Abstract
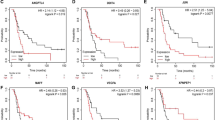
Angiogenesis is crucial to tumor growth and metastasis; it plays a key role in various cancers development and progression. However, the potential effects of angiogenesis-related genes (ARGs) in ovarian cancer (OC) remain to be further investigated. We discussed the characteristics changes of ARGs in 784 OC samples from genomic and transcriptional levels, as well as their expression patterns based on four distinct datasets. First, 784 OC patients were divided into three molecular subtypes, and the findings indicated that ARG changes were correlated with clinicopathological parameters, prognosis, and immune cell-infiltrating tumor microenvironment (TME). OC patients were subsequently divided into two gene subtypes depending on differentially expressed genes (DEGs) of the abovementioned molecular subtypes. We also established an ARGs-related score (ARGs score) model for evaluating overall survival (OS) and determining the immunological landscape of OC patients, therefore predicting patients’ prognosis and therapeutic responses. A lower ARGs’ score accompanied by a high mutation frequency implies a higher probability of survival. Furthermore, the ARG score was correlated with the cancer stem cell (CSC) index and chemotherapeutic sensitivity. The significant involvement of ARGs in the tumor-immune-stromal microenvironment, clinicopathological characteristics, and prognosis were established in our systematic investigation of ARGs for OC patients. These discoveries might help us to better understand the role of ARGs in OC, as well as give new insight for predicting the prognosis and providing promising immunotherapy.







Similar content being viewed by others
Data Availability
Publicly available datasets were analyzed in this study; these can be found in the Gene Expression Omnibus (GEO) (https://www.ncbi.nlm.nih.gov/geo/) and UCSC Xena (http://xena.ucsc.edu/). All procedure in this study followed corresponding guidelines and relative policies of above public database. The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Code Availability
The R markdown code used to conduct these analyses.
References
Shibuya M. Vascular endothelial growth factor-dependent and -independent regulation of angiogenesis. BMB Rep. 2008;41(4):278–86.
Park JA, Kwon Y-G. Hippo-YAP/TAZ signaling in angiogenesis. BMB Rep. 2018;51(3):157–62.
Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin Oncol. 2002;29(6 Suppl 16):15–8.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2022. CA: a Cancer Journal For Clinicians. 2022;72(1):7–33. https://doi.org/10.3322/caac.21708.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: Globocan Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: a Cancer Journal For Clinicians. 2021;71(3):209–49. https://doi.org/10.3322/caac.21660.
Chen Y, Zhang L, Liu W-X, Wang K. Vegf and Sema4d have synergistic effects on the promotion of angiogenesis in epithelial ovarian cancer. Cell Mol Biol Lett. 2018;23:2. https://doi.org/10.1186/s11658-017-0058-9.
Lim D, Do Y, Kwon BS, Chang W, Lee M-S, Kim J, et al. Angiogenesis and vasculogenic mimicry as therapeutic targets in ovarian cancer. BMB Rep. 2020;53(6):291–8.
Kurnit KC, Fleming GF, Lengyel E. Updates and new options in advanced epithelial ovarian cancer treatment. Obstet Gynecol. 2021;137(1):108–21. https://doi.org/10.1097/AOG.0000000000004173.
Schmitt J, Matei D. Targeting angiogenesis in ovarian cancer. Cancer Treat Rev. 2012;38(4):272–83. https://doi.org/10.1016/j.ctrv.2011.06.004.
Maman S, Witz IP. A history of exploring cancer in context. Nat Rev Cancer. 2018;18(6):359–76. https://doi.org/10.1038/s41568-018-0006-7.
Fukumura D, Kloepper J, Amoozgar Z, Duda DG, Jain RK. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nat Rev Clin Oncol. 2018;15(5):325–40. https://doi.org/10.1038/nrclinonc.2018.29.
Chen B, Gao A, Tu B, Wang Y, Yu X, Wang Y, et al. Metabolic modulation via mtor pathway and anti-angiogenesis remodels tumor microenvironment using Pd-L1-targeting codelivery. Biomaterials. 2020;255:120187. https://doi.org/10.1016/j.biomaterials.2020.120187.
Riera-Domingo C, Audigé A, Granja S, Cheng W-C, Ho P-C, Baltazar F, et al. Immunity, Hypoxia, and Metabolism-the Ménage à Trois of Cancer: Implications for Immunotherapy. Physiol Rev. 2020;100(1):1–102. https://doi.org/10.1152/physrev.00018.2019.
Song M, Yeku OO, Rafiq S, Purdon T, Dong X, Zhu L, et al. Tumor derived Ubr5 promotes ovarian cancer growth and metastasis through inducing immunosuppressive macrophages. Nat Commun. 2020;11(1):6298. https://doi.org/10.1038/s41467-020-20140-0.
Wang N, Wang S, Wang X, Zheng Y, Yang B, Zhang J, et al. Research trends in pharmacological modulation of tumor-associated macrophages. Clin Transl Med. 2021;11(1):e288. https://doi.org/10.1002/ctm2.288.
Koch A, Joosten SC, Feng Z, de Ruijter TC, Draht MX, Melotte V, et al. Analysis of DNA methylation in cancer: location revisited. Nat Rev Clin Oncol. 2018;15(7):459–66. https://doi.org/10.1038/s41571-018-0004-4.
Lambert SA, Jolma A, Campitelli LF, Das PK, Yin Y, Albu M, et al. The human transcription factors. Cell. 2018;172(4):650–65. https://doi.org/10.1016/j.cell.2018.01.029.
Sommerkamp P, Cabezas-Wallscheid N, Trumpp A. Alternative polyadenylation in stem cell self-renewal and differentiation. Trends Mol Med. 2021;27(7):660–72. https://doi.org/10.1016/j.molmed.2021.04.006.
Saygin C, Matei D, Majeti R, Reizes O, Lathia JD. Targeting cancer stemness in the clinic: from hype to hope. Cell Stem Cell. 2019;24(1):25–40. https://doi.org/10.1016/j.stem.2018.11.017.
Tyagi K, Mandal S, Roy A. Recent advancements in therapeutic targeting of the warburg effect in refractory ovarian cancer: a promise towards disease remission. Biochim Biophys Acta Rev Cancer. 2021;1876(1):188563. https://doi.org/10.1016/j.bbcan.2021.188563.
Steinbichler TB, Dudás J, Skvortsov S, Ganswindt U, Riechelmann H, Skvortsova I-I. Therapy resistance mediated by cancer stem cells. Semin Cancer Biol. 2018;53:156–67. https://doi.org/10.1016/j.semcancer.2018.11.006.
Roy L, Cowden Dahl KD. Can Stemness and Chemoresistance Be Therapeutically Targeted Via Signaling Pathways in Ovarian Cancer? Cancers (Basel). 2018;10(8):241. https://doi.org/10.3390/cancers10080241.
Keyvani V, Farshchian M, Esmaeili S-A, Yari H, Moghbeli M, Nezhad S-RK, et al. Ovarian cancer stem cells and targeted therapy. J Ovarian Res. 2019;12(1):120. https://doi.org/10.1186/s13048-019-0588-z.
Folkman J, Merler E, Abernathy C, Williams G. Isolation of a tumor factor responsible for angiogenesis. J Exp Med. 1971;133(2):275–88.
Carmeliet P, Jain RK. Angiogenesis in cancer and other diseases. Nature. 2000;407(6801):249–57.
Carmeliet P, Baes M. Metabolism and therapeutic angiogenesis. N Engl J Med. 2008;358(23):2511–2. https://doi.org/10.1056/NEJMcibr0802500.
Lorusso D, Ceni V, Daniele G, Salutari V, Pietragalla A, Muratore M, et al. Newly diagnosed ovarian cancer: which first-line treatment? Cancer Treat Rev. 2020;91:102111. https://doi.org/10.1016/j.ctrv.2020.102111.
Armstrong DK, Alvarez RD, Bakkum-Gamez JN, Barroilhet L, Behbakht K, Berchuck A, et al. Ovarian Cancer, Version 2.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2021;19(2):191–226. https://doi.org/10.6004/jnccn.2021.0007.
Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19(11):1423–37. https://doi.org/10.1038/nm.3394.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74. https://doi.org/10.1016/j.cell.2011.02.013.
Jiang Y, Wang C, Zhou S. Targeting tumor microenvironment in ovarian cancer: premise and promise. Biochim Biophys Acta Rev Cancer. 2020;1873(2):188361. https://doi.org/10.1016/j.bbcan.2020.188361.
Franklin RA, Li MO. Ontogeny of tumor-associated macrophages and its implication in cancer regulation. Trends Cancer. 2016;2(1):20–34.
Noy R, Pollard JW. Tumor-associated macrophages: from mechanisms to therapy. Immunity. 2014;41(1):49–61. https://doi.org/10.1016/j.immuni.2014.06.010.
Lawrence T, Natoli G. Transcriptional regulation of macrophage polarization: enabling diversity with identity. Nat Rev Immunol. 2011;11(11):750–61. https://doi.org/10.1038/nri3088.
Xue J, Schmidt SV, Sander J, Draffehn A, Krebs W, Quester I, et al. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity. 2014;40(2):274–88. https://doi.org/10.1016/j.immuni.2014.01.006.
Bagchi S, Yuan R, Engleman EG. Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Annu Rev Pathol. 2021;16:223–49. https://doi.org/10.1146/annurev-pathol-042020-042741.
Galluzzi L, Humeau J, Buqué A, Zitvogel L, Kroemer G. Immunostimulation with chemotherapy in the era of immune checkpoint inhibitors. Nat Rev Clin Oncol. 2020;17(12):725–41. https://doi.org/10.1038/s41571-020-0413-z.
Wan C, Keany MP, Dong H, Al-Alem LF, Pandya UM, Lazo S, et al. Enhanced efficacy of simultaneous Pd-1 and Pd-L1 immune checkpoint blockade in high-grade serous ovarian cancer. Cancer Res. 2021;81(1):158–73. https://doi.org/10.1158/0008-5472.CAN-20-1674.
Indini A, Nigro O, Lengyel CG, Ghidini M, Petrillo A, Lopez S, et al. Immune-Checkpoint Inhibitors in Platinum-Resistant Ovarian Cancer. Cancers (Basel). 2021;13(7):1663. https://doi.org/10.3390/cancers13071663.
Lee EK, Konstantinopoulos PA. Combined parp and immune checkpoint inhibition in ovarian cancer. Trends Cancer. 2019;5(9):524–8. https://doi.org/10.1016/j.trecan.2019.06.004.
Acknowledgements
We sincerely thank the R package developers and the maintainers of The Cancer Genome Atlas (TCGA) project and the Gene Expression Omnibus (GEO).
Funding
This work was supported by the National Natural Science Foundation of China (No. 81802605), Gynecological tumor special research Fund of Bei**g Kanghua Traditional Chinese and Western Medicine Development Foundation (No. KH-2021-LLZX-044), Tian** Key Medical Discipline (Specialty) Construction Project, and Natural Science Foundation of Tian** municipality (No. 21JCQNJC00230).
Author information
Authors and Affiliations
Contributions
Yurou Ji and Qi Ge contributed to the conception, data collection and analysis of the study. All authors drafted the manuscript. Wenwen Zhang and Pengpeng Qu revised the manuscript for important intellectual content. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
All data in this article comes from the public databases including the Gene Expression Omnibus (GEO) (https://www.ncbi.nlm.nih.gov/geo/) and UCSC Xena (http://xena.ucsc.edu/) and does not involve animal or human subjects in any way, so the IRB Approval is not available.
Consent to Participate
Not applicable.
Consent for Publication
All the authors read and approved the final manuscript.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ji, Y., Ge, Q., Zhang, W. et al. Molecular Features, Prognostic Value, and Cancer Immune Interactions of Angiogenesis-Related Genes in Ovarian Cancer. Reprod. Sci. 30, 1637–1650 (2023). https://doi.org/10.1007/s43032-022-01123-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-022-01123-6