Abstract
The respiratory tract is a vital, intricate system for several important biological processes including mucociliary clearance, airway conductance, and gas exchange. The Wnt signaling pathway plays several crucial and indispensable roles across lung biology in multiple contexts. This review highlights the progress made in characterizing the role of Wnt signaling across several disciplines in lung biology, including development, homeostasis, regeneration following injury, in vitro directed differentiation efforts, and disease progression. We further note uncharted directions in the field that may illuminate important biology. The discoveries made collectively advance our understanding of Wnt signaling in lung biology and have the potential to inform therapeutic advancements for lung diseases.
Similar content being viewed by others
Introduction
The lung is a structurally and functionally intricate organ, with over 40 different, known cell types1. The advent of single-cell sequencing and other technologies continues to increase this number and contributes to the striking complexity of the lung. At its most proximal portion, the cartilaginous conducting airways harbor a pseudostratified mucociliary epithelium that plays a vital role in host defense. Inhaled harmful particles, pathogenic organisms, and debris are expectorated from the airways via a process known as mucociliary clearance (MCC). Inhaled contents are first entrapped by a layer of mucus, produced by goblet cells, that are then transported proximally by the unidirectional beating of cilia from terminal bronchioles to the trachea2. The coordinated removal of debris by ciliated cells and mucus-producing goblet cells is facilitated by lubrication from a periciliary water layer. The concerted undertaken by these multiple cell types together comprise the mucociliary escalator2. The bronchioles, also referred to as the conducting airways, are additionally important for moving gases to and from the distal lung, which contain the alveolar sacs necessary for gas exchange. Atmospheric oxygen undergoes exchange for blood carbon dioxide, a process that promotes cellular respiration for all tissues of the body.
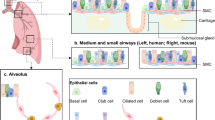
The wingless related-integration site (Wnt)/β-catenin signaling pathway plays an instrumental role in stem cell self-renewal across several tissue epithelia3. R-spondin ligands are cysteine-rich glycoproteins that bind to their cognate leucine-rich repeat-containing G-protein coupled receptor (LGR) LGR4/5/6 receptors and E3 ubiquitin ligases ring finger protein (RNF)43/ZNRF3 via their furin-like domains4,5. In the absence of R-spondins, RNF43/ZNR43 ubiquitinate the Frizzled receptors that targets them for degradation (Fig. 1a, b). As such, Wnt signaling is dampened. Under canonical conditions in the absence of Wnt ligand, β-catenin is in complex with several other proteins including Adenomatous polyposis coli (APC), Glycogen synthase kinase-3a, Glycogen synthase kinase-3β (GSK3β), casein kinase I (CKI), Axin, and Disheveled, among others (Fig. 1a)3. These proteins together comprise a destruction complex. CKIγ first phosphorylates β-catenin at residue serine 45, a priming event that allows for recognition by GSK3β, which phosphorylates β-catenin at serine 33, serine 37, and threonine 413. These N-terminal post-translational modifications mark β-catenin for ubiquitination and subsequent proteasomal degradation by the E3 ubiquitin ligase β-transducin repeat-containing protein (Fig. 1a).
a In the absence of RSPO binding to LGR4/5/6, ubiquitin ligase ZNRF43/RNF43 ubiquitinates the Frizzled receptor which leads to receptor complex endocytosis, β-catenin degradation and subsequent inhibition of Wnt-driven transcriptional activity5. b The binding of RSPO to LGR4/5/6 potentiates Wnt signaling by removing ZNR43/RNF43 ubiquitin ligase from the cell membrane, which would otherwise mark Frizzled receptor for ubiquitination. Frizzled receptors are then able to interact with both Wnt ligand and LRP5/6 co-receptor to drive Wnt signaling cascade. β-catenin then escapes cytoplasmic proteasomal degradation, resulting in its nuclear translocation, interactions with transcription factors. TCF/LEF, and subsequent transactivation of Wnt target genes like c-MYC, CyclinD1, and Axin25.
However, upon Wnt transcription and translation, the ligand enters to the endoplasmic reticulum (ER) and encounters Porcupine, an ER-resident protein that palmitoylates N-terminal cysteine residues of Wnt proteins. This lipid tail modification has been shown to be necessary for Wnt ligand secretion6,7. Wnt ligand then binds to LRP5/6 and appropriate Frizzled co-receptor on the same or neighboring cell. Moreover, R-spondins can potentiate and amplify Wnt signaling by forming complexes with their cognate LGR pair and binding to RNF43/ZNR43 to prevent Frizzled receptor ubiquitination. Together, these events allow for a cascade of molecular events that results in disassembly of the cytoplasmic destruction complex (Fig. 1b). β-catenin then accumulates in the cytoplasm, subsequently translocates to the nucleus, and interacts with transcription factors T-cell factor/Lymphoid enhancer factor 1 (TCF/LEF) (Fig. 1b). In this way, β-catenin drives transactivation of downstream target genes such as c-MYC, AXIN2, and CYCLIN D1 among others (Fig. 1b). The canonical Wnt signaling described above is important in a variety of cellular processes including proliferation, self-renewal, epithelial to mesenchymal transitions, and migration and motility.
In contrast, non-canonical Wnt signaling is often thought of as the β-catenin-independent pathway. One arm of this pathway regulates planar cell polarity (PCP), during which Frizzled receptors trigger downstream activation of RhoA and Rac GTPases that promote cytoskeletal remodeling8. A second arm of non-canonical Wnt signaling lies in Wnt-Frizzled binding that triggers Phospholipase C and downstream Ca2+ activity for regulation of cell migration and fate decisions8. It is important to note that Porcupine palmitoylates all 19 mammalian Wnt ligands and is therefore necessary for their secretion, including those that partake in non-canonical signaling.
Over the past several years, much research has been put forth toward carefully dissecting the nuanced role of Wnt signaling across several disciplines pertaining to lung biology. This review aims to highlight the major contributions made to our current understanding of the Wnt signaling pathway in lung and airway development, its role in proximal and distal airway homeostasis and relevant niche biology, as well as its role in directed differentiation efforts of induced pluripotent stem cells (iPSCs) and embryonic stem cells (ESCs) to the lung lineage, in organoid culture models, and its perturbations in disease states.
Development
Detailed staging and mechanisms of both human and murine lung development have been well reviewed by others9,10,11,12. To briefly summarize murine lung development, during the embryonic stage, the ventral anterior foregut endoderm (AFE) expresses the transcription factor Nkx2.1 at mouse embryonic day (E) 9.0 as a sign of the specification to promote initial lung budding. From E9.5–E12.5, two lung buds with high Nkx2.1 expression and a proximal portion with low Nkx2.1 that later forms the trachea emerge concomitantly with tracheo-esophageal septation. From E12.5–E16.5 during the pseudoglandular stage, the lung buds undergo a period of branching morphogenesis to form the lung tree and terminal bronchioles. Upon completion of the canalicular and saccular stages of development (E16.5-Postnatal (P) day 4), the terminal bronchioles narrow and begin to form epithelial sacs. These structures later form fully mature alveolar structures for gas exchange by P21 during the alveolarization phase9. In contrast, alveolarization begins pre-partum during human lung development and continues postnatally into childhood13.
Coordinated development of the conducting and distal airways is a vital process during which both the epithelial and mesenchymal compartments play integral roles. The develo** lung endodermal buds penetrate the splanchnic mesoderm and mesothelium around E9.5. The develo** distal lung then acquires four distinct layers, each with its own unique anatomical, cellular, morphologic, and molecular profiles: endoderm (epithelium), subepithelial mesoderm (mesenchyme), submesothelial mesoderm (mesenchyme), and mesothelium. Transient amplifying submesothelial cells give rise to a parabronchial smooth muscle cell (PSMC) progenitor population. This cell population then migrates more proximally around the bronchi and differentiates into smooth muscle cells (SMCs)14,15. Studies assessing the expression levels of various Wnt/β-catenin signaling members and activity reporters in these aforementioned compartments during lung development have identified contrasting findings16,17. However, a myriad of studies collectively demonstrates that the develo** lung mesenchyme displays several highly regulated interactions with the lung endoderm in both mouse and human that together coordinate normal lung organogenesis9, many of which are Wnt-mediated.
Embryonic stage (E9.5–E12.5)
Wnt signaling plays a role in some of the earliest stages of cardiopulmonary specification. Wnt2+ Gli1+ Isl1+ cells comprise the multipotent cardiopulmonary mesoderm progenitors (CPPs) that orchestrate heart and lung development. Lineage tracing of Wnt2+ CPPs at E8.5 demonstrates their capacity to generate the cardiac inflow tract and pulmonary mesoderm cell lineage by E17.518. These cells are important for the vital epithelial–mesenchymal interactions that occur during lung development.
Crosstalk from develo** mesenchyme to endoderm
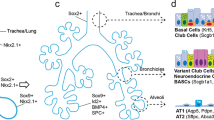
Wnt-driven mesenchymal-to-endodermal crosstalk is critically important from the earliest stages of lung development. From E9.5–12.5, there is active Wnt/β-catenin signaling in both the epithelium and the mesenchyme adjacent to the future proximal airway as measured by TOPGAL and AXIN2-LacZ Wnt activity reporters19,20. Canonical Wnt2/2b ligands are spatiotemporally regulated by Hox5 genes during this stage, with notable mesodermal expression near the ventral aspect of the anterior foregut between E9.0 and E10.521 (Fig. 2a). Together, Wnt2/2b cooperate to promote Nkx2.1+ lung endodermal specification (Fig. 2a), as mice without them display lung agenesis22,23,24. Wnt2/2b converges on canonical signaling in the develo** endoderm, as deletion of β-catenin in the anterior foregut also display lung agenesis, resulting in the upregulation of a Sox2+ digestive progenitor identity19,22. In contrast, constitutive activation of β-catenin prevents tracheo-esophageal septation and instead drives Nkx2.1+ lung endodermal progenitor expansion19,22.
a During the embryonic stage (E9.0–12.5) of development, lung bud emerges from tracheal-esophageal septation occurs9. Mesenchymal (brown) HOX5 spatiotemporally regulates endodermal (pink) Wnt2/2b to establish NKX2.1 lung progenitor via downstream β-catenin signaling21,22,23,24. Endodermal Wnt ligands also promote mesenchymal FGF10 and β-catenin, which then allow for SMC differentiation and cartilage and basal cell development27. b During the pseudoglandular stage (E12.5–E16.5), lung buds undergo branching morphogenesis to develop terminal bronchioles9. Mesenchymal Wnt5a promotes tracheal and cartilage formation via ROR2-dependent mechanisms33. Wntless (Wls)-regulated Notum suppresses mesenchymal Wnt and is necessary for tracheal development and branching morphogenesis39,40. A Wnt7b-BMP4 signaling axis also promotes epithelial proliferation and mesenchymal vascular SMC (VSMC) differentiation and SMC proliferation35,36,37,38. Further, epithelial Wnt5a expression is highest in distal tips25. Similar to constitutive β-catenin activation, deletion of Barx1 results in the loss of tracheo-esophageal septation as evidenced by single, contiguous luminal layer of Nkx2.1+ tracheal and Sox2+ esophageal epithelium by E10.519,22,25.
Crosstalk from develo** endoderm to mesenchyme
As early as E11.5, canonical Wnt2 ligand signaling promotes fibroblast growth factor 10 (Fgf10) signaling that, in turn, facilitates differentiation of immature platelet-derived growth factor receptor (Pdgfr)α/β+ smooth muscle cells by regulating the expression of myocardin and Mrtf-B, critical transcription factors in myogenesis26 (Fig. 2a). Mesenchymal Wnt2 also promotes endodermal Wnt7b expression that later signals back to the subepithelial mesenchyme to further drive SMC differentiation26. More recently, epithelial Wnt ligands were shown to activate mesenchymal β-catenin, which alongside Fgf10/Fgfr2, regulate both cartilage progenitors and basal cell development27 (Fig. 2a).
Pseudoglandular stage (E12.5–E16.5)
Constitutive activation of β-catenin in surfactant protein C (Sftpc)+ cells drives distal conducting airway dilatation concomitant with no differentiation to either the secretory or ciliated cell fate28. Consistent with this, R-spondin 2 (Rspo2) facilitates normal embryonic lung growth and the start of branching morphogenesis by potentiating Wnt/β-catenin signaling29. A recent study identified, however, that Rspo2 antagonizes RNF43 and zinc and ring finger (ZNRF)3 to regulate limb and lung formation in xenopus4. These efforts resulted in a paradigm shift, demonstrating that Rspo2 can potentiate Wnt signaling in the absence of LGR4,30. Interestingly, while Rspo2 drives branching morphogenesis earlier in development, it has no role in the differentiation of epithelial or mesenchymal cell types29. This suggests functional redundancy in the varying upstream regulators of Wnt signaling in embryonic lung development.
Several Wnt ligands have been well characterized in the pseudoglandular stage. Wnt5a is diffusely expressed in both the epithelium and mesenchyme as early as E12.067,68,69. The bronchoalveolar duct junction (BADJ) houses the Scgb1a1+ Sftpc+ Sca1+ bronchoalveolar stem cell (BASC)70,71. Further, Sftpc+ ATII cells in the alveolar region undergo self-renewal and give rise to ATI cells72. Most recently, a family of stem/progenitor cells referred to as the lineage-negative epithelial progenitors (LNEPs) have been characterized as quiescent at homeostasis but are mobilized under injurious conditions to regenerate the lungs73,74. Although the intricate complexity of the identity of these stem cell progenitors continues to be an area of current investigation, Wnt signaling plays critical roles in several of these regional compartments in regeneration.
From a cell biology perspective, several of the aforementioned identified progenitor populations or non-stem supporting niche cells employ Wnt signaling in bronchiolar, bronchioalveolar, or alveolar regeneration. Although an early study reported that β-catenin in club cells is dispensable for bronchiolar epithelial repair75, several subsequent findings indicate a highly important and intricately regulated role for this pathway at the cellular level in repair.
In the bronchioles and BADJ following naphthalene injury, ciliated cells induce Wnt7b expression that signals to the PSMCs to induce Fgf10 expression76,77 (Fig. 3c). Mesenchymal Fgf10 then induces Ak strain transforming (AKT)-mediated phosphorylation of β-catenin at S552 to promote BASC expansion and subsequent epithelial regeneration78 (Fig. 3c). Constitutive activation of β-catenin increases bronchiolar stem cell expansion and attenuates differentiation79. β-catenin stabilization also skews sex-determining region Y-box 2 (Sox2)+ LNEPs toward an ATII-like rather than K5+ cell fate by inhibiting Notch and hypoxia signaling following influenza infection139. BPD also shares a common downstream activated, phosphorylated form of β-catenin at Y489 that is also observed in IPF127,140, though much remains to be explored in this disease context. COPD also displays perturbed non-canonical Wnt signaling biology, as pulmonary fibroblasts secrete Wnt5a to inhibit alveolar canonical Wnt/β-catenin signaling, thereby preventing epithelial repair141. Recent work has also begun to think about the role of Wnt signaling in the context of aging as well. Lehmann et al. reported that aged ATII cells exhibit increase senescence that is driven by activation of Wnt/β-catenin signaling and is associated with profibrotic changes142.
Prospective/discussion of Wnt signaling in disease biology
The work of Nabhan et al. has paved the way for understanding the role of single-cell niches within distal lung regeneration83. The success of this lies, in part, because its generated understanding of the Wnt-producing (Wnt-expressing, Porcupine-positive) cells and the Wnt-responsive (Axin2+) cells via single-cell sequencing studies. This approach should be adopted in the context of disease processes, as it could hold particular promise with disease processes with known intermediates such as the malignant transformation underpinning lung squamous cell carcinoma. There also exists a plethora of outstanding questions largely underexplored within the sphere of aging. For example, how do stem cell niches throughout the airway and lung change with aging, both at the cellular and molecular levels? And how, if at all, do the alterations in Wnt signaling that occur in aging relate to distinct pathophysiology of disease processes?
Summary
Taken together, it is clear that Wnt signaling plays a major role in lung development, lung repair and regeneration, and the progression of many lung diseases. The rapid technological advances in the fields of molecular and cellular biology are greatly facilitating the study of Wnt signaling in lung biology. Advancing our knowledge on the exact mechanisms of Wnt signaling in the lung will allow for the development of more Wnt pathway targeted therapies that will hopefully lead to a therapeutic benefit for patients with lung diseases.
References
Cardoso, W. V. & Whitsett, J. A. Resident cellular components of the lung: developmental aspects. Proc. Am. Thorac. Soc. 5, 767–771 (2008).
Bustamante-Marin, X. M. & Ostrowski, L. E. Cilia and mucociliary clearance. Cold Spring Harb. Perspect. Biol. 9, a028241 (2017).
Nusse, R. & Clevers, H. Wnt/beta-catenin signaling, disease, and emerging therapeutic modalities. Cell 169, 985–999 (2017).
Szenker-Ravi, E. et al. RSPO2 inhibition of RNF43 and ZNRF3 governs limb development independently of LGR4/5/6. Nature 557, 564–569 (2018).
Clevers, H., Loh, K. M. & Nusse, R. Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science 346, 1248012 (2014).
Barrott, J. J., Cash, G. M., Smith, A. P., Barrow, J. R. & Murtaugh, L. C. Deletion of mouse Porcn blocks Wnt ligand secretion and reveals an ectodermal etiology of human focal dermal hypoplasia/Goltz syndrome. Proc. Natl Acad. Sci. USA 108, 12752–12757 (2011).
Biechele, S., Cox, B. J. & Rossant, J. Porcupine homolog is required for canonical Wnt signaling and gastrulation in mouse embryos. Dev. Biol. 355, 275–285 (2011).
Gomez-Orte, E., Saenz-Narciso, B., Moreno, S. & Cabello, J. Multiple functions of the noncanonical Wnt pathway. Trends Genet. 29, 545–553 (2013).
Herriges, M. & Morrisey, E. E. Lung development: orchestrating the generation and regeneration of a complex organ. Development 141, 502–513 (2014).
Morrisey, E. E. & Hogan, B. L. Preparing for the first breath: genetic and cellular mechanisms in lung development. Dev. Cell 18, 8–23 (2010).
Schittny, J. C. Development of the lung. Cell Tissue Res. 367, 427–444 (2017).
Nikolic, M. Z., Sun, D. & Rawlins, E. L. Human lung development: recent progress and new challenges. Development 145, dev163485 (2018).
Warburton, D. et al. Lung organogenesis. Curr. Top. Dev. Biol. 90, 73–158 (2010).
Yin, Y. et al. An FGF-WNT gene regulatory network controls lung mesenchyme development. Dev. Biol. 319, 426–436 (2008).
De Langhe, S. P. et al. Formation and differentiation of multiple mesenchymal lineages during lung development is regulated by beta-catenin signaling. PLoS ONE 3, e1516 (2008).
Al Alam, D. et al. Contrasting expression of canonical Wnt signaling reporters TOPGAL, BATGAL and Axin2(LacZ) during murine lung development and repair. PLoS ONE 6, e23139 (2011).
Tebar, M., Destree, O., de Vree, W. J. & Ten Have-Opbroek, A. A. Expression of Tcf/Lef and sFrp and localization of beta-catenin in the develo** mouse lung. Mech. Dev. 109, 437–440 (2001).
Peng, T. et al. Coordination of heart and lung co-development by a multipotent cardiopulmonary progenitor. Nature 500, 589–592 (2013).
Harris-Johnson, K. S., Domyan, E. T., Vezina, C. M. & Sun, X. beta-Catenin promotes respiratory progenitor identity in mouse foregut. Proc. Natl Acad. Sci. USA 106, 16287–16292 (2009).
De Langhe, S. P. et al. Dickkopf-1 (DKK1) reveals that fibronectin is a major target of Wnt signaling in branching morphogenesis of the mouse embryonic lung. Dev. Biol. 277, 316–331 (2005).
Hrycaj, S. M. et al. Hox5 genes regulate the Wnt2/2b-Bmp4-signaling axis during lung development. Cell Rep. 12, 903–912 (2015).
Goss, A. M. et al. Wnt2/2b and beta-catenin signaling are necessary and sufficient to specify lung progenitors in the foregut. Dev. Cell 17, 290–298 (2009).
Poulain, M. & Ober, E. A. Interplay between Wnt2 and Wnt2bb controls multiple steps of early foregut-derived organ development. Development 138, 3557–3568 (2011).
Rankin, S. A., Gallas, A. L., Neto, A., Gomez-Skarmeta, J. L. & Zorn, A. M. Suppression of Bmp4 signaling by the zinc-finger repressors Osr1 and Osr2 is required for Wnt/beta-catenin-mediated lung specification in Xenopus. Development 139, 3010–3020 (2012).
Woo, J., Miletich, I., Kim, B. M., Sharpe, P. T. & Shivdasani, R. A. Barx1-mediated inhibition of Wnt signaling in the mouse thoracic foregut controls tracheo-esophageal septation and epithelial differentiation. PLoS ONE 6, e22493 (2011).
Goss, A. M. et al. Wnt2 signaling is necessary and sufficient to activate the airway smooth muscle program in the lung by regulating myocardin/Mrtf-B and Fgf10 expression. Dev. Biol. 356, 541–552 (2011).
Hou, Z. et al. Wnt/Fgf crosstalk is required for the specification of basal cells in the mouse trachea. Development 146, dev171496 (2019).
Hashimoto, S. et al. beta-Catenin-SOX2 signaling regulates the fate of develo** airway epithelium. J. Cell Sci. 125, 932–942 (2012).
Bell, S. M. et al. R-spondin 2 is required for normal laryngeal-tracheal, lung and limb morphogenesis. Development 135, 1049–1058 (2008).
Lebensohn, A. M. & Rohatgi, R. R-spondins can potentiate WNT signaling without LGRs. Elife 7 (2018).
Li, C., **ao, J., Hormi, K., Borok, Z. & Minoo, P. Wnt5a participates in distal lung morphogenesis. Dev. Biol. 248, 68–81 (2002).
Yamaguchi, T. P., Bradley, A., McMahon, A. P. & Jones, S. A Wnt5a pathway underlies outgrowth of multiple structures in the vertebrate embryo. Development 126, 1211–1223 (1999).
Kishimoto, K. et al. Synchronized mesenchymal cell polarization and differentiation shape the formation of the murine trachea and esophagus. Nat. Commun. 9, 2816 (2018).
Li, C. et al. Wnt5a regulates Shh and Fgf10 signaling during lung development. Dev. Biol. 287, 86–97 (2005).
Shu, W., Jiang, Y. Q., Lu, M. M. & Morrisey, E. E. Wnt7b regulates mesenchymal proliferation and vascular development in the lung. Development 129, 4831–4842 (2002).
Weidenfeld, J., Shu, W., Zhang, L., Millar, S. E. & Morrisey, E. E. The WNT7b promoter is regulated by TTF-1, GATA6, and Foxa2 in lung epithelium. J. Biol. Chem. 277, 21061–21070 (2002).
Rajagopal, J. et al. Wnt7b stimulates embryonic lung growth by coordinately increasing the replication of epithelium and mesenchyme. Development 135, 1625–1634 (2008).
Cohen, E. D. et al. Wnt signaling regulates smooth muscle precursor development in the mouse lung via a tenascin C/PDGFR pathway. J. Clin. Investig. 119, 2538–2549 (2009).
Gerhardt, B. et al. Notum attenuates Wnt/beta-catenin signaling to promote tracheal cartilage patterning. Dev. Biol. 436, 14–27 (2018).
Cornett, B. et al. Wntless is required for peripheral lung differentiation and pulmonary vascular development. Dev. Biol. 379, 38–52 (2013).
Yates, L. L. et al. The PCP genes Celsr1 and Vangl2 are required for normal lung branching morphogenesis. Hum. Mol. Genet. 19, 2251–2267 (2010).
Zhang, Y. et al. A Gata6-Wnt pathway required for epithelial stem cell development and airway regeneration. Nat. Genet. 40, 862–870 (2008).
Mucenski, M. L. et al. beta-Catenin is required for specification of proximal/distal cell fate during lung morphogenesis. J. Biol. Chem. 278, 40231–40238 (2003).
Shu, W. et al. Wnt/beta-catenin signaling acts upstream of N-myc, BMP4, and FGF signaling to regulate proximal-distal patterning in the lung. Dev. Biol. 283, 226–239 (2005).
Okubo, T. & Hogan, B. L. Hyperactive Wnt signaling changes the developmental potential of embryonic lung endoderm. J. Biol. 3, 11 (2004).
Mucenski, M. L. et al. Beta-catenin regulates differentiation of respiratory epithelial cells in vivo. Am. J. Physiol. Lung Cell Mol. Physiol. 289, L971–L979 (2005).
Frank, D. B. et al. Emergence of a wave of Wnt signaling that regulates lung alveologenesis by controlling epithelial self-renewal and differentiation. Cell Rep. 17, 2312–2325 (2016).
Lynch, T. J. et al. Submucosal gland myoepithelial cells are reserve stem cells that can regenerate mouse tracheal epithelium. Cell Stem Cell 22, 779 (2018).
Montoro, D. T. et al. A revised airway epithelial hierarchy includes CFTR-expressing ionocytes. Nature 560, 319–324 (2018).
Hegab, A. E. et al. Isolation and in vitro characterization of basal and submucosal gland duct stem/progenitor cells from human proximal airways. Stem Cells Transl. Med. 1, 719–724 (2012).
Tata, A. et al. Myoepithelial cells of submucosal glands can function as reserve stem cells to regenerate airways after injury. Cell Stem Cell 22, 668–683.e666 (2018).
Duan, D. et al. Submucosal gland development in the airway is controlled by lymphoid enhancer binding factor 1 (LEF1). Development 126, 4441–4453 (1999).
Duan, D., Sehgal, A., Yao, J. & Engelhardt, J. F. Lef1 transcription factor expression defines airway progenitor cell targets for in utero gene therapy of submucosal gland in cystic fibrosis. Am. J. Respir. Cell Mol. Biol. 18, 750–758 (1998).
Ritchie, T. C. et al. Developmental expression of catenins and associated proteins during submucosal gland morphogenesis in the airway. Exp. Lung Res. 27, 121–141 (2001).
Driskell, R. R. et al. Wnt3a regulates Lef-1 expression during airway submucosal gland morphogenesis. Dev. Biol. 305, 90–102 (2007).
Driskell, R. R. et al. Wnt-responsive element controls Lef-1 promoter expression during submucosal gland morphogenesis. Am. J. Physiol. Lung Cell Mol. Physiol. 287, L752–L763 (2004).
**e, W. et al. Sox2 modulates Lef-1 expression during airway submucosal gland development. Am. J. Physiol. Lung Cell Mol. Physiol. 306, L645–L660 (2014).
Liu, X. et al. Sox17 modulates Wnt3A/beta-catenin-mediated transcriptional activation of the Lef-1 promoter. Am. J. Physiol. Lung Cell Mol. Physiol. 299, L694–L710 (2010).
Lynch, T. J. et al. Wnt signaling regulates airway epithelial stem cells in adult murine submucosal glands. Stem Cells 34, 2758–2771 (2016).
Rock, J. R., Randell, S. H. & Hogan, B. L. Airway basal stem cells: a perspective on their roles in epithelial homeostasis and remodeling. Dis. Model Mech. 3, 545–556 (2010).
Brechbuhl, H. M. et al. beta-catenin dosage is a critical determinant of tracheal basal cell fate determination. Am. J. Pathol. 179, 367–379 (2011).
Giangreco, A. et al. beta-Catenin determines upper airway progenitor cell fate and preinvasive squamous lung cancer progression by modulating epithelial-mesenchymal transition. J. Pathol. 226, 575–587 (2012).
Hsu, H. S. et al. Repair of naphthalene-induced acute tracheal injury by basal cells depends on beta-catenin. J. Thorac. Cardiovasc. Surg. 148, 322–332 (2014).
Aros, C. J. et al. Distinct spatiotemporally dynamic Wnt-secreting niches regulate proximal airway regeneration and aging. Cell Stem Cell 27, 413–429.e414 (2020).
Aros, C. J. et al. High-throughput drug screening identifies a potent wnt inhibitor that promotes airway basal stem cell homeostasis. Cell Rep. 30, 2055–2064.e2055 (2020).
Haas, M. et al. DeltaN-Tp63 mediates Wnt/beta-catenin-induced inhibition of differentiation in basal stem cells of mucociliary epithelia. Cell Rep. 28, 3338–3352.e3336 (2019).
Guha, A. et al. Neuroepithelial body microenvironment is a niche for a distinct subset of Clara-like precursors in the develo** airways. Proc. Natl Acad. Sci. USA 109, 12592–12597 (2012).
Rawlins, E. L. et al. The role of Scgb1a1+ Clara cells in the long-term maintenance and repair of lung airway, but not alveolar, epithelium. Cell Stem Cell 4, 525–534 (2009).
Guha, A., Deshpande, A., Jain, A., Sebastiani, P. & Cardoso, W. V. Uroplakin 3a(+) cells are a distinctive population of epithelial progenitors that contribute to airway maintenance and post-injury repair. Cell Rep. 19, 246–254 (2017).
Kim, C. F. et al. Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 121, 823–835 (2005).
Liu, Q. et al. Lung regeneration by multipotent stem cells residing at the bronchioalveolar-duct junction. Nat. Genet. 51, 728–738 (2019).
Barkauskas, C. E. et al. Type 2 alveolar cells are stem cells in adult lung. J. Clin. Investig. 123, 3025–3036 (2013).
Vaughan, A. E. et al. Lineage-negative progenitors mobilize to regenerate lung epithelium after major injury. Nature 517, 621–625 (2015).
Kathiriya, J. J., Brumwell, A. N., Jackson, J. R., Tang, X. & Chapman, H. A. Distinct airway epithelial stem cells hide among club cells but mobilize to promote alveolar regeneration. Cell Stem Cell (2020).
Zemke, A. C. et al. beta-Catenin is not necessary for maintenance or repair of the bronchiolar epithelium. Am. J. Respir. Cell Mol. Biol. 41, 535–543 (2009).
Volckaert, T. et al. Fgf10-Hippo epithelial-mesenchymal crosstalk maintains and recruits lung basal stem cells. Dev. Cell 43, 48–59.e45 (2017).
Volckaert, T. et al. Hippo signaling promotes lung epithelial lineage commitment by curbing Fgf10 and beta-catenin signaling. Development 146, dev166454 (2019).
Volckaert, T. et al. Parabronchial smooth muscle constitutes an airway epithelial stem cell niche in the mouse lung after injury. J. Clin. Investig. 121, 4409–4419 (2011).
Reynolds, S. D. et al. Conditional stabilization of beta-catenin expands the pool of lung stem cells. Stem Cells 26, 1337–1346 (2008).
**, Y. et al. Local lung hypoxia determines epithelial fate decisions during alveolar regeneration. Nat. Cell Biol. 19, 904–914 (2017).
Lee, J. H. et al. Anatomically and functionally distinct lung mesenchymal populations marked by Lgr5 and Lgr6. Cell 170, 1149–1163 e1112 (2017).
Zepp, J. A. et al. Distinct mesenchymal lineages and niches promote epithelial self-renewal and myofibrogenesis in the lung. Cell 170, 1134–1148 e1110 (2017).
Nabhan, A. N., Brownfield, D. G., Harbury, P. B., Krasnow, M. A. & Desai, T. J. Single-cell Wnt signaling niches maintain stemness of alveolar type 2 cells. Science 359, 1118–1123 (2018).
Flozak, A. S. et al. Beta-catenin/T-cell factor signaling is activated during lung injury and promotes the survival and migration of alveolar epithelial cells. J. Biol. Chem. 285, 3157–3167 (2010).
Zemans, R. L. et al. Neutrophil transmigration triggers repair of the lung epithelium via beta-catenin signaling. Proc. Natl Acad. Sci. USA 108, 15990–15995 (2011).
Malleske, D. T., Hayes, D. Jr., Lallier, S. W., Hill, C. L. & Reynolds, S. D. Regulation of human airway epithelial tissue stem cell differentiation by beta-catenin, P300, and CBP. Stem Cells 36, 1905–1916 (2018).
Rieger, M. E. et al. p300/beta-catenin interactions regulate adult progenitor cell differentiation downstream of WNT5a/protein kinase C (PKC). J. Biol. Chem. 291, 6569–6582 (2016).
Mori, M. et al. Generation of functional lungs via conditional blastocyst complementation using pluripotent stem cells. Nat. Med. 25, 1691–1698 (2019).
Tadokoro, T. et al. IL-6/STAT3 promotes regeneration of airway ciliated cells from basal stem cells. Proc. Natl Acad. Sci. USA 111, E3641–E3649 (2014).
Gouon-Evans, V. et al. BMP-4 is required for hepatic specification of mouse embryonic stem cell-derived definitive endoderm. Nat. Biotechnol. 24, 1402–1411 (2006).
D’Amour, K. A. et al. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nat. Biotechnol. 23, 1534–1541 (2005).
Green, M. D. et al. Generation of anterior foregut endoderm from human embryonic and induced pluripotent stem cells. Nat. Biotechnol. 29, 267–272 (2011).
Christodoulou, C. et al. Mouse ES and iPS cells can form similar definitive endoderm despite differences in imprinted genes. The. J. Clin. Investig. 121, 2313–2325 (2011).
Gomperts BN. Induction of multiciliated cells from induced pluripotent stem cells. Proc Natl Acad Sci USA. 111, 6120–6121 (2014).
Mou, H. et al. Generation of multipotent lung and airway progenitors from mouse ESCs and patient-specific cystic fibrosis iPSCs. Cell Stem Cell 10, 385–397 (2012).
Wong, A. P. et al. Directed differentiation of human pluripotent stem cells into mature airway epithelia expressing functional CFTR protein. Nat. Biotechnol. 30, 876–882 (2012).
Huang, S. X. et al. The in vitro generation of lung and airway progenitor cells from human pluripotent stem cells. Nat. Protoc. 10, 413–425 (2015).
Huang, S. X. et al. Efficient generation of lung and airway epithelial cells from human pluripotent stem cells. Nat. Biotechnol. 32, 84–91 (2014).
Sherwood, R. I., Maehr, R., Mazzoni, E. O. & Melton, D. A. Wnt signaling specifies and patterns intestinal endoderm. Mech. Dev. 128, 387–400 (2011).
Hawkins, F. et al. Prospective isolation of NKX2-1-expressing human lung progenitors derived from pluripotent stem cells. J. Clin. Investig. 127, 2277–2294 (2017).
McCauley, K. B. et al. Efficient derivation of functional human airway epithelium from pluripotent stem cells via temporal regulation of Wnt signaling. Cell Stem Cell 20, 844–857 e846 (2017).
Jacob, A. et al. Differentiation of human pluripotent stem cells into functional lung alveolar epithelial cells. Cell Stem Cell 21, 472–488 e410 (2017).
Hurley, K. et al. Reconstructed single-cell fate trajectories define lineage plasticity windows during differentiation of human PSC-derived distal lung progenitors. Cell Stem Cell 26, 593–608.e598 (2020).
Ikonomou, L. et al. The in vivo genetic program of murine primordial lung epithelial progenitors. Nat. Commun. 11, 635 (2020).
Longmire, T. A. et al. Efficient derivation of purified lung and thyroid progenitors from embryonic stem cells. Cell Stem Cell 10, 398–411 (2012).
Serra, M. et al. Pluripotent stem cell differentiation reveals distinct developmental pathways regulating lung- versus thyroid-lineage specification. Development 144, 3879–3893 (2017).
Firth, A. L. et al. Generation of multiciliated cells in functional airway epithelia from human induced pluripotent stem cells. Proc. Natl Acad. Sci. USA 111, E1723–E1730 (2014).
Konishi, S. et al. Directed induction of functional multi-ciliated cells in proximal airway epithelial spheroids from human pluripotent stem cells. Stem Cell Rep. 6, 18–25 (2016).
Gotoh, S. et al. Generation of alveolar epithelial spheroids via isolated progenitor cells from human pluripotent stem cells. Stem Cell Rep. 3, 394–403 (2014).
Dye, B. R. et al. In vitro generation of human pluripotent stem cell derived lung organoids. Elife 4, e05098 (2015).
Miller, A. J. et al. Generation of lung organoids from human pluripotent stem cells in vitro. Nat. Protoc. 14, 518–540 (2019).
Chen, Y. W. et al. A three-dimensional model of human lung development and disease from pluripotent stem cells. Nat. Cell Biol. 19, 542–549 (2017).
Strikoudis, A. et al. Modeling of fibrotic lung disease using 3D organoids derived from human pluripotent stem cells. Cell Rep. 27, 3709–3723.e3705 (2019).
Kim, M. et al. Patient-derived lung cancer organoids as in vitro cancer models for therapeutic screening. Nat. Commun. 10, 3991 (2019).
Sachs, N. et al. Long-term expanding human airway organoids for disease modeling. EMBO J. 38, e100300 (2019).
Ueda, M. et al. Mutations of the beta- and gamma-catenin genes are uncommon in human lung, breast, kidney, cervical and ovarian carcinomas. Br. J. Cancer 85, 64–68 (2001).
Ohgaki, H. et al. APC mutations are infrequent but present in human lung cancer. Cancer Lett. 207, 197–203 (2004).
Shigemitsu, K. et al. Genetic alteration of the beta-catenin gene (CTNNB1) in human lung cancer and malignant mesothelioma and identification of a new 3p21.3 homozygous deletion. Oncogene 20, 4249–4257 (2001).
Ding, L. et al. Somatic mutations affect key pathways in lung adenocarcinoma. Nature 455, 1069–1075 (2008).
Li, C. et al. Stabilized beta-catenin in lung epithelial cells changes cell fate and leads to tracheal and bronchial polyposis. Dev. Biol. 334, 97–108 (2009).
Pacheco-Pinedo, E. C. et al. Wnt/beta-catenin signaling accelerates mouse lung tumorigenesis by imposing an embryonic distal progenitor phenotype on lung epithelium. J. Clin. Investig. 121, 1935–1945 (2011).
Yin, N. et al. Protein kinase ciota and Wnt/beta-catenin signaling: alternative pathways to Kras/Trp53-driven lung adenocarcinoma. Cancer Cell 36, 692–693 (2019).
Nguyen, D. X. et al. WNT/TCF signaling through LEF1 and HOXB9 mediates lung adenocarcinoma metastasis. Cell 138, 51–62 (2009).
Tammela, T. et al. A Wnt-producing niche drives proliferative potential and progression in lung adenocarcinoma. Nature 545, 355–359 (2017).
Wagner, A. H. et al. Recurrent WNT pathway alterations are frequent in relapsed small cell lung cancer. Nat. Commun. 9, 3787 (2018).
Chilosi, M. et al. Aberrant Wnt/beta-catenin pathway activation in idiopathic pulmonary fibrosis. Am. J. Pathol. 162, 1495–1502 (2003).
Sucre, J. M. S. et al. A shared pattern of beta-catenin activation in bronchopulmonary dysplasia and idiopathic pulmonary fibrosis. Am. J. Pathol. 188, 853–862 (2018).
Lam, A. P. et al. Wnt coreceptor Lrp5 is a driver of idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med 190, 185–195 (2014).
Konigshoff, M. et al. WNT1-inducible signaling protein-1 mediates pulmonary fibrosis in mice and is upregulated in humans with idiopathic pulmonary fibrosis. J. Clin. Investig. 119, 772–787 (2009).
Akhmetshina, A. et al. Activation of canonical Wnt signalling is required for TGF-beta-mediated fibrosis. Nat. Commun. 3, 735 (2012).
Gaskill, C. F. et al. Disruption of lineage specification in adult pulmonary mesenchymal progenitor cells promotes microvascular dysfunction. J. Clin. Investig. 127, 2262–2276 (2017).
Konigshoff, M. et al. Functional Wnt signaling is increased in idiopathic pulmonary fibrosis. PLoS ONE 3, e2142 (2008).
Henderson, W. R. Jr. et al. Inhibition of Wnt/beta-catenin/CREB binding protein (CBP) signaling reverses pulmonary fibrosis. Proc. Natl Acad. Sci. USA 107, 14309–14314 (2010).
Sennello, J. A. et al. Lrp5/beta-Catenin Signaling Controls Lung Macrophage Differentiation and Inhibits Resolution of Fibrosis. Am. J. Respir. Cell Mol. Biol. 56, 191–201 (2017).
Martin-Medina, A. et al. Increased extracellular vesicles mediate WNT-5A signaling in idiopathic pulmonary fibrosis. Am. J. Respir. Crit Care Med. 198, 1527–1538 (2018).
Reyfman, P. A. et al. Single-cell transcriptomic analysis of human lung provides insights into the pathobiology of pulmonary fibrosis. Am. J. Respir. Crit Care Med. 199, 1517–1536 (2019).
Aumiller, V., Balsara, N., Wilhelm, J., Gunther, A. & Konigshoff, M. WNT/beta-catenin signaling induces IL-1beta expression by alveolar epithelial cells in pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 49, 96–104 (2013).
Tanjore, H. et al. beta-catenin in the alveolar epithelium protects from lung fibrosis after intratracheal bleomycin. Am. J. Respir. Crit. Care Med. 187, 630–639 (2013).
Sucre, J. M. S. et al. Hyperoxia injury in the develo** lung is mediated by mesenchymal expression of Wnt5A. Am. J. Respir. Crit Care Med. 201, 1249–1262 (2020).
Sucre, J. M. et al. Posttranslational modification of beta-catenin is associated with pathogenic fibroblastic changes in bronchopulmonary dysplasia. Am. J. Physiol. Lung Cell Mol. Physiol. 312, L186–L195 (2017).
Baarsma, H. A. et al. Noncanonical WNT-5A signaling impairs endogenous lung repair in COPD. J. Exp. Med. 214, 143–163 (2017).
Lehmann, M. et al. Chronic WNT/beta-catenin signaling induces cellular senescence in lung epithelial cells. Cell Signal. 70, 109588 (2020).
Author information
Authors and Affiliations
Contributions
C.J.A., C.J.P.: manuscript writing, figure design and execution. B.N.G.: manuscript writing, figure design and execution, financial support.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aros, C.J., Pantoja, C.J. & Gomperts, B.N. Wnt signaling in lung development, regeneration, and disease progression. Commun Biol 4, 601 (2021). https://doi.org/10.1038/s42003-021-02118-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-021-02118-w
- Springer Nature Limited
This article is cited by
-
Influence of intersignaling crosstalk on the intracellular localization of YAP/TAZ in lung cells
Cell Communication and Signaling (2024)
-
A WNT mimetic with broad spectrum FZD-specificity decreases fibrosis and improves function in a pulmonary damage model
Respiratory Research (2024)
-
Expression and potential molecular mechanism of TOP2A in metastasis of non-small cell lung cancer
Scientific Reports (2024)
-
Role of Sox9 in BPD and its effects on the Wnt/β-catenin pathway and AEC-II differentiation
Cell Death Discovery (2024)
-
HOXA9 versus HOXB9; particular focus on their controversial role in tumor pathogenesis
Journal of Applied Genetics (2024)