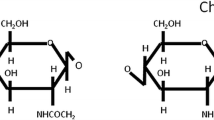
Abstract
Chitin from crustaceans is used in food, pharmaceutical, medical, agricultural and environmental sectors. Edible insects’ chitin is an untapped resource since edible insects have a high biodiversity and considerable amounts of chitin. Therefore, this study aimed at characterizing the functional properties and microstructure of chitin extracted from House cricket (Acheta domesticus), field cricket (Gryllus bimaculatus) and black soldier fly cocoons (Hermetica illucens) and compared with shrimp chitin (commercial). Chitin was chemically extracted and the functional groups were determined by Fourier Transform Infrared Spectroscopy (FTIR). Solubility, emulsion capacity, Water Holding Capacity, Fat Binding Capacity, degree of deacetylation and purity of the extracted chitin were also determined. Based on the FTIR spectra the extracted chitin showed the characteristic functional groups i.e. O–H stretch, C=O stretch, N–H bend, CH2 ending and CH3 deformation, C–N stretch and C–O–C stretch. Chitin extracted from Gryllus bimaculatus recorded the highest values in fat absorption capacity (780.14%), emulsion capacity (65.67%) and emulsion stability (65.67%). Chitin extracted from, Acheta domesticus was more soluble in water as compared to the commercial chitin. The highest level of deacetylation was reported in Hermetia illucens chitin (66.2%) while Acheta domesticus chitin had the least value (47.1%). The commercial chitin and Gryllus bimaculatus had the highest values for purity followed by Hermetia illucens. The microstructure images showed presence of pores and fibers in all the chitin samples. In conclusion, the insect chitin had characteristics similar to commercial chitin and are thus a suitable alternative in industrial application.


Similar content being viewed by others
References
E.B. Ibitoye, I.H. Lokman, M.N.M. Hezmee, Y.M. Goh, A.B.Z. Zuki, A.A. Jimoh, Extraction and physicochemical characterization of chitin and chitosan isolated from house cricket. Biomed. Mater. 13, 1–12 (2018). https://doi.org/10.1088/1748-605X/aa9dde
T. Philibert, B.H. Lee, N. Fabien, Current status and new perspectives on chitin and chitosan as functional biopolymers. Appl. Biochem. Biotechnol. 181, 1314–1337 (2017). https://doi.org/10.1007/s12010-016-2286-2
F. Shahidi, R. Abuzaytoun, Chitin, chitosan, and co-products: chemistry, production, applications, and health effects. Adv. Food Nutr. Res. 49, 93–137 (2005)
M. Zubair, M. Arshad, A. Ullah, in Handbook of Chitin and Chitosan (Elsevier Inc, 2020), pp. 773–809
K. Azuma, T. Osaki, S. Minami, Y. Okamoto, Anticancer and anti-inflammatory properties of chitin and chitosan Oligosaccharides. J. Funct. Biomater. 6, 33–49 (2015). https://doi.org/10.3390/jfb6010033
N. Nwe, S. Chandrkrachang, W.F. Stevens, T. Maw, T.K. Tan, E. Khor, S.M. Wong, Production of fungal chitosan by solid state and submerged fermentation. Carbohydr. Polym. 49, 3–5 (2002)
F. Tajdini, M.A. Amini, N. Nafissi-Varcheh, M.A. Faramarzi, Production, physiochemical and antimicrobial properties of fungal chitosan from Rhizomucor miehei and Mucor racemosus. Int. J. Biol. Macromol. 47, 180–183 (2010). https://doi.org/10.1016/j.ijbiomac.2010.05.002
S. Liu, J. Sun, L. Yu, C. Zhang, J. Bi, F. Zhu, M. Qu, C. Jiang, Q. Yang, Extraction and characterization of chitin from the beetle Holotrichia parallela motschulsky. Molecules 17, 4604–4611 (2012). https://doi.org/10.3390/molecules17044604
S. Crognale, C. Russo, M. Petruccioli, A. D’annibale, Chitosan production by fungi: current state of knowledge, future opportunities and constraints. Fermentation (2022). https://doi.org/10.3390/fermentation8020076
M. Triunfo, E. Tafi, A. Guarnieri, R. Salvia, C. Scieuzo, T. Hahn, S. Zibek, A. Gagliardini, L. Panariello, M.B. Coltelli, A. De Bonis, P. Falabella, Characterization of chitin and chitosan derived from Hermetia illucens, a further step in a circular economy process. Sci. Rep. 12, 1–17 (2022). https://doi.org/10.1038/s41598-022-10423-5
H. Greven, M. Kaya, I. Sargin, T. Baran, R. Møbjerg Kristensen, M. Vinther Sørensen, Characterisation of chitin in the cuticle of a velvet worm (Onychophora). Turk. J. Zool. 43, 416–424 (2019). https://doi.org/10.3906/zoo-1903-37
H. Wang, K. ur Rehman, W. Feng, D. Yang, R. ur Rehman, M. Cai, J. Zhang, Z. Yu, L. Zheng, Physicochemical structure of chitin in the develo** stages of black soldier fly. Int. J. Biol. Macromol. 149, 901–907 (2020). https://doi.org/10.1016/j.ijbiomac.2020.01.293
L. Soetemans, M. Uyttebroek, L. Bastiaens, Characteristics of chitin extracted from black soldier fly in different life stages. Int. J. Biol. Macromol. 165, 3206–3214 (2020). https://doi.org/10.1016/j.ijbiomac.2020.11.041
M.S. Benhabiles, R. Salah, H. Lounici, N. Drouiche, M.F.A. Goosen, N. Mameri, Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocoll. 29, 48–56 (2012). https://doi.org/10.1016/j.foodhyd.2012.02.013
M.D.A. Finke, Estimate of chitin in raw whole insects. Zoo Biol. 26, 105–115 (2007). https://doi.org/10.1002/zoo
A. Hirsch, Y.H. Cho, Y.H.B. Kim, O.G. Jones, Contributions of protein and milled chitin extracted from domestic cricket powder to emulsion stabilization. Curr. Res. Food Sci. 1, 17–23 (2019). https://doi.org/10.1016/j.crfs.2019.09.002
I. Aranaz, M. Mengibar, R. Harris, I. Panos, B. Miralles, N. Acosta, G. Galed, A. Heras, Functional characterization of chitin and chitosan. Curr. Chem. Biol. 3, 203–230 (2012). https://doi.org/10.2174/2212796810903020203
M.V. Tzoumaki, T. Moschakis, E. Scholten, C.G. Biliaderis, In vitro lipid digestion of chitin nanocrystal stabilized o/w emulsions. Food Funct. 4, 121–129 (2013). https://doi.org/10.1039/c2fo30129f
P. Charoenvuttitham, J. Shi, G.S. Mittal, Chitin extraction from black tiger shrimp (Penaeus monodon) waste using organic acids. Sep. Sci. Technol. 41, 1135–1153 (2006). https://doi.org/10.1080/01496390600633725
N. Van Toan, Production of chitin and chitosan from partially autolyzed shrimp shell materials. Open. Biomater. J. 1, 21–24 (2009)
T.S. Trung, W.W. Thein-Han, N.T. Qui, C.H. Ng, W.F. Stevens, Functional characteristics of shrimp chitosan and its membranes as affected by the degree of deacetylation. Bioresour. Technol. 97, 659–663 (2006). https://doi.org/10.1016/j.biortech.2005.03.023
Y.S. Puvvada, S. Vankayalapati, S. Sukhavasi, Extraction of chitin from chitosan from exoskeleton of shrimp for application in the pharmaceutical industry. Int. Curr. Pharm. J. 1, 258–263 (2012)
W. Sajomsang, P. Gonil, Preparation and characterization of α-chitin from cicada sloughs. Mater. Sci. Eng. C 30, 357–363 (2010). https://doi.org/10.1016/j.msec.2009.11.014
M. Naczk, L.J. Rubin, F. Shahidi, Functional properties and phytate content of pea protein preparations. J. Food Sci. 51, 1245–1247 (1986). https://doi.org/10.1111/j.1365-2621.1986.tb13096.x
Cereals and Grains Association, in AACC Approved Methods of Analysis, 11th edn. (AACC, 2000), pp. 3–4
M.J.Y.L. Humbert, ES and FWS, Certain functional properties sunflower meal products. J. Food Sci. 39, 5–7 (1974)
R. Hussain, M. Iman, T.K. Maji, Determination of degree of deacetylation of chitosan and their effect on the release behavior of essential oil from chitosan and chitosan-gelatin complex microcapsules. Int. J. Adv. Eng. Appl. 2, 4–12 (2013)
E.I. Díaz-Rojas, W.M. Argüelles-Monal, I. Higuera-Ciapara, J. Hernández, J. Lizardi-Mendoza, F.M. Goycoolea, Determination of chitin and protein contents during the isolation of chitin from shrimp waste. Macromol. Biosci. 6, 340–347 (2006). https://doi.org/10.1002/mabi.200500233
E.B. Ibitoye, I.H. Lokman, M.N.M. Hezmee, Y.M. Goh, A.B.Z. Zuki, A.A. Jimoh, Extraction and physicochemical characterization of chitin and chitosan isolated from house cricket. Biomed. Mater. (2018). https://doi.org/10.1088/1748-605X/aa9dde
M. Kaya, S. Erdogan, A. Mol, T. Baran, Comparison of chitin structures isolated from seven Orthoptera species. Int. J. Biol. Macromol. 72, 797–805 (2015). https://doi.org/10.1016/j.ijbiomac.2014.09.034
S. Kumari, P. Rath, A. Sri Hari Kumar, T.N. Tiwari, Extraction and characterization of chitin and chitosan from fishery waste by chemical method. Environ. Technol. Innov. 3, 77–85 (2015). https://doi.org/10.1016/j.eti.2015.01.002
A.T. Paulino, J.I. Simionato, J.C. Garcia, J. Nozaki, Characterization of chitosan and chitin produced from silkworm crysalides. Carbohydr. Polym. 64, 98–103 (2006). https://doi.org/10.1016/j.carbpol.2005.10.032
D. Purkayastha, S. Sarkar, Physicochemical structure analysis of chitin extracted from pupa exuviae and dead imago of wild Black soldier fly (Hermetia illucens). J. Polym. Environ. 28, 445–457 (2020). https://doi.org/10.1007/s10924-019-01620-x
A.M. Grumezescu, A. Maria, Therapeutic, Probiotic, and Unconventional Foods (Academic Press, Cambridge, 2018)
Y.I. Cho, H.K. No, S.P. Meyers, Physicochemical characteristics and functional properties of various commercial chitin and chitosan products. J. Agric. Food Chem. 46, 3839–3843 (1998). https://doi.org/10.1021/jf971047f
D. Knorr, Functional properties of chitin and chitosan. J. Food Sci. 47, 593–595 (1982). https://doi.org/10.1111/j.1365-2621.1982.tb10131.x
L. Sampath, S. Ngasotter, P. Layana, A.K. Balenge, B.B. Nayak, K.A.M. Xavier, Effect of chemical treatment duration on physicochemical, rheological, and functional properties of colloidal chitin. Food Hydrocoll. Health 2, 100091 (2022)
N.H. Marei, E.A. El-Samie, T. Salah, G.R. Saad, A.H.M. Elwahy, Isolation and characterization of chitosan from different local insects in Egypt. Int. J. Biol. Macromol. 82, 871–877 (2016). https://doi.org/10.1016/j.ijbiomac.2015.10.024
F. Nessa, S. Masum, M. Asaduzzaman, S.K. Roy, A process for the preparation of chitin and chitosan from prawn shell waste. Bangladesh J. Sci. Ind. Res. 45, 323–330 (2010).
N. Panith, J. Wichaphon, S. Lertsiri, N. Niamsiri, Effect of physical and physicochemical characteristics of chitosan on fat-binding capacities under in vitro gastrointestinal conditions. LWT 71, 25–32 (2016). https://doi.org/10.1016/j.lwt.2016.03.013
J. Liu, J. Zhang, W. **a, Hypocholesterolaemic effects of different chitosan samples in vitro and in vivo. Food Chem. 107, 419–425 (2008). https://doi.org/10.1016/j.foodchem.2007.08.044
Y. Huang, Y. Tsai, Extraction of chitosan from squid pen waste by high hydrostatic pressure: effects on physicochemical properties and antioxidant activities of chitosan. Int. J. Biol. Macromol. 160, 677–687 (2020). https://doi.org/10.1016/j.ijbiomac.2020.05.252
G. Ru, S. Wu, X. Yan, B. Liu, P. Gong, L. Wang, J. Feng, Inverse solubility of chitin/chitosan in aqueous alkali solvents at low temperature. Carbohydr. Polym. 206, 487–492 (2019). https://doi.org/10.1016/j.carbpol.2018.11.016
M. Mahdy Samar, M.H. El-Kalyoubi, M.M. Khalaf, M.M. Abd El-Razik, Physicochemical, functional, antioxidant and antibacterial properties of chitosan extracted from shrimp wastes by microwave technique. Ann. Agric. Sci. 58, 33–41 (2013). https://doi.org/10.1016/j.aoas.2013.01.006
M.S. Hossain, A. Iqbal, Production and characterization of chitosan from shrimp waste. J. Bangladesh Agric. Univ. 12, 153–160 (2014)
K. Kurita, Chitin and chitosan: functional biopolymers from marine crustaceans. Mar. Biotechnol. 8, 203–226 (2006). https://doi.org/10.1007/s10126-005-0097-5
M. Rinaudo, Chitin and chitosan: properties and applications. Prog. Polym. Sci. 31, 603–632 (2006). https://doi.org/10.1016/j.progpolymsci.2006.06.001
C. Gartner, C.A. Peláez, B.L. López, Characterization of chitin and chitosan extracted from shrimp shells by two methods. E-Polymers 10, 1–16 (2010). https://doi.org/10.1515/epoly.2010.10.1.748
A.J. Hirsch, Functional properties of protein and chitin from commercial cricket flour (2018)
B. Mohanty, D.M. Mulhivill, P.F. Fox, Emulsifying and foaming properties of acidic caseins and sodium caseinate. Food Chem. 28, 17–30 (1988)
S.U. Pickering, Pickering: emulsions. J. Chem. Soc. Trans. 91, 2001–2021 (1907)
F. Liu, C.H. Tang, Soy protein nanoparticle aggregates as pickering stabilizers for oil-in-water emulsions. J. Agric. Food Chem. 61, 8888–8898 (2013)
C. Harkin, N. Mehlmer, D.V. Woortman, T.B. Brück, W.M. Brück, Nutritional and additive uses of chitin and chitosan in the food industry (2019)
H. Zhang, S. Yun, L. Song, Y. Zhang, Y. Zhao, The preparation and characterization of chitin and chitosan under large-scale submerged fermentation level using shrimp by-products as substrate. Int. J. Biol. Macromol. 96, 334–339 (2017). https://doi.org/10.1016/j.ijbiomac.2016.12.017
J. Xu, L. Liu, J. Yu, Y. Zou, Z. Wang, Y. Fan, DDA (degree of deacetylation) and pH-dependent antibacterial properties of chitin nanofibers against Escherichia coli. Cellulose 26, 2279–2290 (2019). https://doi.org/10.1007/s10570-019-02287-2
Y. Wu, T. Sasaki, S. Irie, K. Sakurai, A novel biomass-ionic liquid platform for the utilization of native chitin. Polymer (Guildf) 49, 2321–2327 (2008). https://doi.org/10.1016/j.polymer.2008.03.027
F.B. Silva, L.J. Gasparrini, P.A. Cremonez, G.R.M. Burin, B. Machado, M.A. Polinarski, M.K. Arantes, H.J. Alves, Chitosan preparations with improved fat-binding capacity. J. Appl. Polym. Sci. 138, 1–15 (2021). https://doi.org/10.1002/app.50841
J. Zhang, W.-R. Xu, Y.-C. Zhang, Facile production of chitin from shrimp shells using a deep eutectic solvent and acetic acid. R Soc. Chem. 12, 22631–22638 (2022). https://doi.org/10.1039/d2ra03417d
X. Zhu, J. Cai, J. Yang, Q. Su, Determination of glucosamine in impure chitin samples by high-performance liquid chromatography. Carbohydr. Res. 340, 1732–1738 (2005). https://doi.org/10.1016/j.carres.2005.01.045
T. Hahn, A. Roth, R. Ji, E. Schmitt, S. Zibek, Chitosan production with larval exoskeletons derived from the insect protein production. J. Biotechnol. 310, 62–67 (2020). https://doi.org/10.1016/j.jbiotec.2019.12.015
A. Khayrova, S. Lopatin, V. Varlamov, Obtaining chitin/chitosan-melanin complexes from black soldier fly Hermetia illucens. IOP Conf. Ser. Mater. Sci. Eng. (2020). https://doi.org/10.1088/1757-899X/809/1/012020
M. Psarianos, S. Ojha, R. Schneider, O.K. Schlüter, Chitin isolation and chitosan production from house crickets (Acheta domesticus) by environmentally friendly methods. Molecules (2022). https://doi.org/10.3390/molecules27155005
M. Kaya, T. Baran, A. Mentes, M. Asaroglu, G. Sezen, K.O. Tozak, Extraction and characterization of α-chitin and chitosan from six different aquatic invertebrates. Food Biophys. 9, 145–157 (2014). https://doi.org/10.1007/s11483-013-9327-y
J. Chakravarty, M.F. Rabbi, N. Bach, V. Chalivendra, C.L. Yang, C.J. Brigham, Fabrication of porous chitin membrane using ionic liquid and subsequent characterization and modelling studies. Carbohydr. Polym. 198, 443–451 (2018). https://doi.org/10.1016/j.carbpol.2018.06.101
J. Wang, C. Chen, Chitosan-based biosorbents: modification and application for biosorption of heavy metals and radionuclides. Bioresour. Technol. 160, 129–141 (2014). https://doi.org/10.1016/j.biortech.2013.12.110
N. Bhardwaj, S.C. Kundu, Electrospinning: a fascinating fiber fabrication technique. Biotechnol. Adv. 28, 325–347 (2010). https://doi.org/10.1016/j.biotechadv.2010.01.004
S. Hirano, Wet-spinning and applications of functional fibers based on chitin and chitosan. Macromol. Symp. 168, 21–30 (2001). https://doi.org/10.1002/1521-3900(200103)
B. Sibaja, E. Culbertson, P. Marshall, R. Boy, R.M. Broughton, A.A. Solano, M. Esquivel, J. Parker, L.D. Ls Fuente, M.L. Auad, Preparation of alginate-chitosan fibers with potential biomedical applications. Carbohydr. Polym. 134, 598–608 (2015). https://doi.org/10.1016/j.carbpol.2015.07.076
A.M. Abdelgawad, S.M. Hudson, O.J. Rojas, Antimicrobial wound dressing nanofiber mats from multicomponent (chitosan/silver-NPs/polyvinyl alcohol) systems. Carbohydr. Polym. 100, 166–178 (2014). https://doi.org/10.1016/j.carbpol.2012.12.043
Acknowledgements
The authors would like to thank Evelyne Nkirote and Brenda Ruto for their contribution in sample preparation and data collection. We would also like to thank members of staff of the school of food and nutrition sciences of Jomo Kenyatta University of Agriculture and Technology for their support.
Author information
Authors and Affiliations
Contributions
Conceptualization; AN and JN. Methodology; All authors. Validation; All authors; Data collection; AN. Data curation; AN and JN. Formal analysis & drafting paper; AN. Review & Editing of the manuscript; All authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ndiritu, A., Kinyuru, J., Onyango, A. et al. Functional and microstructural characteristics of chitin extracted from field cricket, house cricket, and black soldier fly cocoons. Food Measure 17, 5903–5912 (2023). https://doi.org/10.1007/s11694-023-02086-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-023-02086-1