Abstract
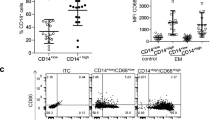
Immunologic disorder has been reported to promote the progression of endometriosis (EMT). It has been known that myeloidderived suppressor cells (MDSCs) drive the progression of many types of diseases. Few studies have shown the relation between MDSCs and EMT. To test whether MDSCs play a role in the progression of EMT, we defined MDSCs, cytokines, and the exosomal microRNA (miRNA) profile in peritoneal fluid (PF) from EMT patients. Characteristics of MDSCs, regulatory T cells (Tregs) and effector T cells were quantified by flow cytometry. Peritoneal fluid monocyte chemoattractant protein (MCP) 1/3, hepatocyte growth factor (HGF), chemokine (C-X-C motif) ligand (CXCL) 1/2, and 13 other cytokines were performed by enzyme-linked immunosorbent assay kit. Exosomal miRNA sequencing was prepared from PF of 3 women with early-stage EMT, 3 women with advanced stage EMT, and 3 women from control group. Our results showed that accumulations of monocytic MDSCs (Mo-MDSCs) and Tregs were detected in advanced patients with EMT. Patients with EMT displayed a significantly higher production of PF CXCL1, CXCL2, MCP-1, MCP-3, and HGF as compared to those from controls. MicroRNA sequencing showed 13 exosomal miRNAs (miRNA-1908, -130b, -451a, -486-5p, -4488, -432, -342, -425, -505, -6508, -145, -365a, and -365b) which are involved in immune alteration and cell proliferation and were differentially expressed in patients with EMT (fold-change ± 2.0). In conclusion, our study revealed that Mo-MDSCs, inflammatory cytokines, and exosomal miRNA seem to be involved in the progression of EMT; however, the relation between Mo-MDSCs, cytokines, and miRNA needs further research.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Domínguez F. Search for new molecular biomarkers to diagnose endometriosis continues. Fertil Steril. 2018;109(4):615–616.
Vercellini P, Viganò P, Somigliana E, Fedele L. Endometriosis: pathogenesis and treatment. Nat Rev Endocrinol. 2014;10(5):261–275.
Koninckx PR, Zupi E, Martin DC. Endometriosis and pregnancy outcome. Fertil Steril. 2018;110(3):406–407.
Sampson JA. Peritoneal endometriosis due to menstrual dissemination of endo-metrial tissues into the peritoneal cavity. Am J Obstet Gynaecol. 1927;14(4):422–469.
Bianco B, André GM, Vilarino FL, et al. The possible role of genetic variants in autoimmune-related genes in the development of endometriosis. Hum Immunol. 2012;73(3):306–315.
Guo SW, Du Y, Liu X. Platelet-derived TGF-β1 mediates the down-modulation of NKG2D expression and may be responsible for impaired natural killer (NK) cytotoxicity in women with endometriosis. Hum Reprod. 2016;31(7):1462–1474.
Gueuvoghlanian-Silva BY, Bellelis P, Barbeiro DF, Hernandes C, Podgaec S. Treg and NK cells related cytokines are associated with deep rectosigmoid endometriosis and clinical symptoms related to the disease. J Reprod Immunol. 2018;126:32–38.
Mier-Cabrera J, González-Gallardo S, Hernández-Guerrero C. Effect of nitric oxide and Th1/Th2 cytokine supplementation over ectopic endometrial tissue growth in a murine model of endometriosis. Reprod Sci. 2013;20(11):1–7.
Kyama CM, Overbergh L, Mihalyi A, et al. Endometrial and peritoneal expression of aromatase, cytokines, and adhesion factors in women with endometriosis. Fertil Steril. 2008;89(2):301–310.
Salmeri FM, Laganà AS, Sofo V, et al. Behavior of tumor necrosis factor-a and tumor necrosis factor receptor 1/tumor necrosis factor receptor 2 system in mononuclear cells recovered from peritoneal fluid of women with endometriosis at different stages. Reprod Sci. 2015;22(2):165–172.
Laganà AS, Vitale SG, Salmeri FM, et al. Unus pro omnibus, omnes pro uno: a novel, evidence-based, unifying theory for the pathogenesis of endometriosis. Med Hypotheses. 2017;103:10–20.
Gogacz M, Gałczyński K, Wojtaś M, et al. Fas-related apoptosis of peritoneal fluid macrophages in endometriosis patients: understanding the disease. J Immunol Res. 2017;2017:3175394.
Sturlese E, Salmeri FM, Retto G, et al. Dysregulation of the Fas/ FasL system in mononuclear cells recovered from peritoneal fluid of women with endometriosis. J Reprod Immunol. 2011;92(1–2):74–81.
Vetvicka V, Laganà AS, Salmeri FM, et al. Regulation of apoptotic pathways during endometriosis: from the molecular basis to the future perspectives. Arch Gynecol Obstet. 2016;294(5):897–904.
Jorgensen H, Hill AS, Beste MT, et al. Peritoneal fluid cytokines related to endometriosis in patients evaluated for infertility. Fertil Steril. 2017;107(5):1191–1199.
Jiang H, Gebhardt C, Umansky L, et al. Elevated chronic inflammatory factors and myeloid-derived suppressor cells indicate poor prognosis in advanced melanoma patients. Int J Cancer. 2015;136(10):2352–2360.
Hassel JC, Jiang H, Bender C, et al. Tadalafil has biologic activity in human melanoma. Results of a pilot trial with tadalafil in patients with metastatic melanoma (TaMe). Oncoimmunology. 2017;6(9):e1326440.
Ben-Meir K, Twaik N, Baniyash M. Plasticity and biological diversity of myeloid derived suppressor cells. Curr Opin Immunol. 2018;51:154–161.
Donnez J, Binda MM, Donnez O, Dolmans MM. Oxidative stress in the pelvic cavity and its role in the pathogenesis of endometriosis. Fertil Steril. 2016;106(5):1011–1017.
Vitale SG, Capriglione S, Peterlunger I, et al. The role of oxidative stress and membrane transport systems during endometriosis: a fresh look at a busy corner. Oxid Med Cell Longev. 2018;2018:7924021.
Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654–659.
Bracamonte-Baran W, Florentin J, Zhou Y, et al. Modification of host dendritic cells by microchimerism-derived extracellular vesicles generates split tolerance. Proc Natl Acad Sci U S A. 2017;114(5):1099–1104.
Marí-Alexandre J, Sánchez-Izquierdo D, Gilabert-Estellés J, Barceló-Molina M, Braza-Boïls A, Sandoval J. miRNAs regulation and its role as biomarkers in endometriosis. Int J Mol Sci. 2016;17(1):E93.
Marí-Alexandre J, Barceló-Molina M, Belmonte-López E, et al. Micro-RNA profile and proteins in peritoneal fluid from women with endometriosis: their relationship with sterility. Fertil Steril. 2018;109(4):675–684.e2.
Zhou M, Fu J, **ao L, et al. miR-196a overexpression activates the MEK/ERK signal and represses the progesterone receptor and decidualization in eutopic endometrium from women with endometriosis. Hum Reprod. 2016;31(11):2598–2608.
Chen H, Qin S, Lei A, et al. Expansion of monocytic myeloidderived suppressor cells in endometriosis patients: a pilot study. Int Immunopharmacol. 2017;47:150–158.
Wang D, Sun H, Wei J, Cen B, DuBois RN. CXCL1 is critical for premetastatic niche formation and metastasis in colorectal cancer. Cancer Res. 2017;77(13):3655–3665.
Zhang H, Ye YL, Li MX, et al. CXCL2/MIF-CXCR2 signaling promotes the recruitment of myeloid-derived suppressor cells and is correlated with prognosis in bladder cancer. Oncogene. 2017;36(15):2095–2104.
Zhang T, Zhou J, Man GCW, et al. MDSCs drive the process of endometriosis by enhancing angiogenesis and are a new potential therapeutic target. Eur J Immunol. 2018;48(6):1059–1073.
Yen BL, Yen ML, Hsu PJ, et al. Multipotent human mesenchymal stromal cells mediate expansion of myeloid-derived suppressor cells via hepatocyte growth factor/c-met and STAT3. Stem Cell Reports. 2013;1(2):139–151.
Jackson AR, Hegde VL, Nagarkatti PS, Nagarkatti M. Characterization of endocannabinoid-mediated induction of myeloid-derived suppressor cells involving mast cells and MCP-1. J Leukoc Biol. 2014;95(4):609–619.
Geis-Asteggiante L, Belew AT, Clements VK, et al. Differential content of proteins, mRNAs, and miRNAs suggests that MDSC and their exosomes may mediate distinct immune suppressive functions. J Proteome Res. 2018;17(1):486–498.
Burke M, Choksawangkarn W, Edwards N, Ostrand-Rosenberg S, Fenselau C. Exosomes from myeloid-derived suppressor cells carry biologically active proteins. J Proteome Res. 2014;13(2):836–843.
Fang F, Song T, Zhang T, Cui Y, Zhang G, **ong Q. MiR-425-5p promotes invasion and metastasis of hepatocellular carcinoma cells through SCAI-mediated dysregulation of multiple signaling pathways. Oncotarget. 2017;8(19):31745–31757.
Wei Y, Nazari-Jahantigh M, Chan L, et al. The microRNA-342- 5p fosters inflammatory macrophage activation through an Akt1- and microRNA-155- dependent pathway during atherosclerosis. Circulation. 2013;127(15):1609–1619.
Ye SB, Li ZL, Luo DH, et al. Tumor-derived exosomes promote tumor progression and T-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma. Oncotarget. 2014;5(14):5439–5452.
Zhang M, Zhou Z, Wang J, Li S. MiR-130b promotes obesity associated adipose tissue inflammation and insulin resistance in diabetes mice through alleviating M2 macrophage polarization via repression of PPAR-g. Immunol Lett. 2016;180:1–8.
Acknowledgments
The authors acknowledge staff from Obstetrics and Gynecology department and staff from Anhui Province Key Laboratory of Reproductive Health and Genetics.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Chen, Y., Wang, K., Xu, Y. et al. Alteration of Myeloid-Derived Suppressor Cells, Chronic Inflammatory Cytokines, and Exosomal miRNA Contribute to the Peritoneal Immune Disorder of Patients With Endometriosis. Reproductive Sciences 26, 1130–1138 (2019). https://doi.org/10.1177/1933719118808923
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719118808923