Abstract
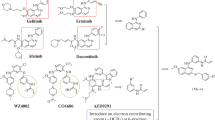
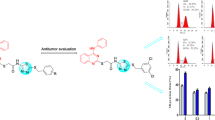
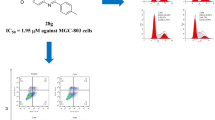
Objective: In order to enrich the library of anti-tumor small molecule compounds, 19 compounds, with highly effective anti-tumor, have been designed and synthesized. Methods: MTT assay was used to detect the anti-proliferation activity of 19 compounds on four human tumor cell lines (PC-3, H1975, A549 and Eca-109). Cell cycle experiment, cell migration experiment, cell clone experiment and cell apoptosis experiment were used to study the anti-tumor mechanism of compound (XIVm). Results: The compound (XIVm) showed the strongest anti-tumor activity against the above four human tumor cells, especially against H1975 cells, with an IC50 value of 1.09 ± 0.04 μM, which was significantly lower than that of 5-fluorouracil (5-FU). The results of a variety of cell experiments showed that the compound (XIVm) significant anti-tumor activity, such as inhibiting the proliferation and migration of H1975 cells, arresting H1975 cells at G2/M phase, and inducing apoptosis in H1975 cells. Discussion: Slight changes in the R group can cause significant changes in the in vitro anti-tumor activity. And when R is a strong electron withdrawing group of cyano and trifluoromethyl, compound (XIVm) exhibits the strongest inhibitory effect, with an IC50 value of 1.09 ± 0.04 μM on H1975 cells. Conclusions: 19 compounds showed significant anti-tumor activity, and the compound (XIVm), with a strong electron withdrawing group of cyano and trifluoromethyl, showed the most significant effect, and the anti-proliferation and anti-migration effects of compound (XIVm) was further investigated.






Similar content being viewed by others
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
Siegel, R.L., Miller, K.D., Fuchs, H.E., and Jemal, A., CA: Cancer J. Clinicians, 2022, vol. 72, pp. 7–33. https://doi.org/10.3322/caac.21708
Lipengolts, A.A., Finogenova, Y.A., Skribitsky, V.A., and Grigorieva, E.Y., J. Physics: Conf. Series, 2021, vol. 2058, p. 011002. https://doi.org/10.1088/1742-6596/2058/1/012039
Tao, D., Xu, J., Zou, S., Tan, Y., and Ai, S., Medicine (Baltimore), 2021, vol. 100, p. e23471. https://doi.org/10.1097/MD.0000000000023471
Sato, C., Okuda, K., Tamiya, H., Yamamoto, K., Hoshina, K., Narumoto, O., Urushiyama, H., Noguchi, S., Amano, Y., Watanabe, K., Mitani, A., Kage, H., Tanaka, G., Yamauchi, Y., Takai, D., and Nagase, T., Intern. Med., 2018, vol. 57, pp. 557–561. https://doi.org/10.2169/internalmedicine.8996-17
Xu, W., Yang, Z., and Lu, N., J. Exp. Clin. Cancer Res., 2016, vol. 35, p. 1. https://doi.org/10.1186/s13046-015-0276-9
Li, X., Xu, J., **e, J., and Yang, W., Chin. Med. J. (Engl.), 2022, vol. 135, pp. 1299–1313. https://doi.org/10.1097/CM9.0000000000002185
Lian, Z., Sang, C., Li, N., Zhai, H., and Bai, W., Front Pharmacol., 2023, vol. 14, p. 1124895. https://doi.org/10.3389/fphar.2023.1124895
Rehuman, N.A., Al-Sehemi, A.G., Parambi, D.G.T., Rangarajan, T.M., Nicolotti, O., Kim, H., and Mathew, B., ChemistrySelect, 2021, vol. 6, pp. 7162–7182. https://doi.org/10.1002/slct.202101077
Wang, D. and Gao, F., Chem. Central J., 2013, vol. 7, p. 95. https://doi.org/10.1186/1752-153X-7-95
Cohen, M.H., Williams, G.A., Sridhara, R., Chen, G., McGuinn, W.D.Jr., Morse, D., Abraham, S., Rahman, A., Liang, C., Lostritto, R., Baird, A., and Pazdur, R., Clinical Cancer Res., 2004, vol. 10, pp. 1212–1218. https://doi.org/10.1158/1078-0432.CCR-03-0564
Verhaeghe, P., Dumetre, A., Castera-Ducros, C., Hutter, S., Laget, M., Fersing, C., Prieri, M., Yzombard, J., Sifredi, F., Rault, S., Rathelot, P., Vanelle, P., and Azas, N., Bioorg. Med. Chem. Lett., 2011, vol. 21, pp. 6003–6006. https://doi.org/10.1016/j.bmcl.2011.06.113
Zhang, G., Wang, M., Zhao, J., Wang, Y., Zhu, M., Wang, J., Cen, S., and Wang, Y., Eur. J. Med. Chem., 2020, vol. 206, p. 112706. https://doi.org/10.1016/j.ejmech.2020.112706
Kubacka, M., Kotanska, M., Szafarz, M., Pociecha, K., Waszkielewicz, A.M., Marona, H., Filipek, B., and Mogilski, S., Nutr. Metab. Cardiovasc Dis., 2019, vol. 29, pp. 751–760. https://doi.org/10.1016/j.numecd.2019.04.003
Li, Y., **ao, J., Zhang, Q., Yu, W., Liu, M., Guo, Y., He, J., and Liu, Y., Bioorg. Med. Chem., 2019, vol. 27, pp. 568–577. https://doi.org/10.1016/j.bmc.2018.12.032
Shagufta and Ahmad, I., Med. Chem. Commun., 2017, vol. 8, pp. 871–885. https://doi.org/10.1039/C7MD00097A
Palazzesi, F., Grundl, M.A., Pautsch, A., Weber, A., and Tautermann, C.S., J. Chem. Inf. Model, 2019, vol. 59, pp. 3565–3571. https://doi.org/10.1021/acs.jcim.9b00316
Amaral, D.N., Lategahn, J., Fokoue, H.H., Silva, E.M.B., Sant'Anna, C.M.R., Rauh, D., Barreiro, E.J., Laufer, S., and Lima, L.M., Sci. Rep., 2019, vol. 9, p. 14. https://doi.org/10.1038/s41598-018-36846-7
Heo, Y.A., Drugs, 2018, vol. 78, pp. 693–697. https://doi.org/10.1007/s40265-018-0899-1
Lorthiois, E., Gerspacher, M., Beyer, K.S., Vaupel, A., Leblanc, C., Stringer, R., Weiss, A., Wilcken, R., Guthy, D.A., Lingel, A., Bomio-Confaglia, C., Machauer, R., Rigollier, P., Ottl, J., Arz, D., Bernet, P., Desjonqueres, G., Dussauge, S., Kazic-Legueux, M., Lozac’h, M.A., Mura, C., Sorge, M., Todorov, M., Warin, N., Zink, F., Voshol, H., Zecri, F.J., Sedrani, R.C., Ostermann, N., Brachmann, S.M., and Cotesta, S., J. Med. Chem., 2022, vol. 65, pp. 16173–16203. https://doi.org/10.1021/acs.jmedchem.2c01438
Jia, H.W., Yang, H.L., **ong, Z.L., Deng, M.H., Wang, T., Liu, Y., and Cheng, M., Bioorg. Chem., 2022, vol. 129, p. 106213.
Zhao, Y., Feng, J.H., Ding, H.X., **ong, Y., Cheng, C.H.K., Hao, X.J., Zhang, Y.M., Pan, Y.J., Guéritte, F., Wu, X.M., Bai, H. and Stöckigt, J., J. Natural Prod., 2006, vol. 69, pp. 1145–1152. https://doi.org/10.1021/np050547x
Grela, E., Kozlowska, J., and Grabowiecka, A., Acta. Histochem., 2018, vol. 120, pp. 303–311. https://doi.org/10.1016/j.acthis.2018.03.007
Funding
This work was supported by the the National Natural Science Foundation of China (no. U21A20416).
Author information
Authors and Affiliations
Contributions
The authors GD, YZ, and WS—carried out the experiment, analyzed and interpreted the data, and wrote the manuscript. The authors CL, YF, and WH—contributed to sample preparation. The authors SX and GC—helped to supervise the data analysis. The authors KY, LH, and ZQ—supervised and defend the findings of this work and revised the manuscript.
Corresponding authors
Ethics declarations
This article does not contain any studies involving patients or animals as test objects.
Informed consent was not required for this article. No conflict of interest was declared by the authors.
Additional information
Publisher's Note. Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Dongling, G., Zichen, Y., Shihao, W. et al. Design, Synthesis, and Anti-Tumor Activity Evaluation In Vitro of 4,6,7–Trisubstituted Quinazoline Derivatives Containing Acrylamide Group. Russ J Bioorg Chem 50, 530–543 (2024). https://doi.org/10.1134/S1068162024020031
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162024020031