Abstract
Lung function testing is an essential modality of investigation in children as it provides objective evidence of lung disease/health. With advances in technology, various tests are available that can aid in the diagnosis of lung disease, assess the progression and response to therapy and document the lung development and evolving lung diseases in infants. This narrative review discusses lung function tests in infants and children. Currently, lung function tests can be performed in every age group, from neonates to the elderly. Spirometry and peak expiratory flow rate (PEFR) are the most employed tests in children more than six years of age. Spirometry helps diagnose and monitoring of both obstructive and restrictive diseases. There is a need for expertise to perform and interpret spirometry correctly. The forced oscillation technique (FOT) or impulse oscillometry (IOS) is done with tidal volume breathing and is feasible even in preschool children. Their utility is mainly restricted to asthma in children at present. Lung function tests can be performed in neonates, infants and children using infant pulmonary function test (PFT) equipment, although their availability is limited. Diffusion capacity for carbon monoxide (DLCO) is a valuable tool in restrictive lung diseases. Lung volumes can be assessed by body plethysmography and multiple washout technique. The latter can also assess lung clearance index. It is essential to perform and interpret the lung function test results correctly and correlate them with the clinical condition for optimum treatment and outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lung function testing is becoming increasingly helpful in pediatric practice to diagnose and monitor various lung conditions. There is a wide array of available lung function tests in children, namely spirometry, forced oscillation technique (FOT)/ impulse oscillometry (IOS), infant pulmonary function test (PFT), plethysmography, diffusing capacity of the lungs for carbon monoxide (DLCO), multiple breath washout technique, exercise testing etc. Most of these tests measure the physiological lung volumes and provide objective evidence of the disease state and lung health. With the expanding knowledge of lung diseases in children and technological advancements, a sound understanding of these tests is essential. In this review, authors present an overview of lung function tests available in children and describe their principles, technique, interpretation, advantages, and limitations.
Spirometry
Spirometry is the most commonly used and available lung function test. Spirometry parameters are influenced by weight, height, age, sex, ethnicity, environmental factors, patient cooperation and effort, and technical factors [1]. The spirometer should fulfil International Organization for Standardization (ISO) 26782 standards with a maximum permissible accuracy error of ±2.5% as per the American Thoracic Society/European Respiratory Society (ATS/ERS) recommendations for spirometry [2].
Spirometry can be performed easily in children older than six years of age. A few studies have also been shown to produce technically satisfactory results in preschool children [3, 4]. As spirometry results depend highly on patient cooperation and effort, the technician/ doctor performing the test must be well-trained and patient-friendly. Children who cannot understand instructions or are uncooperative would not be able to complete the manoeuvre. Table 1 shows various indications and relative contraindications for spirometry in children [2].
Performing a Spirometry Test
After recording height, weight, sex and ethnicity, the child is seated on a chair or may stand. There are four steps of the forced vital capacity (FVC) manoeuvre, the most commonly used manoeuvre: 1. Rapid and maximal inspiration with lips tightly around the mouthpiece, 2. A “blast” of expiration, 3. continued blowing of air fast for 6 s in children (although it may be 3 s in younger children) and finally, 4. complete inspiration. The last inspiration step is optional and only required if we need inspiratory parameters. According to the 2019 ATS spirometry update, there is no requirement for a minimum forced expiratory time [2]. Computer-generated incentive graphics can help to encourage preschool children to perform spirometry properly. A nose clip is recommended while performing spirometry to avoid exhalation through the nose [2]. However, no differences in FEV1 or FVC measurements in children with and without nose clips were noted [5]. Bacterial filters are recommended and were not found to produce any clinically significant changes in spirometry measurements [6]. A maximum of eight complete manoeuvres can be attempted in one session [2]. FEV0.75 (the forced expiratory volume in the first 0.75 s) may be used for children aged six years or younger.
Interpretation of Spirometry Test
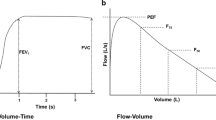
Before interpreting any spirogram, the test must meet the acceptability and repeatability criteria (Fig. 1) [2]. The acceptability criteria are (all must be fulfilled): (1) Must be free from artifacts: poor start, cough (Fig. 1a), double expiration (Fig. 1b), and poor patient efforts (Fig. 1c); (2) Must not have premature finish (glottis closure) before 1 s; (3) Back-extrapolated volume ≤5% of FVC or 100 ml, whichever is greater; (4) Expiration reached to a plateau (<0.025 L in the last 1 s of expiration); (5) Expiratory time at least for 3 s (6 s for older children); (6) There is no obstruction in mouthpiece (no flow on graph); (7) There should be no leak (interrupted line). Recently, no minimum forced expiratory time is defined as acceptability criteria. The repeatability criteria are (applied to acceptable FVC and FEV1 values): Age >6 y: the difference between the two largest FVC and FEV1 <150 ml; and for age <6 y: the difference between the two largest FVC and FEV1 <100 ml or 10% of the highest value, whichever is greater [3, 7].
The volume–time and flow–volume curves should be evaluated for this. The standard flow volume curve will have a sharp rise with a peak followed by a descent at an angle of about 45° (Fig. 1d). A small flow-volume curve suggests a restrictive pattern (Fig. 1e) or poor patient effort. A concavity (or scoo**) in the descending limb of the curve indicates airway obstruction (Fig. 1f). A flat FVC flow-volume curve without a peak suggests extrathoracic obstruction. Next, interpret the spirometry parameters shown in Fig. 2 [8]. The parameters must be compared with reference values. The recent ATS/ERS standard for interpreting routine lung function tests recommends using Global Lung function Initiative (GLI) reference equations for spirometry, DLCO and lung volumes [9, 10]. Spirometry results can be described as normal, obstructive, restrictive, or mixed pattern [11]. A value of mid-expiratory flow (FEF25-75%) below 60% of predicted also suggests an obstructive pattern and may be more sensitive to airway obstruction [12,13,14]. Till recently, FEV1, FVC, and FEV1/FVC values of more than 80% were taken as normal. Now, the normal range is defined between the 5th percentile [lower limit of normal (LLN)] and the 95th percentile [upper limit of normal (ULN)] [9]. The severity of obstruction and restriction was defined by % predicted. Currently, ATS/ERS statement defines the severity based on z-score: mild –1.65 to –2.5, moderate –2.51 to –4.0 and severe < –4.1 z-score spirometry and DLCO [9].
Bronchodilator Response (BDR)
BDR is assessed after 15 min of 4–5 doses of 100 μg salbutamol inhalation using a metered dose inhaler with a spacer or nebulization. A >12% improvement in FEV1 or 15–25% improvement in FEF25-75% suggests reversibility of airway obstruction (Fig. 1f) [2, 13]. The recent ATS/ERS standard defines the BDR as a >10% increase in FEV1 or FVC predicted [9]. Before conducting a bronchodilator test, it is necessary to ensure that the patient has not used a short-acting β2-agonist in the last 4–6 h or a short-acting muscarinic antagonist (ipratropium bromide) in the last 12 h or a long-acting β2- agonist in the last 24 h or a long-acting muscarinic antagonist in last 36–48 h [2]. Inhaled corticosteroids and leukotriene modifiers need not be withheld.
Forced Oscillation Technique (FOT)/ Impulse Oscillometry (IOS)
FOT/IOS is used to assess the mechanical properties of the respiratory system (airways, lung parenchyma and chest wall) in a passive manner during tidal breathing [7, 15].
Principle
The sound waves generated with the help of a loudspeaker are superimposed on normal tidal breathing. Three types of oscillation signals are used: 1. Monofrequency – Sound waves transmitted are of single frequency generating a sinusoidal waveform as in a FOT. 2. In IOS, recurrent pressure oscillations are applied at a fixed frequency of 5 Hz, generating a square wave pattern. 3. Pseudorandom noise (PRN) signal–Impulses of several frequencies are simultaneously applied [7, 16, 17]. According to the law of physics, the lower the airways' resistance, the higher the flow will be and vice versa. Therefore, the higher frequency impulses (15–20 Hz) get reflected from the large airways to the mouth, whereas the lower (5–10 Hz) frequencies travel deep into the lung before returning. The recommended frequency range is between 4–30 Hz [15]. Studies have demonstrated comparable measurements of airway resistance by devices using FOT and IOS, though FOT was more sensitive to airway elastase properties [18]. In clinical practice, any device, either based on FOT or IOS, may be used,
The main advantages of FOT/IOS are that it is effort independent, requires minimal patient cooperation, is quick and easy and can be performed in children from 2–3 y of age to the elderly [19,20,21]. It can also be used where spirometry is contraindicated, such as in recent surgery, recurrent pneumothorax, spirometry-related bronchospasm and during pandemics where forceful blowing is discouraged [7].
The commonly reported parameters in FOT/IOS are impedance [Z-sum of all the forces (resistance and reactance), resistance (R5, R20, R5-20), reactance (Xrs-out-of-phase component of lung impedance and reflects the elastic and inertial properties of the airway), Resonant frequency (Fres- frequency at which total reactance is null), and area of reactance [total reactance (area under the curve) at all frequencies between 5 Hz and Fres] [16, 17, 22]. R5 represents the total airway resistance, R20 represents the resistance of the large airways, and R5-R20 is the resistance of the small airways. In patients with small airway disease, resistance at low frequencies (R5) will be more apparent, and R5 will increase significantly compared to R20 [20, 22]. At lower frequencies, the tissue elastance dominates; therefore, Xrs is negative, whereas, at higher frequencies, the inertial properties dominate, denoted by a positive value of Xrs. The conditions that reduce the elasticity of the lung, such as fibrosis, interstitial disease and hyperinflation/ obstructive diseases and heterogeneity of the lung, make capacitance (Xrs) increasingly negative [17]. The normal Fres in adults is 8–12 Hz. In children, it can be as high as 30 Hz [15].
Coherence (Co) and Coefficient of variation (CoV) help interpret the validity of IOS results. The Co value is between 0 and 1. It should be between 0.9 and 1 at 20 Hz. CoV should be ≤10% in adults and ≤15% in children for two sets of Rrs [15]. The cut-off values have not been validated in younger children; hence, the recent ATS/ ERS guidelines have suggested using CoV% over coherence to determine quality control whenever available. Coherence is decreased by improper technique, swallowing, glottis closure, obstruction of airflow by the tongue, or irregular breathing.
Technique
After the child is seated with the head in the neutral position, the nose is clipped, and the cheeks and lower jaw are supported to prevent the shunting of oscillations into the upper airway. The patient takes tidal breathing through a tight mouthpiece in a closed system. The loudspeaker delivers multiple impulses over a period. The minimum acquisition time without artefacts is 16 s in children <12 y of age and 3 s in adults [15]. IOS should be performed before performing forced expiratory manoeuvres, such as spirometry, if both tests are done in the same session [15].
Interpretation
The accurate- time visualization of tidal flow is essential to identify artefacts like swallowing, irregular breathing, mouthpiece obstruction, glottis closure and mouth leaks. R5 and R20 values are considered normal if they are lower than 150% of predicted. X5 is considered normal if it is higher than X5 pred – 0.15 kps/l/. The recommended cut-offs for significant bronchodilator response in adults and children are –40% in R5, +50% in X5, and –80% in AX [15, 23]. Interpretation of IOS in certain common pathological conditions is shown in Table 2. The FEV1, FVC and PEF significantly co-relate with IOS parameters, as observed in a study by Dawman et al. [24]. IOS is more sensitive than spirometry in children for asthma diagnosis and treatment response [22, 23]. IOS has also been used to measure lung function in children with chronic lung disease of prematurity, cystic fibrosis, interstitial lung disease and neuromuscular disease [25, 26]. A global reference range for IOS is not available, and there is a need to develop multiethnic normative values. In the absence of these, reference values derived from a device most similar to the device being used are recommended [15].
Infant Pulmonary Function Tests
Infant PFTs are being performed at advanced centres specialized in pediatric respiratory care and are a way forward in early diagnosis, monitoring and intervention in evolving lung diseases in small children. The various tests that can be performed in this age group include- Tidal breathing flow-volume loop (TBFVL) manoeuvre, rapid thoracoabdominal compression (RTC), raised volume rapid thoracoabdominal compression (RVRTC) manoeuvres and lung volume measurement using body plethysmography.
TBFVL is performed in an awake infant, breathing quietly. The uncooperative babies may require some sedation, oral chloral hydrate being the preferred agent. A pneumotachograph is attached to a face mask (Fig. 3a), and the baby is allowed to breathe quietly for around 4–50 breath cycles. A minimum of 30 s of tidal breathing with 10 stable breaths should be recorded [27]. In an infant with normal lung function, oval/ round flow-volume curves are obtained (Fig. 3b). Various indices can be derived from these curves (Fig. 3c), viz. time to peak tidal expiratory flow (PTEF), total expiratory time (tE), the ratio of tPTEF/tE, volume at peak tidal expiratory flow (VPTEF), expired tidal volume (VE) and the ratio of VPTEF/VE. The most reported parameter in relationship to bronchial obstruction is the tPTEF/tE ratio and VPTEF/VE. Children with obstructive airway diseases or wheezing disorders reach peak tidal flow earlier during expiration and therefore have lower mean tPTEF/tE and VPTEF/VE values than control subjects [27, 28]. Most studies have reported a normal peak ratio between 0.3–0.4 [28, 29].
Infant PFT. a Child with ultrasonic flow measuring system attached to mask; b Real time graph on the screen during the test; c A format of report along with various parameters measured; d Performing RVRTC with a jacket wrapped around chest and abdomen. PFT Pulmonary function test, RVRTC Rapid thoracoabdominal compression
The RTC manoeuvres can be performed in spontaneously breathing infants by applying positive external pressure to the infant's chest wall and abdomen of the infant and thus squeezing the air out of the lungs (Fig. 3d). It can either be done in the tidal volume range (tidal volume RTC technique) or over an extended volume range by raising lung volume above tidal inspiration through an inflation procedure before the RTC manoeuvre (RVRTC technique). RVRTC appears similar to spirometry in older children. The same parameters are used to interpret the test as in TBFVL.
The various uses of infant PFT include evaluation of lung development during infancy and diagnosis and monitoring of different respiratory diseases starting during infancy. It has also been shown that acute respiratory infection during infancy is associated with impaired pulmonary function indices, such as increased resistance at three years of age [30]. Recently normative data for various infant PFT indices have been published in Indian babies [31].
Diffusion Capacity of Lungs for Carbon Monoxide (DLCO)/ Transfer Factor For CO (TLCO)
The ability of the lungs to transfer gas/ oxygen from inhaled air to the red blood cells in pulmonary capillaries is measured by DLCO. The commonly used gas is carbon monoxide as it has a high affinity for hemoglobin, its uptake across pulmonary capillary is diffusion limited, and it can be measured easily with infrared detectors or gas chromatography. The diffusion capacity of the lung is determined by the thickness of the alveolar-capillary membrane (alveolar epithelium, basement membrane, interstitial space and the capillary endothelium), hemoglobin concentration and cardiac output [32, 33]. The main indication of DLCO is for diagnosing restrictive lung diseases like interstitial lung diseases (ILD), hypersensitivity pneumonitis, sarcoidosis, and drug-induced ILD. It is also helpful to differentiate between the pulmonary and extrapulmonary causes of restriction, asthma and emphysema and diagnose pulmonary vascular disease and pulmonary hypertension. The presence of carbon monoxide toxicity and significant hypoxemia without supplemental oxygen are absolute contraindications.
It can be performed as a single breath manoeuvre, steady state manoeuvre or intra-breath analysis method using standardized techniques as per ATS/ERS guidelines [34]. Now GLI reference equations are available for DLCO [35]. In authors’ experience, steady state manoeuvre is relatively easy to perform in children. However, it is not widely used due to its variability, lack of studies and unavailability of normative data [34]. Boro et al. observed a significant correlation between DLCO is steady state (DLCOss) and DLCO is single breadth (DLCOsb) in adult patients with chronic obstructive lung disease [36].
Measurement of Lung Volumes (Body Plethysmography)
Body plethysmography evaluates various lung volumes, including functional residual capacity (FRC) and airway resistance [37]. In body plethysmography, the patient is enclosed in a closed chamber capable of measuring flow, volume and pressure. The washout techniques cannot measure non-communicating or poorly communicating air spaces, whereas plethysmography can measure lung volumes in all situations. The common indications of plethysmography include diagnosis of restrictive lung disease, differentiating between restrictive and obstructive lung disease by measuring lung volumes, diagnosis of bronchiolitis obliterans, assessing airway resistance with bronchodilation and broncho provocation tests, and to assess the progression of a disease or to see a response to therapy. The relative contraindications of body plethysmography include claustrophobia, altered sensorium, children receiving continuous oxygen supplementation, children with devices that may interfere with change in pressure like chest tube or ruptured tympanic membrane etc. The child may develop hypoxia, hypercapnia, and increased intrathoracic pressure by improper technique. The patient should be monitored continuously, and the box should be vented periodically. GLI reference equations are available for lung volumes [38].
Multiple-Breath Washout Test (Lung Clearance Index)
The multiple-breath washout (MBW) testing measures the efficiency of inert gas mixing in the lungs. The inert gas usually used is nitrogen. It is generally used in children with cystic fibrosis but is now increasingly used in obstructive diseases, bronchiectasis, and post-transplant bronchiolitis obliterans. The results are reported as lung clearance index (LCI) [39]. LCI indicates the number of lung turnovers needed to wash out the gas from the lungs. LCI is calculated as the total volume of air exhaled from the lungs during the washout period of the test, and it is corrected for the patient’s functional residual capacity (FRC), which is also measured during the test. LCI can detect early lung function impairment and pathology in peripheral airways. ATS published a technical document for MBW in preschool children in 2017 [40]. The MBW test is done during tidal breathing so that it can be done on preschool children (even infants) to the elderly. While interpreting results, we should ensure no leak during the measurement.
Exercise Testing
Exercise testing or cardiopulmonary exercise testing (CPET) is a useful, non-invasive investigation to evaluate physical fitness in children and adolescents with a potential role in the diagnosis, management and prognosis of several conditions in the pediatric population. By putting the respiratory system under stress, CPET may reveal subtle physiological abnormalities that may not be identified by routine static lung function tests [41]. CPET may be helpful in children with bronchopulmonary dysplasia, asthma, exercise-induced bronchospasm, and cystic fibrosis to evaluate the disease severity and to see the response of various therapeutic measures [42]. CPET can be performed as a free running field test or standardised protocols in an exercise testing laboratory using a treadmill or cycle ergometer. A few essential parameters measured in CPET include heart rate, oximetry, blood pressure, tidal volume, minute ventilation, rate of O2 consumption, maximum/peak oxygen uptake, rate of CO2 elimination, anerobic threshold etc. [41, 42].
Lung Function Tests During the COVID-19 Pandemic
Lung function tests, especially those with forceful manoeuvres, may be an aerosol-generating procedure. During the COVID-19 pandemic, lung function tests should be performed only when they are essential, and the latest recommendations should be followed [43].
Conclusions
In summary, PFT is an essential tool for pediatricians and pulmonologists. Apart from the limitations specific to each modality, the availability of standardized devices and reference values for children remains challenging. The results of any test should always be interpreted in conjunction with the clinical condition. The GLI reference equations are available for spirometry [9], DLCO [35], and lung volumes [38] and should be used while interpreting these. Follow-up tests help monitor the disease progression and response to therapy.
References
Kotecha SJ, Watkins WJ, Paranjothy S, Dunstan FD, Henderson AJ, Kotecha S. Effect of late preterm birth on longitudinal lung spirometry in school age children and adolescents. Thorax. 2012;67:54–61.
Graham BL, Steenbruggen I, Miller MR, et al. Standardization of spirometry 2019 update. An official american thoracic society and european respiratory society technical statement. Am J Respir Crit Care Med. 2019;200:e70–88.
Aurora P, Stocks J, Oliver C, et al. Quality control for spirometry in preschool children with and without lung disease. Am J Respir Crit Care Med. 2004;169:1152–9.
Crenesse D, Berlioz M, Bourrier T, Albertini M. Spirometry in children aged 3 to 5 years: reliability of forced expiratory maneuvers. Pediatr Pulmonol. 2001;32:56–61.
Chavasse R, Johnson P, Francis J, Balfour-Lynn I, Rosenthal M, Bush A. To clip or not to clip? Noseclips for spirometry. Eur Respir J. 2003;21:876–8.
Kamps AW, Vermeer K, Roorda RJ, Brand PL. Effect of bacterial filters on spirometry measurements. Arch Dis Child. 2001;85:346–7.
Komarow HD, Myles IA, Uzzaman A, Metcalfe DD. Impulse oscil- lometry in the evaluation of diseases of the airways in children. Ann Allergy Asthma Immunol. 2011;106:191–9.
Glady CA, Aaron SD, Lunau M, Clinch J, Dales RE. A spirometry-based algorithm to direct lung function testing in the pulmonary function laboratory. Chest. 2003;123:1939–46.
Stanojevic S, Kaminsky DA, Miller MR, et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur Respir J. 2022;60:2101499.
Quanjer PH, Stanojevic S, Cole TJ, et al. ERS global lung function initiative. Multi-ethnic reference values for spirometry for the 3-95-yr age range: the global lung function 2012 equations. Eur Respir J. 2012;40:1324–43.
Barreiro TJ, Perillo I. An approach to interpreting spirometry. Am Fam Physician. 2004;69:1107–14.
Lebecque P, Kiakulanda P, Coates AL. Spirometry in the asthmatic child: is FEF25-75 a more sensitive test than FEV1/FVC? Pediatr Pulmonol. 1993;16:19–22.
Jat KR. Spirometry in children. Prim Care Respir J. 2013;22:221–9.
Simon MR, Chinchilli VM, Phillips BR, et al. Childhood asthma research and education network of the national heart, lung, and blood institute. Forced expiratory flow between 25% and 75% of vital capacity and FEV1/forced vital capacity ratio in relation to clinical and physiological parameters in asthmatic children with normal FEV1 values. J Allergy Clin Immunol. 2010;126:527–34.
King GG, Bates J, Berger KI, et al. Technical standards for respiratory oscillometry. Eur Respir J. 2020;55:1900753.
Oostveen E, MacLeod D, Lorino H, et al. The forced oscillation technique in clinical practice: methodology, recommendations and future developments. Eur Respir J. 2003;22:1026–41.
Smith HJ, Reinhold P, Goldman MD. Forced oscillation technique and impulse oscillometry. Eur Respir Mon. 2005;31:72–105.
Soares M, Richardson M, Thorpe J, Owers-Bradley J, Siddiqui S. Comparison of forced and impulse oscillometry measurements: A clinical population and printed airway model study. Sci Rep. 2019;9:2130.
Frei J, Jutla J, Kramer G, Hatzakis GE, Ducharme FM, Davis GM. Impulse oscillometry: reference values in children 100 to 150 cm in height and 3 to 10 years of age. Chest. 2005;128:1266–73.
Bickel S, Popler J, Lesnick B, Eid N. Impulse oscillometry: Interpretation and practical applications. Chest. 2014;146:841–7.
Komarow HD, Skinner J, Young M, et al. A study of the use of impulse oscillometry in the evaluation of children with asthma: analysis of lung parameters, order effect, and utility compared with spirometry. Pediatr Pulmonol. 2012;47:18–26.
Shi Y, Aledia AS, Galant SP, George SC. Peripheral airway impairment measured by oscillometry predicts loss of asthma control in children. J Allergy Clin Immunol. 2013;131:718–23.
Desiraju K, Agrawal A. Impulse oscillometry: The state-of-art for lung function testing. Lung India. 2016;33:410–6.
Dawman L, Mukherjee A, Sethi T, Agrawal A, Kabra SK, Lodha R. Role of Impulse oscillometry in assessing asthma control in children. Indian Pediatr. 2020;57:119–23.
Raj D, Sharma GK, Lodha R, Kabra SK. Correlation between impulse oscillometry and spirometry parameters in Indian patients with cystic fibrosis. Chron Respir Dis. 2014;11:139–49.
Sugiyama A, Hattori N, Haruta Y, et al. Characteristics of inspiratory and expiratory reactance in interstitial lung disease. Respir Med. 2013;107:875–82.
Beydon N, Davis SD, Lombardi E, et al. American thoracic society/european respiratory society working group on infant and young children pulmonary function testing. An official american thoracic society/european respiratory society statement: pulmonary function testing in preschool children. Am J Respir Crit Care Med. 2007;175:1304–45.
van der Ent CK, Brackel HJ, van der Laag J, Bogaard JM. Tidal breathing analysis as a measure of airway obstruction in children three years of age and older. Am J Respir Crit Care Med. 1996;153:1253–8.
Mayer OH, Clayton RG, Jawad AF, McDonough JM, Allen JL. Respiratory inductance plethysmography in healthy 3- to 5-year-old children. Chest. 2003;124:1812–9.
Kumar P, Mukherjee A, Randev S, et al. Effect of acute respiratory infections in infancy on pulmonary function test at 3 years of age: a prospective birth cohort study. BMJ Open Respir Res. 2020;7:e000436.
Kumar P, Mukherjee A, Randev S, Jat KR, Lodha R, Kabra SK. Normative data of infant pulmonary function testing: A prospective birth cohort study from India. Indian Pediatr. 2020;57:25–33.
Forster R. Exchange of gases between alveolar air and pulmonary capillary blood: pulmonary diffusing capacity. Physiol Rev. 1957;37:391–452.
Hughes J, Pride N. In defense of the carbon monoxide transfer coefficient KCO (TL/VA). Eur Respir J. 2001;17:168–74.
Graham BL, Brusasco V, Burgos F, et al. ERS/ATS standards for single-breath carbon monoxide uptake in the lung. Eur Respir J. 2017;49:1600016.
Stanojevic S, Graham BL, Cooper BG, et al. Official ERS technical standards: global lung function initiative reference values for the carbon monoxide transfer factor for caucasians. Eur Respir J. 2017;50:1700010.
Boro AK, Sharma SK, Pande JN. Comparison of single breath and steady state methods for the measurement of pulmonary diffusing capacity for carbon monoxide in normal subjects, patients with bronchial asthma and chronic obstructive airway disease. Indian J Chest Dis Allied Sci. 1992;34:1–5.
Zysman-Colman Z, Lands LC. Whole body plethysmography: Practical considerations. Paediatr Respir Rev. 2016;19:39–41.
Hall GL, Filipow N, Ruppel G, et al. Official ERS technical standard: Global lung function initiative reference values for static lung volumes in individuals of European ancestry. Eur Respir J. 2021;57:2000289.
Stanojevic S, Bowerman C, Robinson P. Multiple breath washout: measuring early manifestations of lung pathology. Breathe. 2021;17:210016.
Robinson PD, Latzin P, Ramsey KA, et al. ATS assembly on pediatrics. preschool multiple-breath washout testing. an official american thoracic society technical statement. Am J Respir Crit Care Med. 2018;197:e1–19.
Wołoszyn F, Mazur A. Comprehensive use of cardiopulmonary exercise testing in pediatrics. Pediatr Endocrinol Diabetes Metab. 2021;27:42–6.
Teoh OH, Trachsel D, Mei-Zahav M, Selvadurai H. Exercise testing in children with lung diseases. Paediatr Respir Rev. 2009;10:99–104.
McGowan A, Laveneziana P, Bayat S, et al. International consensus on lung function testing during the COVID-19 pandemic and beyond. ERJ Open Res. 2022;8:00602–2021.
Author information
Authors and Affiliations
Contributions
KRJ: Conceptualized the manuscript, literature review, data synthesis, finalized manuscript; SA: Literature review, data synthesis, prepared initial manuscript. Both authors critically reviewed the manuscript and approved it for publication. KRJ will act as a guarantor for this manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jat, K.R., Agarwal, S. Lung Function Tests in Infants and Children. Indian J Pediatr 90, 790–797 (2023). https://doi.org/10.1007/s12098-023-04588-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-023-04588-8