Abstract
In patients with end-stage renal disease, excessive blood flow through an arteriovenous fistula (AVF) may lead to volume overload-induced cardiac remodeling and heart failure. It is unclear which patients with hyperfunctional AVF may benefit from AVF reduction or ligation. The indication for the procedure is often based on AVF flow. Because cardiac remodeling is driven by increased venous return, which is equivalent to cardiac output, we hypothesized that an elevated cardiac index (CI) might better identify subjects with reverse remodeling after AVF reduction. Thirty patients (age 52±12 years, 73% male) with AVF flow ⩾1.5 l min−1 underwent comprehensive echocardiographic evaluations before and after AVF reduction. At baseline, 16 patients had a normal CI (2.5–3.8 l min−1 m−2) and 14 had a high CI (4.0–6.0 l min−1 m−2). A left ventricular end-diastolic diameter decrease after operation was predicted by elevated baseline CI (P<0.01), but not elevated AVF flow (P=0.07). There was a significant decrease in CI, left ventricular mass, left atrial and right ventricular diameter and pulmonary systolic pressure in the high CI group but not in the normal CI group. After AVF reduction, systemic vascular resistance decreased in the normal CI group, whereas it did not change in the high CI group. In conclusion, reduction of high-flow AVF leads to reverse cardiac remodeling but only in patients with elevated CI. The variability of the response of systemic vascular resistance to AVF flow may explain this observation. Increased CI but not increased AVF flow may better determine candidates for AVF reduction.
Similar content being viewed by others
Introduction
A native arteriovenous fistula (AVF) is recommended for primary hemodialysis vascular access.1 AVF is simultaneously like both a ‘lifeline’ and an ‘Achilles heel,’ because of volume overload of the heart.2 AVF creation decreases systemic vascular resistance, thus leading to an increased cardiac index via increases in stroke volume, heart rate and left ventricular (LV) ejection fraction.3 Extremely elevated AVF flow may lead to marked cardiac index elevation, volume overload-induced cardiac remodeling and clinical heart failure.4, 5
To treat or prevent the development of heart failure, high-flow AVF are commonly closed or reduced. However, there are currently no clear indications to guide this practice. Increases in AVF flow are commonly considered when entertaining AVF modification. Basile et al.6 have observed that vascular access flow ⩾2 l min−1 is associated with increased risk of high-output heart failure, and the Vascular Access Society defines high-flow AVF as AVF flow >1–1.5 l min−1.6 Nonetheless, the association of AVF flow and cardiac index is nonlinear, and there is high inter-individual variability in CI at the same level of AVF flow.6, 7 Until now, no study has evaluated whether the effect of high-flow AVF reduction on heart remodeling is modified by cardiac index (CI). Because cardiac remodeling is driven by increased venous return, which is equivalent to cardiac output, we hypothesized that an elevated cardiac index might better identify subjects with reverse remodeling after AVF reduction than increased AVF flow.
The aim of this study was to identify predictors of cardiac reverse remodeling after AVF reduction and to evaluate the effects of the baseline cardiac index on structural and functional changes in the heart.
Methods
Population
All consecutive patients with high-flow (defined as AVF flow ⩾1.5 l min−1) type I aneurysmatic AVF according to the Balaz classification scheme8 referred to the Institute for Clinical and Experimental Medicine, Prague, Czech Republic for AVF reduction between January 2011 and October 2014 were included in this study. AVF flow and echocardiography were performed before the operation and at 3 months and 1 year after the operation. The study complied with the Declaration of Helsinki. Informed consent was obtained from all patients. The study was approved by the local ethics committee.
AVF reduction
A reinforced aneurysmorrhaphy with external polytetrafluoroethylene mesh tube was used to reduce the high-flow AVF. This technique was developed by our group in 20089 and has been validated10 and internationally adopted for aneurysmal AVF salvage.11 A detailed video of this procedure can be found online (http://aneurysmorrhaphy.eu—How to use section). In brief, the aneurysmal fistula was mobilized, and general heparinization (5000–10 000 IU) was performed. The supplying artery and non-dilated vein above the aneurysm were clamped, and the aneurysmal sac was resected to an appropriate diameter. The vein wall was sewn with a continuous running suture. The repaired vein was scaffolded with an external mesh polytetrafluoroethylene prosthesis (ProVena; BBraun Medical, Melsungen, Germany), and arteriovenous re-anastomosis was performed. The distal anastomosis of an upper-arm AVF was relocated to forearm arteries, whereas a reduction of the distal anastomosis was performed in the case of a forearm AVF. Heparin was neutralized by protamine sulfate, and the wound was closed with drainage. All patients used 100 mg of acetylsalicylic acid after the operation. The AVF after aneurysmorrhaphy was usually used for dialysis after 4 weeks.
AVF flow measurement
AVF flow was measured in the brachial artery using an Aplio500 ultrasound system (Toshiba, Tokyo, Japan) as previously described.2 AVF flow was calculated by multiplying the brachial artery cross-sectional area by the time-averaged mean velocity. The examinations were conducted by one angiology specialist with expertise in ultrasound examination.
Echocardiographic examination and hemodynamics
Echocardiographic examination was performed 24–48 h after dialysis to obtain similar and representative body fluid status. The examination was conducted by one medical doctor (VP) with expertise in echocardiography, and a Vivid7 ultrasound system (General Electric Healthcare, Wauwatosa, WI, USA) was used. The velocity–time integral in the LV outflow tract and the LV outflow tract diameter were used to calculate stroke volume and cardiac output. The cardiac index was calculated by dividing the cardiac output by the body surface area. A cardiac index ⩾3.9 l min −1m−2 was considered the cutoff for an elevated cardiac index.12 The LV mass was calculated by using the cube formula, as previously recommended.13 The right ventricular systolic pressure was estimated from the tricuspid regurgitation velocity (available in 58% of patients), and the right atrial pressure estimate was based on the inferior vena cava diameter and collapsibility. The LV ejection fraction was used as a parameter of LV function, and the tricuspid annular plane systolic excursion was used as a parameter of right ventricular systolic function. The mean blood pressure (MBP) was calculated by using the equation MBP=1/3 × SBP+2/3 × DBP, where SBP is the systolic blood pressure and DBP is the diastolic blood pressure. The total vascular resistance (TVR) was calculated as TVR=80 (MBP/CO). The systemic vascular resistance (SVR, vascular resistance omitting AVF) was calculated as SVR=80 (MBP/(CO-AVF flow)).
Statistical analysis
Continuous data with normal distributions are presented as the mean±s.d. and non-normally distributed variables are presented as the median (interquartile range—IQR). Categorical data are shown as frequencies and percentages. To account for the correlation of measures on the same patient, a random-effect mixed-linear model14 was used to assess the ability of baseline CI and AVF flow to predict LV diameter change after aneurysmorrhaphy. To increase the statistical power, and because we were interested in comparing the changes after operation between groups rather than determining the time point when these changes occurred, data examined at 3 months and 1 year after the operation were analyzed as a single time point (the after-operation time point). Patients were dichotomized on the basis of baseline CI (<3.9 l min−1m−2—normal CI group, ⩾3.9 l min−1 m−2—high CI group) and median AVF flow (<3.2 , ⩾3.2 l min−1). The null hypothesis tested was that the change in end-diastolic diameter after operation is independent of the baseline CI or AVF group. Differences between normal and high CI groups at baseline were compared using the t-test, Mann–Whitney U-test, χ2 or Fisher exact test, as appropriate. The random-effect mixed-linear model was also used to analyze sequential data by the baseline cardiac index group. Gamma regression was used for right-skewed data. Calculations were performed with SPSS version 21 (IBM SPSS Statistics; IBM Corporation, Armonk, New York, NY, USA) and JMP10 (SAS Institute, Cary, NC, USA). A two-sided P value <0.05 was considered to be statistically significant.
Results
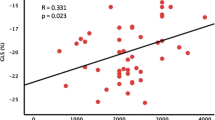
In total, 32 patients with high-flow AVF without clinical signs of heart failure underwent AVF reduction between January 2011 and October 2014 in our institution. Two patients were excluded from analyses, owing to valvular heart surgery during the follow-up (one aortic valve replacement, one mitral valve replacement). Data from thirty patients (mean age 52±12 years, 73% male) with high-flow AVF and a mean AVF flow of 3.3±1.1 l min−1 (range 1.5–5 l min−1) were analyzed. None of the patients included in the analyses had a significant valvular disease. As assessed by the corrected Akaike information criterion weight, linear regression (Figure 1) provided a better fit of the association between AVF flow and cardiac index (Akaike information criterion weight=0.65, R2=0.13) than did quadratic regression (Akaike information criterion weight=0.35, R2=0.17). At baseline, the LV end-diastolic diameter was associated with cardiac output (Pearson r=0.45, P=0.03) but not with AVF flow (Pearson r=0.13, P=0.51).
Association between arteriovenous fistula flow and cardiac index. Dark shaded area represents 95% CI of fit; light shaded area represents 95% CI of prediction. Data before and after operation are shown together. A full colour version of this figure is available online at the Hypertension Research website.
Predictors of reverse remodeling
The cardiac index discriminated patients with end-diastolic diameter (EDD) decrease after operation (interaction between CI group and time P<0.01), and increased CI was associated with EDD decrease after aneurysmorrhaphy (P=0.01) (Figure 2a). However, AVF flow was not able to discriminate patients with EDD decrease after operation (interaction between AVF flow and time P=0.94), and AVF flow over the median (3.2 l min−1) was not associated with an EDD decrease (P=0.07) (Figure 2b). When EDD was adjusted for body size (height or body surface area), the results did not change. Age (P=0.78), sex (P=0.49), baseline hemoglobin (P=0.11) and systolic blood pressure (P=0.57) did not predict EDD decrease after operation. On the basis of a univariate search for predictors of reverse remodeling, CI was the only predictor of LV diameter decrease after aneurysmorrhaphy. Thus, changes in other parameters of cardiac structure and function were analyzed by baseline CI category.
High CI and normal CI groups
There were 16 patients (54%) in the normal CI group (nCI) with CI<3.9 l min−1m−2 (range 2.5–3.8 l min−1m−2) and 14 patients in the high CI group (hCI) (46%) with an increased cardiac index (range 4.0–6.0 l min−1m−2). Whereas the mean AVF flow did not differ between groups (3.1±1.4 vs. 3.4±0.8 l min−1, P=0.34 for nCI vs. hCI groups), there were significant differences in cardiac index, systemic vascular resistance, hemoglobin level and LV diameter between groups (Table 1).
The effect of aneurysmorrhaphy
After aneurysmorrhaphy, the nCI and hCI groups had similar reductions in AVF flow (−1.22 (−1.81 to −0.64) vs. −1.55 (−2.18 to −0.92) l min−1, P=0.45) (Figure 3a). In contrast, the cardiac index decreased significantly only in hCI (−1.01 (−1.43 to −0.58) l min−1m−2, P<0.001) and not in the nCI group (−0.01 (−0.40 to 0.38) l min−1m−2, P=0.97) (Figure 3b). The total vascular resistance did not change in the nCI and increased in the hCI group (Figure 3c), whereas the systemic vascular resistance decreased in the nCI group and did not change in the hCI group (Figure 3d).
In the hCI group, there were significant decreases in LV end-diastolic diameter, mass, left atrial and right ventricular diameters, and estimated pulmonary systolic blood pressure (Table 2). In contrast, none of these parameters changed in the nCI group. There was a small decrease in the LV ejection fraction in both groups, but no patients developed new systolic dysfunction (EF<55%). There was a small decrease in the parameters of right ventricular systolic function (tricuspid annular plane systolic excursion, Sm) in the hCI group, but after correction for right ventricular diameter change, this difference was not significant.
Discussion
In the present study in patients with high-flow arteriovenous fistulas, we demonstrated that the effect of AVF reduction on heart remodeling is dependent on cardiac index before operation but does not depend on increased AVF flow. AVF flow reduction decreased LV end-diastolic diameter and mass, left atrial and right ventricular diameter and pulmonary pressure in the high CI group but not in the normal CI group. The variability of the response of systemic vascular resistance to AVF flow reduction may explain these observations.
Although several previous studies7, 15, 16 have assessed the effects of AVF closure on the heart, this study is the first to address the effect of AVF reduction on heart remodeling. The advantages of aneurysmorrhaphy compared with ligation are the preservation of vascular access and an excellent long-term patency with minimal periprocedural complications.10 In previously published studies, AVF closure has been shown to decrease left ventricular diameter and mass.7, 15, 16 However, results of the present study show that the effect of AVF reduction on heart remodeling is not present in all patients and is dependent on cardiac index before operation but does not depend on increased AVF flow. This conclusion is supported by a previous observation of patients after kidney transplant undergoing AVF closure,16 in which the magnitude of LV mass reduction was independent of AVF flow but correlated with baseline LV diameter and mass. In our study, left ventricular diameter at baseline was associated with cardiac output but not AVF flow, thus suggesting that LV dilatation is driven by increased cardiac output but not high-flow AVF.
In previous studies, the association between AVF flow and cardiac index/output has been found to be nonlinear.6, 7 In the present study, the association was linear but with high CI variability at the same level of AVF flow. This finding may be explained by a variable degree of systemic vasoconstriction/vasodilatation as a response to AVF creation. At baseline, despite similar AVF flow, patients in the hCI group had lower total vascular resistance and a higher cardiac index. Aneurysmorrhaphy in patients in the nCI group caused a decrease in SVR as a response to the AVF resistance increase, and thus, total vascular resistance and cardiac output did not change. In the hCI group, systemic vascular resistance did not change after aneurysmorrhaphy. Instead, the increase in total vascular resistance in these patients was coupled to decreasing cardiac output. The importance of the systemic vascular response found in our study is in line with results from a study by Unger et al.,17 in which the decrease in LV mass and diameter after AVF closure was predicted by the acute increase in total vascular resistance and MBP during pneumatic compression of AVF. In another study,18 the post-AVF closure 24 h diastolic blood pressure change (which is related to SVR) has been found to be negatively associated with LV mass change. This finding suggests that LV remodeling after AVF closure is present only if the increase in total vascular resistance by AVF closure/reduction is not offset by the decrease in SVR.
The inability to increase systemic vascular resistance as a response to high-flow AVF and to decrease systemic vascular resistance as a response to increased AVF resistance after AVF closure/reduction suggests a vasomotor dysfunction in patients in the hCI group. One of the mechanisms affecting vasomotor function is anemia, which decreases systemic vascular resistance19 and may cause high-output heart failure.20 In the present study, though hemoglobin levels increased in patients in the hCI group, the hemoglobin increase did not significantly increase systemic vascular resistance, and adjustment for hemoglobin did not attenuate the decrease in LV diameter in this group (Supplementary Table S1). Another mechanism that may explain abnormal vascular reactivity is autonomic dysfunction, which is common in patients with end-stage renal disease.21 However, autonomic dysfunction was not evaluated in this study. Furthermore, we did not find any significant differences in baseline antihypertensive medications, thus suggesting that vasodilatative therapy is not responsible for this phenomenon.
There is an ongoing debate regarding whether AVF is beneficial or harmful in patients with end-stage renal disease.22 In a retrospective study of dialyzed patients, no increased risk of death associated with high levels of AVF flow has been observed.23 Thus, in most patients, AVF reduction/closure is not required; but clearly, among patients who develop adverse remodeling or heart failure, intervention may be required. On the basis of previous case reports, it seems reasonable to intervene in subjects with high-output heart failure, in which symptoms usually disappear after operation.24 However, in more advanced stages of heart failure, patients may not benefit from the procedure.23, 25 The current results indicate that among patients without symptoms of heart failure, subjects with elevated cardiac index and left ventricular eccentric hypertrophy might be most likely to respond favorably to AVF reduction. In contrast, patients with high-flow AVF, normal cardiac index, no LV dilation and without symptoms of heart failure may not require surgical AVF reduction but instead would need close follow-up. Other causes of high cardiac index such as anemia, liver disease and hypervolemia should be excluded before intervention. Furthermore, other risk factors increasing morbidity and mortality in this population should be addressed.26, 27
Our study should be interpreted within the context of its strengths and limitations. Though the sample size of the present study is relatively low, it is the first study to assess the influence of AVF reduction on the heart. Non-invasive hemodynamic data inherently have greater variability than invasive measurements. Echocardiographic assessments were performed after a short interval, and chronic effects of AVF reduction were not assessed. Because we did not measure symptoms of heart failure and functional capacity during the study period, we are unable to compare the effect of AVF reduction on those parameters. We also did not measure cardiac biomarkers, which may provide deeper insight into cardiac changes.
In conclusion, this study shows that in patients with end-stage renal disease, high-flow arteriovenous fistula reduction causes reverse cardiac remodeling only in patients with elevated cardiac output and thus high venous return. This finding suggests that an increased cardiac index, but not increased arteriovenous flow, may be the optimal parameter to assess when considering aneurysmorrhaphy or other AVF reduction techniques in a patient with a high-flow arteriovenous fistula.
References
Vascular Access Work Group. Clinical practice guidelines for vascular access. Am J Kidney Dis 2006; 48 (Suppl 1): S248–S273.
Malik J, Kudlicka J, Tesar V, Linhart A . Cardiac safety in vascular access surgery and maintenance. Contrib Nephrol 2015; 184: 75–86.
Ori Y, Korzets A, Katz M, Perek Y, Zahavi I, Gafter U . Haemodialysis arteriovenous access—a prospective haemodynamic evaluation. Nephrol Dial Transplant 1996; 11: 94–97.
Singh S, Elramah M, Allana SS, Babcock M, Keevil JG, Johnson MR, Yevzlin AS, Chan MR . A case series of real-time hemodynamic assessment of high output heart failure as a complication of arteriovenous access in dialysis patients. Semin Dial 2014; 27: 633–638.
Raza F, Alkhouli M, Rogers F, Vaidya A, Forfia P . Case series of 5 patients with end-stage renal disease with reversible dyspnea, heart failure, and pulmonary hypertension related to arteriovenous dialysis access. Pulm Circ 2015; 5: 398–406.
Basile C, Lomonte C, Vernaglione L, Casucci F, Antonelli M, Losurdo N . The relationship between the flow of arteriovenous fistula and cardiac output in haemodialysis patients. Nephrol Dial Transplant 2008; 23: 282–287.
Aitken E, Kerr D, Geddes C, Berry C, Kingsmore D . Cardiovascular changes occurring with occlusion of a mature arteriovenous fistula. J Vasc Access 2015; 16: 459–466.
Balaz P, Bjorck M . True aneurysm in autologous hemodialysis fistulae: definitions, classification and indications for treatment. J Vasc Access 2015; 16: 446–453.
Balaz P, Rokosny S, Klein D, Adamec M . Aneurysmorrhaphy is an easy technique for arteriovenous fistula salvage. J Vasc Access 2008; 9: 81–84.
Rokosny S, Balaz P, Wohlfahrt P, Palous D, Janousek L . Reinforced aneurysmorrhaphy for true aneurysmal haemodialysis vascular access. Eur J Vasc Endovasc Surg 2014; 47: 444–450.
Berard X, Brizzi V, Mayeux S, Sassoust G, Biscay D, Ducasse E, Bordenave L, Corpataux JM, Midy D . Salvage treatment for venous aneurysm complicating vascular access arteriovenous fistula: Use of an exoprosthesis to reinforce the vein after aneurysmorrhaphy. Eur J Vasc Endovasc Surg 2010; 40: 100–106.
Nixon JV . American Heart Association The AHA Cinical Cardiac Consult. Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA. 2011.
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, Flachskampf FA, Foster E, Goldstein SA, Kuznetsova T, Lancellotti P, Muraru D, Picard MH, Rietzschel ER, Rudski L, Spencer KT, Tsang W, Voigt JU . Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 2015; 16: 233–270.
Cleophas TJ, Zwinderman AH, van Ouwerkerk BM . Methods for analysing cardiovascular studies with repeated measures. Neth Heart J 2009; 17: 429–433.
Unger P, Velez-Roa S, Wissing KM, Hoang AD, van de Borne P . Regression of left ventricular hypertrophy after arteriovenous fistula closure in renal transplant recipients: a long-term follow-up. Am J Transplant 2004; 4: 2038–2044.
van Duijnhoven EC, Cheriex EC, Tordoir JH, Kooman JP, van Hooff JP . Effect of closure of the arteriovenous fistula on left ventricular dimensions in renal transplant patients. Nephrol Dial Transplant 2001; 16: 368–372.
Unger P, Wissing KM, de Pauw L, Neubauer J, van de Borne P . Reduction of left ventricular diameter and mass after surgical arteriovenous fistula closure in renal transplant recipients. Transplantation 2002; 74: 73–79.
Unger P, Xhaet O, Wissing KM, Najem B, Dehon P, van de Borne P . Arteriovenous fistula closure after renal transplantation: a prospective study with 24-hour ambulatory blood pressure monitoring. Transplantation 2008; 85: 482–485.
Anand IS . Heart failure and anemia: mechanisms and pathophysiology. Heart Fail Rev 2008; 13: 379–386.
Mehta PA, Dubrey SW . High output heart failure. QJM 2009; 102: 235–241.
Salman IM . Cardiovascular autonomic dysfunction in chronic kidney disease: a comprehensive review. Curr Hypertens Rep 2015; 17: 571.
Basile C, Lomonte C . Pro: the arteriovenous fistula is a blessing of god. Nephrol Dial Transplant 2012; 27: 3752–3756.
Al-Ghonaim M, Manns BJ, Hirsch DJ, Gao Z, Tonelli M . Relation between access blood flow and mortality in chronic hemodialysis patients. Clin J Am Soc Nephrol 2008; 3: 387–391.
MacRae JM, Pandeya S, Humen DP, Krivitski N, Lindsay RM . Arteriovenous fistula-associated high-output cardiac failure: a review of mechanisms. Am J Kidney Dis 2004; 43: e17–e22.
Ingram CW, Satler LF, Rackley CE . Progressive heart failure secondary to a high output state. Chest 1987; 92: 1117–1118.
Cha RH, Kim S, Ae Yoon S, Ryu DR, Eun OhJ, Han SY, Young Lee E, Ki Kim D, Kim YS . Association between blood pressure and target organ damage in patients with chronic kidney disease and hypertension: results of the APrODiTe study. Hypertens Res 2014; 37: 172–178.
Kaneko H, Suzuki S, Uejima T, Kano H, Matsuno S, Takai H, Oikawa Y, Yajima J, Aizawa T, Yamashita T . Functional mitral regurgitation and left ventricular systolic dysfunction in the recent era of cardiovascular clinical practice, an observational cohort study. Hypertens Res 2014; 37: 1082–1087.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
BP and RS are the owners of BalRok clamp registration in the Office for Harmonization in the Internal Market (OHIM) Trade Marks and Designs and Community Design, RCD application number: 002024539. The other authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Hypertension Research website
Supplementary information
Rights and permissions
About this article
Cite this article
Wohlfahrt, P., Rokosny, S., Melenovsky, V. et al. Cardiac remodeling after reduction of high-flow arteriovenous fistulas in end-stage renal disease. Hypertens Res 39, 654–659 (2016). https://doi.org/10.1038/hr.2016.50
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2016.50
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
Have a Heart: Tie It Off — Update on Recommendations Regarding Vascular Access Ligation After Successful Renal Transplant
Current Transplantation Reports (2023)
-
NT-pro-BNP as marker for cardiac strain that may be caused by high-output arteriovenous shunting in a haemodialysis patient. A case report
BMC Nephrology (2020)
-
Cardiac impact of arteriovenous fistulas: what tools to assess?
Heart and Vessels (2020)
-
Relationships of high cardiac output with ventricular morphology, myocardial energetics, and energy costs in hemodialysis patients with preserved ejection fraction
The International Journal of Cardiovascular Imaging (2019)
-
Reply to ‘Cardiac remodeling after reduction of high-flow arteriovenous fistulas in end-stage renal disease: methodological issues’
Hypertension Research (2017)