Abstract
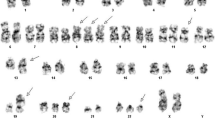
The 2017 revision of the World Health Organization classification of lymphoid neoplasms defined a new provisional entity, Burkitt-like lymphoma with 11q aberration (BLL-11q). BLL-11q lacks MYC rearrangement but has a chromosome 11q-gain/loss due to an inverted duplication event such as dup(11)(q23q13). Despite this classification, the association between 11q aberration and diffuse large B cell lymphoma (DLBCL) has not been fully characterized. Here, we describe an unusual case of DLBCL that presented as dup(11)(q11q24) without MYC rearrangement. A 68-year-old woman with left cervical lymphadenopathy was admitted to our hospital. Lymph node biopsy revealed diffuse proliferation of atypical large lymphoid cells positive for CD20, CD10, BCL6, MUM1, and BCL2, which led to a diagnosis of DLBCL. G-banding showed 46,X,-X,+7,dup(11)(q11q24),del(20)(q1?). Fluorescence in situ hybridization (FISH) detected duplicated CCND1 signals at 11q13.3 and duplicated KMT2A signals at 11q23.3 with similar intervals on dup(11)(q11q24). These results indicated that the chromosomal aberration was a simple duplication without inversion. FISH did not detect split MYC signals. Almost all cells were negative for MYC and CCND1. In contrast to BLL-11q, DLBCL seems to have a non-inverted simple duplication without terminal deletion. Thus, the distinction between an inverted duplication and a simple duplication by metaphase FISH is crucial, since the resultant genetic changes and disease phenotypes are different. Duplication of genes including KMT2A located on 11q may contribute to the pathogenesis of DLBCL.




Similar content being viewed by others
Data availability
Not applicable.
References
Leoncini L, Campo E, Stein H, et al. Burkitt-like lymphoma with 11q aberration. In: Swerdlow SH, Campo E, Harris NL, et al., editors. WHO classification of tumors of haematopoietic and lymphoid tissues. 4th ed. Lyon: IARC; 2017. p. 334.
Pienkowska-Grela B, Rymkiewicz G, Grygalewicz B, Woroniecka R, Krawczyk P, Czyz-Domanska K, et al. Partial trisomy 11, dup(11)(q23q13), as a defect characterizing lymphomas with Burkitt pathomorphology without MYC gene rearrangement. Med Oncol. 2011;28:1589–95.
Salaverria I, Martin-Guerrero I, Wagener R, Kreuz M, Kohler CW, Richter J, et al. A recurrent 11q aberration pattern characterizes a subset of MYC-negative high-grade B cell lymphomas resembling Burkitt lymphoma. Blood. 2014;123:1187–98.
Ferreiro JF, Morscio J, Dierickx D, Marcelis L, Verhoef G, Vandenberghe P, et al. Post-transplant molecularly defined Burkitt lymphomas are frequently MYC-negative and characterized by the 11q-gain/loss pattern. Haematologica. 2015;100:e275–9.
Havelange V, Ameye G, Théate I, Callet-Bauchu E, Lippert E, Luquet I, et al. The peculiar 11q-gain/loss aberration reported in a subset of MYC-negative high-grade B cell lymphomas can also occur in a MYC-rearranged lymphoma. Cancer Genet. 2016;209:117–8.
Grygalewicz B, Woroniecka R, Rymkiewicz G, Rygier J, Borkowska K, Kotyl A, et al. The 11q-gain/loss aberration occurs recurrently in MYC-negative Burkitt-like lymphoma with 11q aberration, as well as MYC-positive Burkitt lymphoma and MYC-positive high-grade B cell lymphoma, NOS. Am J Clin Pathol. 2018;149:17–28.
Gascoyne RD, Campo E, Jaffe ES, et al. Diffuse large B cell lymphoma, NOS. In: Swerdlow SH, Campo E, Harris NL, et al., editors. WHO classification of tumors of haematopoietic and lymphoid tissues. 4th ed. IARC: Lyon; 2017. p. 291–7.
Mitelman F, Johansson B, Mertens F, editors. Mitelman database of chromosome aberrations and gene fusions in cancer. 2020. http://mitelmandatabase.isb-cgc.org/. Accessed 31 October 2020.
Bloomfield CD, Arthur DC, Frizzera G, Levine EG, Peterson BA, Gajl-Peczalska KJ. Nonrandom chromosome abnormalities in lymphoma. Cancer Res. 1983;43:2975–84.
Richardson ME, Quanguang C, Filippa DA, et al. Intermediate- to high-grade histology of lymphomas carrying t(14;18) is associated with additional nonrandom chromosome changes. Blood. 1987;70:444–7.
Koduru PRK, Filippa DA, Richardson ME, Jhanwar SC, Chaganti SR, Koziner B, et al. Cytogenetic and histologic correlations in malignant lymphoma. Blood. 1987;69:97–102.
Cabanillas F, Pathak S, Trujillo J, Manning J, Katz R, McLaughlin P, et al. Frequent nonrandom chromosome abnormalities in 27 patients with untreated large cell lymphoma and immunoblastic lymphoma. Cancer Res. 1988;48:5557–64.
Fujii H, Yashige H, Misawa S, Tanaka S, Urata Y, Matuyama F. Ph chromosome in a patient with non-leukemic non-Hodgkin B cell lymphoma. Am J Hematol. 1990;35:213–5.
Mikraki V, Jhanwar SC, Filippa DA, Wollner N, Chaganti RSK. Distinct patterns of chromosome abnormalities characterize childhood non-Hodgkin’s lymphoma. Br J Haematol. 1992;80:15–20.
Jerkeman M, Johansson B, Åkerman M, et al. Prognostic implications of cytogenetic aberrations in diffuse large B cell lymphomas. Eur J Haematol. 1999;62:184–90.
Murase T, Nakamura S, Kawaguchi K, et al. An Asian variant of intravascular large B cell lymphoma: clinical, pathological and cytogenetic approaches to diffuse large B cell lymphoma associated with haemophagocytic syndrome. Br J Haematol. 2000;111:826–34.
Fan YS, Rizkalla K. Comprehensive cytogenetic analysis including multicolor spectral karyoty** and interphase fluorescence in situ hybridization in lymphoma diagnosis: a summary of 154 cases. Cancer Genet Cytogenet. 2003;143:73–9.
Katzenberger T, Lohr A, Schwarz S, Dreyling M, Schoof J, Nickenig C, et al. Genetic analysis of de novo CD5+ diffuse large B cell lymphomas suggests an origin from a somatically mutated CD5+ progenitor B cell. Blood. 2003;101:699–702.
Cook JR, Shekhter-Levin S, Swerdlow SH. Utility of routine classical cytogenetics studies in the evaluation of suspected lymphomas. Am J Clin Pathol. 2004;121:826–35.
Bosga-Bouwer AG, Kok K, Booman M, Boven L, van der Vlies P, van den Berg A, et al. Array comparative genomic hybridization reveals a very high frequency of deletions of the long arm of chromosome 6 in testicular lymphoma. Genes Chromosom Cancer. 2006;45:976–81.
Narayan G, **e D, Freddy AJ, Ishdorj G, Do C, Satwani P, et al. PCDH10 promoter hypermethylation is frequent in most histologic subtypes of mature lymphoid malignancies and occurs early in lymphomagenesis. Genes Chromosom Cancer. 2013;52:1030–41.
Bonetti P, Testoni M, Scandurra M, Ponzoni M, Piva R, Mensah AA, et al. Deregulation of ETS1 and FLI1 contributes to the pathogenesis of diffuse large B cell lymphoma. Blood. 2013;122:2233–41.
Ferdová E, Ferda J, Baxa J. 18F-FDG-PET/MRI in lymphoma patients. Eur J Radiol. 2017;94:A52–63.
McGowan-Jordan J, Hastings RJ, Moore S, editors. An international system for human cytogenetic nomenclature (2020). Basel: Karger; 2020.
Rymkiewicz G, Grygalewicz B, Chechlinska M, Blachnio K, Bystydzienski Z, Romejko-Jarosinska J, et al. A comprehensive flow-cytometry-based immunophenotypic characterization of Burkitt-like lymphoma with 11q aberration. Mod Pathol. 2018;31:732–43.
Wagener R, Seufert J, Raimondi F, Bens S, Kleinheinz K, Nagel I, et al. The mutational landscape of Burkitt-like lymphoma with 11q aberration is distinct from that of Burkitt lymphoma. Blood. 2019;133:962–6.
Deisch J, Fuda FB, Chen W, et al. Segmental tandem triplication of the MLL gene in an intravascular large B cell lymphoma with multisystem involvement. Arch Pathol Lab Med. 2009;133:1477–82.
Mater DV, Goodman BK, Wang E, Gaca AM, Wechsler DS. MLL duplication in a pediatric patient with B cell lymphoblastic lymphoma. J Pediatr Hematol Oncol. 2012;34:e120–3.
Acknowledgements
The authors thank Dr. S. Mitani (Hyogo Prefectural Amagasaki General Medical Center) for providing clinical information.
Code Availability
Not applicable.
Author information
Authors and Affiliations
Contributions
Ka. Y. provided the concept and design, collected and interpreted the data, and drafted the manuscript. Ke. K., A. H., S. S., Ki. Y., S. K., H. Ma, and H. Mi took care of the patient and collected the clinical data. Ka. K. and Y. H. performed pathological analyses. H. Mi revised and gave final approval of the manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All procedures performed in this study were in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Medicine
Rights and permissions
About this article
Cite this article
Yamamoto, K., Kurata, K., Higashime, A. et al. Non-inverted Duplication of 11q, dup(11)(q11q24), in a Diffuse Large B Cell Lymphoma Without MYC Rearrangement: Case Report. SN Compr. Clin. Med. 3, 1455–1462 (2021). https://doi.org/10.1007/s42399-021-00866-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42399-021-00866-8