Abstract
Schisandrin B (SchB) is the highest content of biphenyl cyclooctene lignans in Schisandra chinensis. It has been reported to have a variety of pharmacological effects, including anti-inflammatory, anti-oxidant, anti-cancer, heart protection, liver protection. In this study, we found that SchB can promote the proliferation of MC3T3-E1 subclone 14 cells. Meanwhile, we found that SchB can regulate the BMP2-SMADs signaling pathway by increasing gene and protein expression of those relative biomolecules. Furthermore, SchB can raise the RUNX2 and SP7 expression in both mRNA and protein levels. Since the role of BMP2-SMADs-RUNX2-SP7 signaling axis in osteoblast proliferation and differentiation has been well documented. The present experimental findings indicate that SchB could promote the proliferation and differentiation of osteoblasts through BMP2-SMADs-RUNX2-SP7 signaling axis.
Similar content being viewed by others
Introduction
Schisandrin B (SchB) is one of the lignans with high content from Schisandra chinensis1. SchB has been reported to have a variety of pharmacological activities, including anti-inflammation2, antioxidation3, anti-cancer4,5 etc. It is worth noting that SchB has been reported to ameliorate chondrocytes inflammation and osteoarthritis by inhibiting NF-κB and MAPK signaling pathways6. These findings suggest that SchB may play a potential role in alleviate bone disease. However, the effect of SchB on osteoblasts is unknown.
MC3T3-E1 subclone 14 cells are often used to study the proliferation and differentiation of osteoblasts7,8,9. It is well known BMP2-SMADs signaling pathway play a key role in mediating osteoblast differentiation and osteogenesis10. While Runx2 and SP7 are transcription factors that are essential for osteoblast proliferation and differentiation11,12.
Here, the effects of SchB on the proliferation of MC3T3-E1 subclone 14 cells were evaluated by MTT assay, and the effects of SchB on the expression of genes and proteins related to BMP2-SMADs-RUNX2-SP7 signaling axis were investigated by quantitative PCR and western blot, respectively.
Results
SchB promotes MC3T3-E1 subclone 14 cells proliferation in a certain concentration range
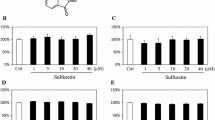
Osteoblasts are responsible for bone synthesis, remodeling and healing13. Osteoblast proliferation plays an important role in bone maintenance and development. In the present study, SchB stimulated the proliferation of MC3T3-E1 subclone 14 cells in gradually increase degrees at the concentration of 1.25–40 μM, while showed cytotoxicity at concentrations of 80 μM and 100 μM. In addition, after 24 h of treatment, the effect of SchB on the proliferation of MC3T3-E1 cells tended to be flat, and the intensity of action did not increase significantly with the extension of time (Fig. 1).
SchB promotes BMP2, SMADs and Runx2 gene expression
The positive correlation between BMP2, SMADs, RUNX2, and osteoblast proliferation and differentiation has been clearly elucidated11,14. At first, we found SchB could stimulate the proliferation of MC3T3-E1 subclone 14 cells by the MTT assay. Then attributing to the qPCR test we have confirmed that SchB could promote the expression of BMP2 gene in the concentration range of 5–40 μM, especially at the concentrations of 20 μM and 40 μM. In addition, similar findings have been found in the detection of SMAD1,4,5,9 and RUNX2 gene expression (Fig. 2).
SchB promotes BMP2-SMADs gene expression in MC3T3-E1 subclone 14 cells. (A) BMP2 mRNA expression; (B) qPCR product length of BMP2; (C) SMAD1 mRNA expression; (D) qPCR product length of SMAD1; (E) SMAD4 mRNA expression; (F) qPCR product length of SMAD4; (G) SMAD5 mRNA expression; (H) qPCR product length of SMAD5; (I)SMAD9 mRNA expression; (J) qPCR product length of SMAD9; (K) RUNX2 mRNA expression ; (L) qPCR product length of RUNX2.
SchB promotes BMP2, SMADs and Runx2 protein expression
Based on the findings at the mRNA level, we further examined the effects of SchB on these molecules at the protein level. Given that 2.5 μM SchB has no significant effect on BMP2-SMADs at the mRNA level, we chose 5–40 μM for protein level detection. The data showed that SchB could promote the protein expression of BMP2 in the concentration range of 5–40 μM. Meanwhile, SchB can significantly up-regulate the expression of SMAD4 protein only at the concentration of 20 and 40 μM, and up-regulate the expression of SMAD5,9 protein at the concentration of 40 μM. However, there was no significant change in the protein expression of SMAD1 (Fig. 3).
SchB promotes SP7 mRNA and protein expression
Sp7 is an essential transcription factor for osteoblast differentiation, which induced by Runx212. Bglap also known as osteocalcin, is an osteoblast marker. Sp7 directly regulates the expression of Bglap through Sp7-binding sites on the promoter region of the gene15. In the present study, we observed that SchB could upregulate the mRNA and protein expression of Sp7 while show no significant effect on Bglap (Fig. 4).
Discussion
In this study, we found that SchB can promote the proliferation of MC3T3-E1 subclone 14 cells and up-regulate the gene and protein expression of biomolecules in BMP2-SMADS signaling pathway. Firstly, SchB can promote the proliferation of MC3T3-E1 subclone 14 cells at the concentration of 1.25–40 μM. However, we note that extending the treating time of SchB does not enhance its effectiveness. This data indicated that SchB can achieve its strongest effect within 24 h. At the level of mRNA, SchB can promote the expression of BMP2, SMADs, Runx2, Sp7 genes a dose-dependent manner but have no impact on Bglap. The expression of BMP2, SMADs, Runx2, SP7 protein was up-regulated to varying degrees, but only SMAD4 and SP7 protein showed a dose-dependent relationship, while the expression of SMAD1 protein had no significant change. These results indicate that SchB may have a high selectivity for SMAD4 and SP7.
In addition, we noticed that 10–20 μM SchB markedly up-regulated the expression of Runx2, while 40 μM SchB had no significant effect on the protein expression of RUNX2 (Fig. 3K, L). Then we found 40 μM SchB also have no impact on mRNA expression of Sp7 (Fig. 4A). Given that SP7 induced by Runx2, this finding is reasonable. However, these series of results suggest that 40 μM SchB may be too high for MC3T3-E1 subclone 14 cells.
Taken together, our findings reveal the effect and mechanism of SchB on MC3T3-E1 subclone 14 cells (Fig. 5), and further confirm its potential value in the treatment of bone-related diseases, especially osteoarthritis and rheumatoid arthritis, which are closely related to inflammation and osteoblasts. Based on the findings of this study, we will further examine the potential role of SchB in the treatment of osteoarthritis and rheumatoid arthritis in vitro and in vivo.
Materials and methods
Reagents and antibodies
SchB was purchased from aladdin (Shanghai, China). MEM-α was purchased from Gibco (Bei**g, China). Fetal Bovine Serum was from Gemini bio-products (USA). Trypsin-EDTA solution and DMSO were from Solarbio (Bei**g, China). Penicillin–Streptomycin solution was from HyClone (USA). TrIzol Reagent, RevertAid First Strand cDNA Systhesis kit and PowerUp SYBR Green Master Mix were purchased from Thermo Fisher Scientific (USA). RIPA buffer and PMSF were purchased from Solarbio (Bei**g, China). BMP2 (Cat#18933-1), SMAD1 (Cat#10429-1-AP), SMAD4 (Cat#10231-1-AP), SMAD5 (Cat#12167-1-AP), SMAD9 (Cat#16397-1-AP) rabbit polyclonal antibody, HRP-conjugated Affinipure Goat Anti-Rabbit IgG(H + L) and HRP-conjugated Affinipure Goat Anti-Mouse IgG(H + L) Antibody were purchased from proteintech. RUNX2 (D1L7F) Rabbit mAb was purchased from cell signaling technology (Cat#12556). SP7 Polyclonal antibody was purchased from Invitrogen (Cat#PA5-40509). Chemiluminescent HRP substrate was purchased from Millipore.
Cell culture
MC3T3-E1 subclone 14 cell line obtained from the committee of type culture collection of Chinese Academy of sciences (Shanghai, China). It was maintained in MEM-α supplemented with 10% (v/v) fetal bovine serum (FBS), 100 units/mL penicillin, and 100 μg/μL streptomycin.
MTT assay
MTT assay was performed in 96-well plates in sextuplicate. MC3T3-E1 subclone 14 cells were seeded at a density of 5 × 103 cells/well overnight, and treated with compounds for 24 h, 48 h, 72 h respectively. OD490 values of compounds were detected using the Epoch 2 Microplate Spectrophotometer from BioTek Instruments.
Endogenous gene expression
MC3T3-E1 subclone 14 cells were seeded into 60 mm dishes and grown for 24 h in medium containing 10% FBS. Cells were then treated with DMSO or SchB with the indicated concentrations for 24 h. RNA was extracted and purified using the TrIzol Reagent. cDNA was prepared from 1 μg of RNA with the RevertAid First Strand cDNA Systhesis kit. Diluted cDNA was used to perform qPCR using SYBR Green (Light cycler 96, Roche) with ACTB as the internal standard. Primers for quantitative RT-PCR were listed in Table 1.
Western blot
MC3T3-E1 subclone 14 cells were seeded into 60 mm dishes and grown for 24 h in medium containing 10% FBS. Then cells received fresh medium containing the indicated treatments. Whole cell extracts were prepared after 24 h of treatment using RIPA buffer supplemented with 1 mM PMSF. 40 μg of protein per lane was analyzed on 10% SDS-PAGE gels and transferred to PVDF transfer membranes. BMP2, SMADs, Runx2, SP7 protein was detected using antibodies listed above. Images were captured using the Chemidoc CD Touch (Bio-Rad, USA), and images analyzing and processing using the Image Lab 6.0 (Bio-Rad, Chinese edition).
Statistical analysis
All results were presented as mean ± standard deviation (SD). Statistical significance was determined with One-Way ANOVA. p < 0.05 was considered statistically significant.
References
Dai, Z. et al. Exploration and optimization of conditions for quantitative analysis of lignans in Schisandra chinensis by an online supercritical fluid extraction with supercritical fluid chromatography system. J. Sep. Sci. 42, 2444–2454 (2019).
Ji, Z. R., Xue, W. L. & Zhang, L. Schisandrin B attenuates inflammation in LPS-induced sepsis through miR-17-5p downregulating TLR4. Inflammation 42, 731–739 (2019).
Chiu, P. Y., Lam, P. Y., Yan, C. W. & Ko, K. M. Schisandrin B protects against solar irradiation-induced oxidative injury in BJ human fibroblasts. Fitoterapia 82, 682–691 (2011).
**ang, S. S. et al. Schisandrin B induces apoptosis and cell cycle arrest of gallbladder cancer cells. Molecules (Basel, Switzerland) 19, 13235–13250 (2014).
Liu, Z., Zhang, B., Liu, K., Ding, Z. & Hu, X. Schisandrin B attenuates cancer invasion and metastasis via inhibiting epithelial-mesenchymal transition. PLoS ONE 7, e40480. https://doi.org/10.1371/journal.pone.0040480 (2012).
Ran, J. et al. Schisandrin B ameliorated chondrocytes inflammation and osteoarthritis via suppression of NF-kappaB and MAPK signal pathways. Drug Des. Dev. Ther. 12, 1195–1204 (2018).
Wu, Z. et al. Icaritin induces MC3T3-E1 subclone14 cell differentiation through estrogen receptor-mediated ERK1/2 and p38 signaling activation. Biomed. Pharmacother. 94, 1–9 (2017).
Shen, M., Wang, G., Wang, Y., **e, J. & Ding, X. Nell-1 enhances osteogenic differentiation of pre-osteoblasts on titanium surfaces via the MAPK-ERK signaling pathway. Cell. Physiol. Biochem. 50, 1522–1534 (2018).
Sun, K. et al. Lead exposure inhibits osteoblastic differentiation and inactivates the canonical Wnt signal and recovery by icaritin in MC3T3-E1 subclone 14cells. Chem. Biol. Interact. 303, 7–13 (2019).
Brunner, M. et al. β1 integrins mediate the BMP2 dependent transcriptional control of osteoblast differentiation and osteogenesis. PLoS ONE 13, e0196021 (2018).
Komori, T. Regulation of proliferation, differentiation and functions of osteoblasts by Runx2. Int. J. Mol. Sci. 20, 1694 (2019).
Komori, T. Runx2, an inducer of osteoblast and chondrocyte differentiation. Histochem. Cell Biol. 149, 313–323 (2018).
Titorencu, I., Pruna, V., **ga, V. V. & Simionescu, M. Osteoblast ontogeny and implications for bone pathology: an overview. Cell Tissue Res. 355, 23–33 (2014).
Karner, C. M., Lee, S.-Y. & Long, F. Bmp induces osteoblast differentiation through both Smad4 and mTORC1 signaling. Mol. Cell. Biol. 37, e00253-00216 (2017).
Chen, Z., Song, Z., Yang, J., Huang, J. & Jiang, H. Sp7/osterix positively regulates dlx2b and bglap to affect tooth development and bone mineralization in zebrafish larvae. J. Biosci. 44, 127 (2019).
Funding
This work was supported by the National Natural Science Foundation of China (81973976), the Initial Scientific Research Fund of Guangxi University of Chinese Medicine (2017BS040).
Author information
Authors and Affiliations
Contributions
X.W. and Y.P. initiated and designed the study; X.L., Y.Z., L.W. completed the experiment under the supervision of X.W.; X.W., X.L. and Y.Z. were involved in writing manuscript and drawing figures.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, X., Liao, X., Zhang, Y. et al. Schisandrin B regulates MC3T3-E1 subclone 14 cells proliferation and differentiation through BMP2-SMADs-RUNX2-SP7 signaling axis. Sci Rep 10, 14476 (2020). https://doi.org/10.1038/s41598-020-71564-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-71564-z
- Springer Nature Limited