Abstract
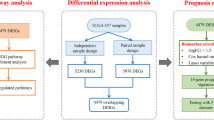
Lung cancer remains a leading cause of global cancer-related mortality, and the exploration of innovative therapeutic approaches, such as PD1/PDL1 immunotherapy, is critical. This study leverages comprehensive data from the Cancer Genome Atlas (TCGA) to investigate the differential expression of PD1/PDL1 in lung cancer patients and explores its implications. Clinical data, RNA expression, somatic mutations, and copy number variations of 1017 lung cancer patients were obtained from TCGA. Patients were categorized into high (HE) and low (LE) PD1/PDL1 expression groups based on mRNA levels. Analyses included differential gene expression, functional enrichment, protein-protein interaction networks, and mutational landscape exploration. The study identified 391 differentially expressed genes, with CD4 and PTPRC among the upregulated genes in the HE group. Although overall survival did not significantly differ between HE and LE groups, enrichment analysis revealed a strong association with immunoregulatory signaling pathways, emphasizing the relevance of PD1/PDL1 in immune response modulation. Notably, TP53 mutations were significantly correlated with high PD1/PDL1 expression. This study provides a comprehensive analysis of PD1/PDL1 expression in lung cancer, uncovering potential biomarkers and highlighting the intricate interplay between PD1/PDL1 and the immune response. The identified upregulated genes, including CD4 and PTPRC, warrant further investigation for their roles in the context of lung cancer and immunotherapy. The study underscores the importance of considering molecular heterogeneity in sha** personalized treatment strategies for lung cancer patients. Limitations, such as the retrospective nature of TCGA data, should be acknowledged.








Similar content being viewed by others
Data availability
All data are provided in this study and raw data can be requested to the corresponding author.
References
Bade BC, Dela Cruz CS (2020) Lung Cancer 2020: epidemiology, etiology, and prevention. Clin Chest Med 41(1):1–24
Bolandi N, Derakhshani A, Hemmat N, Baghbanzadeh A, Asadzadeh Z, Afrashteh Nour M, Brunetti O, Bernardini R, Silvestris N, Baradaran B (2021) The positive and negative immunoregulatory role of B7 family: promising novel targets in gastric cancer treatment. Int J Mol Sci 22(19):10719
Burova E, Hermann A, Waite J et al (2017) Characterization of the anti-PD-1 antibody REGN2810 and its antitumor activity in human PD-1 knock-in mice. Mol Cancer Ther 16(5):861–870
Cha JH, Chan LC, Li CW et al (2019) Mechanisms controlling PD-L1 expression in cancer. Mol Cell 76(3):359–370
Chen EJ, Chen S, Zhou FL (2021) Mechanism of TRIM27 promoting inflammatory response in lung cancer cells. Zhonghua Zhong Liu Za Zhi 43(10):1076–1081
Dong ZY, Zhong WZ, Zhang XC et al (2017) Potential predictive value of TP53 and KRAS mutation status for response to PD-1 blockade immunotherapy in lung adenocarcinoma. Clin Cancer Res 23(12):3012–3024
Ferlay J, Colombet M, Soerjomataram I et al (2019) Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer 144(8):1941–1953
Gómez-Henao W, Tenorio EP, Sanchez FRC et al (2021) Relevance of glycans in the interaction between T lymphocyte and the antigen presenting cell. Int Rev Immunol 40(4):274–288
Herbst RS, Morgensztern D, Boshoff C (2018) The biology and management of non-small cell lung cancer. Nature 553(7689):446–454
Hughes PE, Caenepeel S, Wu LC (2016) Targeted therapy and checkpoint immunotherapy combinations for the treatment of cancer. Trends Immunol 37(7):462–476
Lagou V, Garcia-Perez JE, Smets I et al (2018) Genetic architecture of adaptive immune system identifies key immune regulators. Cell Rep 25(3):798–810.e6
Leng C, Li Y, Qin J et al (2016) Relationship between expression of PD-L1 and PD-L2 on esophageal squamous cell carcinoma and the antitumor effects of CD8+ T cells. Oncol Rep 35(2):699–708
Liu C, Zheng S, ** R et al (2020) The superior efficacy of anti-PD-1/PD-L1 immunotherapy in KRAS-mutant non-small cell lung cancer that correlates with an inflammatory phenotype and increased immunogenicity. Cancer Lett 470:95–105
McDermott DF, Atkins MB (2013) PD-1 as a potential target in cancer therapy. Cancer Medicine 2(5):662–673
Mezzadra R, Sun C, Jae LT et al (2017) Identification of CMTM6 and CMTM4 as PD-L1 protein regulators. Nature 549(7670):106–110
Mogi A, Kuwano H (2011) TP53 mutations in nonsmall cell lung cancer. J Biomed Biotechnol 2011:583929
Raphael I, Joern RR, Forsthuber TG (2020) Memory CD4+ T cells in immunity and autoimmune diseases. Cells 9(3):531
Ritchie ME, Phipson B, Wu D et al (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43(7):e47
Roulleaux Dugage M, Nassif EF, Italiano A et al (2021) Improving immunotherapy efficacy in soft-tissue sarcomas: a biomarker driven and histotype tailored review. Front Immunol 12:775761
Salmaninejad A, Valilou SF, Shabgah AG et al (2019) PD-1/PD-L1 pathway: basic biology and role in cancer immunotherapy. J Cell Physiol 234(10):16824–16837
Singh AK, Stock P, Akbari O (2011) Role of PD-L1 and PD-L2 in allergic diseases and asthma. Allergy 66(2):155–162
Sun Y, Jiang L, Wen T et al (2021) Trends in the research into immune checkpoint blockade by anti-PD1/PDL1 antibodies in cancer immunotherapy: a bibliometric study. Front Pharmacol 12:670900
Toumazis I, Bastani M, Han SS et al (2020) Risk-based lung cancer screening: a systematic review. Lung Cancer 147:154–186
Tumeh PC, Harview CL, Yearley JH et al (2014) PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515(7528):568–571
Watza D, Lusk CM, Dyson G et al (2018) Prognostic modeling of the immune-centric transcriptome reveals interleukin signaling candidates contributing to differential patient outcomes. Carcinogenesis 39(12):1447–1454
Wei W, Zeng H, Zheng R et al (2020) Cancer registration in China and its role in cancer prevention and control. Lancet Oncol 21(7):e342–e3e9
Xu F, Lin H, He P et al (2020) A TP53-associated gene signature for prediction of prognosis and therapeutic responses in lung squamous cell carcinoma. Oncoimmunology 9(1):1731943
Yu G, Wang LG, Han Y et al (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. Omics 16(5):284–287
Zhou J, Zhang D, Zhang W (2023) Cross-view enhancement network for underwater images. Eng Appl Artif Intel 121:105952
Funding
This study did not receive any funding in any form.
Author information
Authors and Affiliations
Contributions
ZY, TH, & LW: concepts, design, data analysis, statistical analysis, manuscript preparation, manuscript review, guarantor; ZY, KH, HZ, LH, & LW: definition of intellectual content, literature search, experimental studies, data acquisition, manuscript editing
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by: Ewa Ziętkiewicz
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ye, Z., Huang, T., Hu, K. et al. Genomic Profiling Reveals Immune-Related Gene Differences in Lung Cancer Patients Stratified by PD1/PDL1 Expression: Implications for Immunotherapy Efficacy. J Appl Genetics (2024). https://doi.org/10.1007/s13353-024-00841-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13353-024-00841-8