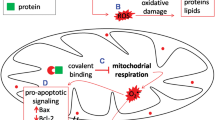
Abstract
Microsomal epoxide hydrolase/epoxide hydrolase 1 (mEH/EPHX1) works in conjunction with cytochromes P450 to metabolize a variety of compounds, including xenobiotics, pharmaceuticals and endogenous lipids. mEH has been most widely studied for its role in metabolism of xenobiotic and pharmaceutical compounds where it converts hydrophobic and reactive epoxides to hydrophilic diols that are more readily excreted. Inhibition or genetic disruption of mEH can be deleterious in the face of many industrial, environmental or pharmaceutical exposures and EPHX1 polymorphisms are associated with the development of exposure-related cancers. The role of mEH in endogenous epoxy-fatty acid (EpFA) metabolism has been less well studied. In vitro, mEH metabolizes most EpFAs at a far slower rate than soluble epoxide hydrolase (sEH) and has thus been generally considered to exert a minor role in EpFA metabolism in vivo. Indeed, sEH inhibitors or sEH-deficiency increase EpFA levels and are protective in animal models of cardiovascular disease. Recently, however, mEH was found to have a previously unrecognized and substantial role in EpFA metabolism in vivo. While few studies have examined the role of mEH in cardiovascular homeostasis, there is now substantial evidence that mEH can regulate cardiovascular function through regulation of EpFA metabolism. The discovery of a prominent role for mEH in epoxyeicosatrienoic acid (EET) metabolism, in particular, suggests that additional studies on the role of mEH in cardiovascular biology are warranted.


Similar content being viewed by others
References
Oesch F, Hengstler JG, Arand M (2004) Detoxication strategy of epoxide hydrolase-the basis for a novel threshold for definable genotoxic carcinogens. Nonlinearity Biol Toxicol Med 2:21–26. https://doi.org/10.1080/15401420490426963
El-Sherbeni AA, El-Kadi AO (2014) The role of epoxide hydrolases in health and disease. Arch Toxicol 88:2013–2032. https://doi.org/10.1007/s00204-014-1371-y
Vaclavikova R, Hughes DJ, Soucek P (2015) Microsomal epoxide hydrolase 1 (EPHX1): Gene, structure, function, and role in human disease. Gene 571:1–8. https://doi.org/10.1016/j.gene.2015.07.071
Arnold C, Markovic M, Blossey K, Wallukat G, Fischer R, Dechend R, Konkel A, von Schacky C, Luft FC, Muller DN, Rothe M, Schunck WH (2010) Arachidonic acid-metabolizing cytochrome P450 enzymes are targets of {omega}-3 fatty acids. J Biol Chem 285:32720–32733. https://doi.org/10.1074/jbc.M110.118406
Morisseau C, Hammock BD (2013) Impact of soluble epoxide hydrolase and epoxyeicosanoids on human health. Annu Rev Pharmacol Toxicol 53:37–58. https://doi.org/10.1146/annurev-pharmtox-011112-140244
Moghaddam MF, Grant DF, Cheek JM, Greene JF, Williamson KC, Hammock BD (1997) Bioactivation of leukotoxins to their toxic diols by epoxide hydrolase. Nat Med 3:562–566. https://doi.org/10.1038/nm0597-562
Edin ML, Gholipour Hamedani B, Gruzdev A, Graves JP, Lih FB, Arbes SJ, Singh R, Orjuela Leon AC, Bradbury JA, DeGraff LM, Hoopes SL, Arand M, Zeldin D (2018) Epoxide hydrolase 1 (EPHX1) hydrolyzes epoxyeicosanoids and impairs cardiac recovery after ischemia. J Biol Chem. https://doi.org/10.1074/jbc.ra117.000298
Marowsky A, Meyer I, Erismann-Ebner K, Pellegrini G, Mule N, Arand M (2017) Beyond detoxification: a role for mouse mEH in the hepatic metabolism of endogenous lipids. Arch Toxicol 91:3571–3585. https://doi.org/10.1007/s00204-017-2060-4
Decker M, Adamska M, Cronin A, Di Giallonardo F, Burgener J, Marowsky A, Falck JR, Morisseau C, Hammock BD, Gruzdev A, Zeldin DC, Arand M (2012) EH3 (ABHD9): the first member of a new epoxide hydrolase family with high activity for fatty acid epoxides. J Lipid Res 53:2038–2045. https://doi.org/10.1194/jlr.M024448
Marowsky A, Burgener J, Falck JR, Fritschy JM, Arand M (2009) Distribution of soluble and microsomal epoxide hydrolase in the mouse brain and its contribution to cerebral epoxyeicosatrienoic acid metabolism. Neuroscience 163:646–661. https://doi.org/10.1016/j.neuroscience.2009.06.033
Sinal CJ, Miyata M, Tohkin M, Nagata K, Bend JR, Gonzalez FJ (2000) Targeted disruption of soluble epoxide hydrolase reveals a role in blood pressure regulation. J Biol Chem 275:40504–40510. https://doi.org/10.1074/jbc.M008106200
Miyata M, Kudo G, Lee YH, Yang TJ, Gelboin HV, Fernandez-Salguero P, Kimura S, Gonzalez FJ (1999) Targeted disruption of the microsomal epoxide hydrolase gene. Microsomal epoxide hydrolase is required for the carcinogenic activity of 7,12-dimethylbenz[a]anthracene. J Biol Chem 274:23963–8. https://doi.org/10.1074/jbc.274.34.23963
Oesch F (1987) Significance of various enzymes in the control of reactive metabolites. Arch Toxicol 60:174–178. https://doi.org/10.1007/BF00296975
Oesch F, Daly J (1972) Conversion of naphthalene to trans-naphthalene dihydrodiol: evidence for the presence of a coupled aryl monooxygenase-epoxide hydrase system in hepatic microsomes. Biochem Biophys Res Commun 46:1713–1720. https://doi.org/10.1016/0006-291x(72)90807-8
Taura KI, Yamada H, Hagino Y, Ishii Y, Mori MA, Oguri K (2000) Interaction between cytochrome P450 and other drug-metabolizing enzymes: evidence for an association of CYP1A1 with microsomal epoxide hydrolase and UDP-glucuronosyltransferase. Biochem Biophys Res Commun 273:1048–1052. https://doi.org/10.1006/bbrc.2000.3076
Taura Ki K, Yamada H, Naito E, Ariyoshi N, Mori Ma MA, Oguri K (2002) Activation of microsomal epoxide hydrolase by interaction with cytochromes P450: kinetic analysis of the association and substrate-specific activation of epoxide hydrolase function. Arch Biochem Biophys 402:275–280. https://doi.org/10.1016/S0003-9861(02)00079-6
Orjuela Leon AC, Marwosky A, Arand M (2017) Evidence for a complex formation between CYP2J5 and mEH in living cells by FRET analysis of membrane protein interaction in the endoplasmic reticulum (FAMPIR). Arch Toxicol 91:3561–3570. https://doi.org/10.1007/s00204-017-2072-0
Lacourciere GM, Vakharia VN, Tan CP, Morris DI, Edwards GH, Moos M, Armstrong RN (1993) Interaction of hepatic microsomal epoxide hydrolase derived from a recombinant baculovirus expression system with an azarene oxide and an aziridine substrate analogue. Biochemistry 32:2610–2616. https://doi.org/10.1021/bi00061a019
Hartsfield JK Jr, Holmes LB, Morel JG (1995) Phenytoin embryopathy: effect of epoxide hydrolase inhibitor on phenytoin exposure in utero in C57BL/6J mice. Biochem Mol Med 56:131–143. https://doi.org/10.1006/bmme.1995.1068
Carlson GP (2011) Comparison of styrene oxide enantiomers for hepatotoxic and pneumotoxic effects in microsomal epoxide hydrolase-deficient mice. J Toxicol Environ Health A 74:347–350. https://doi.org/10.1080/15287394.2011.539130
Carlson GP (2010) Metabolism and toxicity of styrene in microsomal epoxide hydrolase-deficient mice. J Toxicol Environ Health A 73:1689–1699. https://doi.org/10.1080/15287394.2010.516240
Decker M, Arand M, Cronin A (2009) Mammalian epoxide hydrolases in xenobiotic metabolism and signalling. Arch Toxicol 83:297–318. https://doi.org/10.1007/s00204-009-0416-0
Tsuji PA, Walle T (2006) Inhibition of benzo[a]pyrene-activating enzymes and DNA binding in human bronchial epithelial BEAS-2B cells by methoxylated flavonoids. Carcinogenesis 27:1579–1585. https://doi.org/10.1093/carcin/bgi358
Morisseau C, Wecksler AT, Deng C, Dong H, Yang J, Lee KS, Kodani SD, Hammock BD (2014) Effect of soluble epoxide hydrolase polymorphism on substrate and inhibitor selectivity and dimer formation. J Lipid Res 55:1131–1138. https://doi.org/10.1194/jlr.M049718
Genomes Project C, Auton A, Brooks LD, Durbin RM, Garrison EP, Kang HM, Korbel JO, Marchini JL, McCarthy S, McVean GA, Abecasis GR (2015) A global reference for human genetic variation. Nature 526:68–74. https://doi.org/10.1038/nature15393
Hassett C, Aicher L, Sidhu JS, Omiecinski CJ (1994) Human microsomal epoxide hydrolase: genetic polymorphism and functional expression in vitro of amino acid variants. Hum Mol Genet 3:421–428. https://doi.org/10.1093/hmg/3.3.421
Beranek M, Fiala Z, Kremlacek J, Andrys C, Hamakova K, Chmelarova M, Palicka V, Borska L (2016) Genetic polymorphisms in biotransformation enzymes for benzo[a]pyrene and related levels of benzo[a]pyrene-7,8-diol-9,10-epoxide-DNA adducts in Goeckerman therapy. Toxicol Lett 255:47–51. https://doi.org/10.1016/j.toxlet.2016.05.009
Carbonari D, Mansi A, Proietto AR, Paci E, Bonanni RC, Gherardi M, Gatto MP, Sisto R, Tranfo G (2015) Influence of genetic polymorphisms of styrene-metabolizing enzymes on the levels of urinary biomarkers of styrene exposure. Toxicol Lett 233:156–162. https://doi.org/10.1016/j.toxlet.2015.01.002
Benhamou S, Reinikainen M, Bouchardy C, Dayer P, Hirvonen A (1998) Association between lung cancer and microsomal epoxide hydrolase genotypes. Cancer Res 58:5291–5293
Park JY, Chen L, Elahi A, Lazarus P, Tockman MS (2005) Genetic analysis of microsomal epoxide hydrolase gene and its association with lung cancer risk. Eur J Cancer Prev 14:223–230. https://doi.org/10.1097/00008469-200506000-00005
Erkisi Z, Yaylim-Eraltan I, Turna A, Gormus U, Camlica H, Isbir T (2010) Polymorphisms in the microsomal epoxide hydrolase gene: role in lung cancer susceptibility and prognosis. Tumori 96:756–763. https://doi.org/10.1177/030089161009600519
Fathy M, Hamed M, Youssif O, Fawzy N, Ashour W (2014) Association between environmental tobacco smoke exposure and lung cancer susceptibility: modification by antioxidant enzyme genetic polymorphisms. Mol Diagn Ther 18:55–62. https://doi.org/10.1007/s40291-013-0051-6
Zhang P, Zhang Y, Yang H, Li W, Chen X, Long F (2015) Association between EPHX1 rs1051740 and lung cancer susceptibility: a meta-analysis. Int J Clin Exp Med 8:17941–17949
Peluso ME, Munnia A, Srivatanakul P, Jedpiyawongse A, Sangrajrang S, Ceppi M, Godschalk RW, van Schooten FJ, Boffetta P (2013) DNA adducts and combinations of multiple lung cancer at-risk alleles in environmentally exposed and smoking subjects. Environ Mol Mutagen 54:375–383. https://doi.org/10.1002/em.21788
Xu X, Hua H, Fan B, Sun Q, Guo X, Zhang J (2015) EPHX1 rs2234922 polymorphism and lung cancer susceptibility in Asian populations: a meta-analysis. J Thorac Dis 7:1125–1129. https://doi.org/10.3978/j.issn.2072-1439.2015.07.03
Bauer AK, Faiola B, Abernethy DJ, Marchan R, Pluta LJ, Wong VA, Gonzalez FJ, Butterworth BE, Borghoff SJ, Everitt JI, Recio L (2003) Male mice deficient in microsomal epoxide hydrolase are not susceptible to benzene-induced toxicity. Toxicol Sci 72:201–209. https://doi.org/10.1093/toxsci/kfg024
Spector AA, Norris AW (2007) Action of epoxyeicosatrienoic acids on cellular function. Am J Physiol Cell Physiol 292:C996-C1012. https://doi.org/10.1152/ajpcell.00402.2006
Deng Y, Edin ML, Theken KN, Schuck RN, Flake GP, Kannon MA, DeGraff LM, Lih FB, Foley J, Bradbury JA, Graves JP, Tomer KB, Falck JR, Zeldin DC, Lee CR (2011) Endothelial CYP epoxygenase overexpression and soluble epoxide hydrolase disruption attenuate acute vascular inflammatory responses in mice. Faseb J 25:703–713. https://doi.org/10.1096/fj.10-171488
Seubert JM, Sinal CJ, Graves J, DeGraff LM, Bradbury JA, Lee CR, Goralski K, Carey MA, Luria A, Newman JW, Hammock BD, Falck JR, Roberts H, Rockman HA, Murphy E, Zeldin DC (2006) Role of soluble epoxide hydrolase in postischemic recovery of heart contractile function. Circ Res 99:442–450. https://doi.org/10.1161/01.RES.0000237390.92932.37
Larsen BT, Miura H, Hatoum OA, Campbell WB, Hammock BD, Zeldin DC, Falck JR, Gutterman DD (2006) Epoxyeicosatrienoic and dihydroxyeicosatrienoic acids dilate human coronary arterioles via BK(Ca) channels: implications for soluble epoxide hydrolase inhibition. Am J Physiol Heart Circ Physiol 290:H491–H499. https://doi.org/10.1152/ajpheart.00927.2005
Wang Y, Wei X, **ao X, Hui R, Card JW, Carey MA, Wang DW, Zeldin DC (2005) Arachidonic acid epoxygenase metabolites stimulate endothelial cell growth and angiogenesis via mitogen-activated protein kinase and phosphatidylinositol 3-kinase/Akt signaling pathways. J Pharmacol Exp Ther 314:522–532. https://doi.org/10.1124/jpet.105.083477
Panigrahy D, Edin ML, Lee CR, Huang S, Bielenberg DR, Butterfield CE, Barnes CM, Mammoto A, Mammoto T, Luria A, Benny O, Chaponis DM, Dudley AC, Greene ER, Vergilio JA, Pietramaggiori G, Scherer-Pietramaggiori SS, Short SM, Seth M, Lih FB, Tomer KB, Yang J, Schwendener RA, Hammock BD, Falck JR, Manthati VL, Ingber DE, Kaipainen A, D’Amore PA, Kieran MW, Zeldin DC (2012) Epoxyeicosanoids stimulate multiorgan metastasis and tumor dormancy escape in mice. J Clin Invest 122:178–191. https://doi.org/10.1172/JCI58128
Ellis EF, Amruthesh SC, Police RJ, Yancey LM (1991) Brain synthesis and cerebrovascular action of cytochrome P-450/monooxygenase metabolites of arachidonic acid. Adv Prostaglandin Thromboxane Leukot Res 21A:201–204
Arand M, Muller F, Mecky A, Hinz W, Urban P, Pompon D, Kellner R, Oesch F (1999) Catalytic triad of microsomal epoxide hydrolase: replacement of Glu404 with Asp leads to a strongly increased turnover rate. Biochem J 337:37–43. https://doi.org/10.1042/0264-6021:3370037
Marowsky A, Haenel K, Bockamp E, Heck R, Rutishauser S, Mule N, Kindler D, Rudin M, Arand M (2016) Genetic enhancement of microsomal epoxide hydrolase improves metabolic detoxification but impairs cerebral blood flow regulation. Arch Toxicol 90:3017–3027. https://doi.org/10.1007/s00204-016-1666-2
Chaudhary KR, Abukhashim M, Hwang SH, Hammock BD, Seubert JM (2010) Inhibition of soluble epoxide hydrolase by trans-4- [4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid is protective against ischemia-reperfusion injury. J Cardiovasc Pharmacol 55:67–73. https://doi.org/10.1097/FJC.0b013e3181c37d69
Seubert J, Yang B, Bradbury JA, Graves J, Degraff LM, Gabel S, Gooch R, Foley J, Newman J, Mao L, Rockman HA, Hammock BD, Murphy E, Zeldin DC (2004) Enhanced postischemic functional recovery in CYP2J2 transgenic hearts involves mitochondrial ATP-sensitive K+ channels and p42/p44 MAPK pathway. Circ Res 95:506–514. https://doi.org/10.1161/01.RES.0000139436.89654.c8
Katragadda D, Batchu SN, Cho WJ, Chaudhary KR, Falck JR, Seubert JM (2009) Epoxyeicosatrienoic acids limit damage to mitochondrial function following stress in cardiac cells. J Mol Cell Cardiol 46:867–875. https://doi.org/10.1016/j.yjmcc.2009.02.028
Seubert JM, Zeldin DC, Nithipatikom K, Gross GJ (2007) Role of epoxyeicosatrienoic acids in protecting the myocardium following ischemia/reperfusion injury. Prostaglandins Other Lipid Mediat 82:50–59. https://doi.org/10.1016/j.prostaglandins.2006.05.017
Groten T, Schleussner E, Lehmann T, Reister F, Holzer B, Danso KA, Zeillinger R (2014) eNOSI4 and EPHX1 polymorphisms affect maternal susceptibility to preeclampsia: analysis of five polymorphisms predisposing to cardiovascular disease in 279 Caucasian and 241 African women. Arch Gynecol Obstet 289:581–593. https://doi.org/10.1007/s00404-013-2991-9
Laasanen J, Romppanen EL, Hiltunen M, Helisalmi S, Mannermaa A, Punnonen K, Heinonen S (2002) Two exonic single nucleotide polymorphisms in the microsomal epoxide hydrolase gene are jointly associated with preeclampsia. Eur J Hum Genet 10:569–573. https://doi.org/10.1038/sj.ejhg.5200849
Pinarbasi E, Percin FE, Yilmaz M, Akgun E, Cetin M, Cetin A (2007) Association of microsomal epoxide hydrolase gene polymorphism and pre-eclampsia in Turkish women. J Obstet Gynaecol Res 33:32–37. https://doi.org/10.1111/j.1447-0756.2007.00473.x
Zusterzeel PL, Peters WH, Visser W, Hermsen KJ, Roelofs HM, Steegers EA (2001) A polymorphism in the gene for microsomal epoxide hydrolase is associated with pre-eclampsia. J Med Genet 38:234–237. https://doi.org/10.1136/jmg.38.4.234
Sari I, Okten H, Aktan C, Cihan E (2020) Association of the sEH gene promoter polymorphisms and haplotypes with preeclampsia. J Med Biochem 39:428–435. https://doi.org/10.5937/jomb0-27745
Sari I, Pinarbasi H, Pinarbasi E, Yildiz C (2017) Association between the soluble epoxide hydrolase gene and preeclampsia. Hypertens Pregnancy 36:315–325. https://doi.org/10.1080/10641955.2017.1388390
Catella F, Lawson J, Braden G, Fitzgerald DJ, Shipp E, FitzGerald GA (1991) Biosynthesis of P450 products of arachidonic acid in humans: increased formation in cardiovascular disease. Adv Prostaglandin Thromboxane Leukot Res 21A:193–196
Dalle Vedove F, Fava C, Jiang H, Zanconato G, Quilley J, Brunelli M, Guglielmi V, Vattemi G, Minuz P (2016) Increased epoxyeicosatrienoic acids and reduced soluble epoxide hydrolase expression in the preeclamptic placenta. J Hypertens 34:1364–1370. https://doi.org/10.1097/HJH.0000000000000942
Herse F, Lamarca B, Hubel CA, Kaartokallio T, Lokki AI, Ekholm E, Laivuori H, Gauster M, Huppertz B, Sugulle M, Ryan MJ, Novotny S, Brewer J, Park JK, Kacik M, Hoyer J, Verlohren S, Wallukat G, Rothe M, Luft FC, Muller DN, Schunck WH, Staff AC, Dechend R (2012) Cytochrome P450 subfamily 2J polypeptide 2 expression and circulating epoxyeicosatrienoic metabolites in preeclampsia. Circulation 126:2990–2999. https://doi.org/10.1161/CIRCULATIONAHA.112.127340
Enayetallah AE, French RA, Grant DF (2006) Distribution of soluble epoxide hydrolase, cytochrome P450 2C8, 2C9 and 2J2 in human malignant neoplasms. J Mol Histol 37:133–141. https://doi.org/10.1007/s10735-006-9050-9
Panigrahy D, Greene ER, Pozzi A, Wang DW, Zeldin DC (2011) EET signaling in cancer. Cancer Metastasis Rev 30:525–540. https://doi.org/10.1007/s10555-011-9315-y
Fritz P, Behrle E, Zanger UM, Murdter T, Schwarzmann P, Kroemer HK (1996) Immunohistochemical assessment of human microsomal epoxide hydrolase in primary and secondary liver neoplasm: a quantitative approach. Xenobiotica 26:107–116. https://doi.org/10.3109/00498259609046692
Murray GI, Weaver RJ, Paterson PJ, Ewen SW, Melvin WT, Burke MD (1993) Expression of xenobiotic metabolizing enzymes in breast cancer. J Pathol 169:347–353. https://doi.org/10.1002/path.1711690312
Lee HT, Lee KI, Chen CH, Lee TS (2019) Genetic deletion of soluble epoxide hydrolase delays the progression of Alzheimer’s disease. J Neuroinflammation 16:267. https://doi.org/10.1186/s12974-019-1635-9
Nelson JW, Young JM, Borkar RN, Woltjer RL, Quinn JF, Silbert LC, Grafe MR, Alkayed NJ (2014) Role of soluble epoxide hydrolase in age-related vascular cognitive decline. Prostaglandins Other Lipid Mediat 113–115:30–37. https://doi.org/10.1016/j.prostaglandins.2014.09.003
Liu M, Sun A, Shin EJ, Liu X, Kim SG, Runyons CR, Markesbery W, Kim HC, Bing G (2006) Expression of microsomal epoxide hydrolase is elevated in Alzheimer’s hippocampus and induced by exogenous beta-amyloid and trimethyl-tin. Eur J Neurosci 23:2027–2034. https://doi.org/10.1111/j.1460-9568.2006.04724.x
Morisseau C, Newman JW, Wheelock CE, Hill Iii T, Morin D, Buckpitt AR, Hammock BD (2008) Development of metabolically stable inhibitors of Mammalian microsomal epoxide hydrolase. Chem Res Toxicol 21:951–957. https://doi.org/10.1021/tx700446u
Acknowledgements
This work was supported by the Division of Intramural Research, National Institute of Environmental Health Sciences, NIH (Z01 ES025034 to D.C.Z.)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Rights and permissions
About this article
Cite this article
Edin, M.L., Zeldin, D.C. Regulation of cardiovascular biology by microsomal epoxide hydrolase. Toxicol Res. 37, 285–292 (2021). https://doi.org/10.1007/s43188-021-00088-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43188-021-00088-z