thanks to the participants of District 13 of Tehran for their enthusiastic support in this study.
Funding
This study was supported by the Shahid Beheshti University of Medical Sciences (SBMU).
Author information
Authors and Affiliations
Contributions
DK and AH raised the presented idea and designed the study. NSA, KK and MH conducted the analyses. AH, NSA, and MK interpreted the results. MK and DK developed the first draft of the manuscript. KK, DK, MT, FA, and FH critically reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All studies were conducted using standardized approaches, and all methods were carried out in accordance with relevant guidelines and regulations. The ethics committee approved all experimental protocols of Shahid Beheshti University of Medical Sciences. Informed consent was obtained from all patients to be included in the study.
Consent for publication
Not applicable.
Competing interests
The author(s) has/have no competing interests to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1.
Baseline characteristics of study subjects with missing and non-missing data for insulin. Table S2. Baseline characteristics of study subjects subjects who were followed and not followed. Table S3. Odds ratios of incidence of different study outcomes for HOMA-IR and HOMA-B in multiply imputed data.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Khalili, D., Khayamzadeh, M., Kohansal, K. et al. Are HOMA-IR and HOMA-B good predictors for diabetes and pre-diabetes subtypes?. BMC Endocr Disord 23, 39 (2023). https://doi.org/10.1186/s12902-023-01291-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12902-023-01291-9