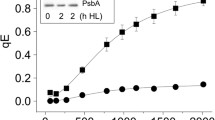
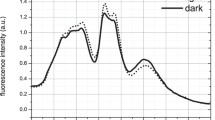
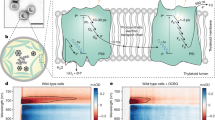
Abstract
Ultrapurified Photosystem II complexes crystalize as uniform microcrystals (PSIIX) of unprecedented homogeneity that allow observation of details previously unachievable, including the longest sustained oscillations of flash-induced O2 yield over > 200 flashes and a novel period-4.7 water oxidation cycle. We provide new evidence for a molecular-based mechanism for PSII-cyclic electron flow that accounts for switching from linear to cyclic electron flow within PSII as the downstream PQ/PQH2 pool reduces in response to metabolic needs and environmental input. The model is supported by flash oximetry of PSIIX as the LEF/CEF switch occurs, Fourier analysis of O2 flash yields, and Joliot-Kok modeling. The LEF/CEF switch rebalances the ratio of reductant energy (PQH2) to proton gradient energy (H+o/H+i) created by PSII photochemistry. Central to this model is the requirement for a regulatory site (QC) with two redox states in equilibrium with the dissociable secondary electron carrier site QB. Both sites are controlled by electrons and protons. Our evidence fits historical LEF models wherein light-driven water oxidation delivers electrons (from QA−) and stromal protons through QB to generate plastoquinol, the terminal product of PSII-LEF in vivo. The new insight is the essential regulatory role of QC. This site senses both the proton gradient (H+o/H+i) and the PQ pool redox poise via e−/H+ equilibration with QB. This information directs switching to CEF upon population of the protonated semiquinone in the Qc site (Q−H+)C, while the WOC is in the reducible S2 or S3 states. Subsequent photochemical primary charge separation (P+QA−) forms no (QH2)B, but instead undergoes two-electron backward transition in which the QC protons are pumped into the lumen, while the electrons return to the WOC forming (S1/S2). PSII-CEF enables production of additional ATP needed to power cellular processes including the terminal carboxylation reaction and in some cases PSI-dependent CEF.








Similar content being viewed by others
Data availability
All supporting data is available from C.G. or G.C.D. on request.
References
Amesz J (1964) Spectrophotometric evidence for the participation of a quinone in photosynthesis of intact blue-green algae. Biochim et Biophys Acta (BBA) Spec Sec Biophys Subj 79(2):257–265
Ananyev G, Dismukes GC (2005) How fast can Photosystem II split water? kinetic performance at high and low frequencies. Photosynth Res 84(1–3):355–365. https://doi.org/10.1007/s11120-004-7081-1
Ananyev G, Carrieri D, Dismukes GC (2008) Optimizing metabolic capacity and flux through environmental cues to maximize hydrogen production by cyanobacterium arthrospira maxima. Appl Environ Microbiol 74(19):6102–6113
Ananyev G, Gates C, Dismukes GC (2017a) The multiplicity of roles for (Bi)carbonate in photosystem II operation in the hypercarbonate-requiring cyanobacterium arthrospira maxima. Photosynthetica 56:217–228
Ananyev G, Gates C, Kaplan A (1858b) Dismukes GC (2017b) Photosystem II-cyclic electron flow powers exceptional photoprotection and record growth in the microalga Chlorella ohadii. Biochim et Biophys Acta (BBA)-Bioenerg 11:873–883. https://doi.org/10.1016/j.bbabio.2017b.07.001
Ananyev G, Gates C, Dismukes GC (2016) The oxygen quantum yield in diverse algae and cyanobacteria is controlled by partitioning of flux between linear and cyclic electron flow within photosystem II. Biochem Biophys Acta 1857:1380–1391
Ananyev G, Roy-Chowdhury S, Gates C, Fromme P, Dismukes GC (2019) The catalytic cycle of water oxidation in crystallized photosystem II complexes: performance and requirements for formation of intermediates. ACS Catal 9(2):1396–1407. https://doi.org/10.1021/acscatal.8b04513
Belyaeva NE, Schmitt F-J, Steffen R, Paschenko VZ, Riznichenko GY, Chemeris YK, Renger G, Rubin AB (2008) PS II model-based simulations of single turnover flash-induced transients of fluorescence yield monitored within the time domain of 100 ns–10 s on dark-adapted Chlorella pyrenoidosa cells. Photosynth Res 98(1):105–119. https://doi.org/10.1007/s11120-008-9374-2
Belyaeva NE, Bulychev AA, Riznichenko GY, Rubin AB (2019) Analyzing both the fast and the slow phases of chlorophyll a fluorescence and P700 absorbance changes in dark-adapted and preilluminated pea leaves using a Thylakoid Membrane model. Photosynth Res 140(1):1–19. https://doi.org/10.1007/s11120-019-00627-8
Bouges-Bocquet B (1980) Kinetic models for the electron donors of photosystem II of photosynthesis. Biochim Biophys Acta 594(2–3):85–103
Causmaecker SD, Douglass JS, Fantuzzi A, Nitschke W, Rutherford AW (2019) Energetics of the exchangeable quinone, Q<sub>B</sub>, in Photosystem II. Proc Natl Acad Sci 116(39):19458–19463. https://doi.org/10.1073/pnas.1910675116
Coe J, Kupitz C, Basu S, Conrad CE, Roy-Chowdhury S, Fromme R, Fromme P (2015) Chapter twenty-two-crystallization of photosystem II for time-resolved structural studies using an X-ray free electron laser. Methods Enzymol 557:459–482
Crane F, Ehrlich B, Kegel L (1960) Plastoquinone reduction in illuminated chloroplasts. Biochem Biophys Res Commun 3(1):37–40
Dau H (1994) Molecular mechanisms and quantitative models of variable photosystem II fluorescence. Photochem Photobiol 60:1–23
Dau H, Haumann M (2007) Time-resolved X-ray spectroscopy leads to an extension of the classical S-state cycle model of photosynthetic oxygen evolution. Photosynth Res 92(3):327–343. https://doi.org/10.1007/s11120-007-9141-9
Dismukes GC, Siderer Y (1981) Intermediates of a polynuclear manganese center involved in photosynthetic oxidation of water. Proc Natl Acad Sci 78(1):274–278. https://doi.org/10.1073/pnas.78.1.274
Falkowski P, Fujita Y, Ley A, Mauzerall D (1986) Evidence for cyclic electron flow around photosystem II in Chlorella pyrenoidosa. Plant Physiol 81(1):310
Feikema WO, Marosvolgyi MA, Lavaud J, van Gorkom HJ (2006) Cyclic electron transfer in photosystem II in the marine diatom Phaeodactylum tricornutum. BBA-Bioenergetics 1757(7):829–834. https://doi.org/10.1016/j.bbabio.2006.06.003
Gates C, Ananyev G, Dismukes GC (2016) The strontium inorganic mutant of the water oxidizing center (CaMn4O5) of PSII improves WOC efficiency but slows electron flux through the terminal acceptors. Biochim et Biophys Acta (BBA)-Bioenerg 1857(9):1550–1560. https://doi.org/10.1016/j.bbabio.2016.06.004
Gates C, Ananyev G, Roy-Chowdhury S, Cullinane B, Miller M, Fromme P, Dismukes GC (2022) Why did nature choose manganese over cobalt to make oxygen photosynthetically on the earth? J Phys Chem B 126(17):3257–3268
Guskov A, Kern J, Gabdulkhakov A, Broser M, Zouni A, Saenger W (2009) Cyanobacterial Photosystem II at 29-angstrom resolution and the role of quinones, lipids, channels and chloride. Nat Struct Mol Biol 16(3):334–342. https://doi.org/10.1038/nsmb.1559
Hasegawa K, Noguchi T (2014) Molecular interactions of the quinone electron acceptors QA, QB, and QC in photosystem II as studied by the fragment molecular orbital method. Photosynth Res 120(1):113–123
Huang J-Y, Chiu Y-F, Ortega JM, Wang H-T, Tseng T-S, Ke S-C, Roncel M, Chu H-A (2016) Mutations of cytochrome b 559 and PsbJ on and near the QC site in Photosystem II influence the regulation of short-term light response and photosynthetic growth of the cyanobacterium Synechocystis sp. PCC 6803. Biochemistry 55(15):2214–2226
Jenson DL, Barry BA (2009) Proton-coupled electron transfer in photosystem II: proton inventory of a redox active tyrosine. J Am Chem Soc 131(30):10567–10573. https://doi.org/10.1021/ja902896e
Joliot P, Joliot A (1968) A polarographic method for detection of oxygen production and reduction of Hill reagent by isolated chloroplasts. Biochim et Biophys Acta (BBA)-Bioenerg 153(3):625–634
Joliot P, Barbieri G, Chabaud R (1969) A new model of photochemical centers in system-2. Photochem Photobiol 10(5):309–329
Kato Y, Akita F, Nakajima Y, Suga M, Umena Y, Shen J-R, Noguchi T (2018) Fourier transform infrared analysis of the S-state cycle of water oxidation in the microcrystals of photosystem II. J Phys Chem Lett 9(9):2121–2126
Kedem I, Milrad Y, Kaplan A, Yacoby I (2021) Juggling lightning: how Chlorella ohadii handles extreme energy inputs without damage. Photosynth Res 147(3):329–344
Kok B, Forbush B, McGloin M (1970) Cooperation of charges in photosynthetic oxygen evolution part 1: a linear 4 step mechanism. Photochem Photobiol 11(6):457–475. https://doi.org/10.1111/j.1751-1097.1970.tb06017.x
Krivanek R, Kern J, Zouni A, Dau H, Haumann M (2007) Spare quinones in the QB cavity of crystallized photosystem II from Thermosynechococcus elongatus. Biochim et Biophys Acta (BBA)-Bioenerg 1767(6):520–527
Kupitz C, Basu S, Grotjohann I, Fromme R, Zatsepin NA, Rendek KN, Hunter MS, Shoeman RL, White TA, Wang D (2014a) Serial time-resolved crystallography of photosystem II using a femtosecond X-ray laser. Nature 513(7517):261–265
Kupitz C, Grotjohann I, Conrad CE, Roy-Chowdhury S, Fromme R, Fromme P (2014b) Microcrystallization techniques for serial femtosecond crystallography using photosystem II from Thermosynechococcus elongatus as a model system. Phil Trans R Soc B 369(1647):20130316
Laisk A, Oja V (2018) Kinetics of photosystem II electron transport: a mathematical analysis based on chlorophyll fluorescence induction. Photosynth Res 136(1):63–82. https://doi.org/10.1007/s11120-017-0439-y
Lambreva MD, Russo D, Polticelli F, Scognamiglio V, Antonacci A, Zobnina V, Campi G, Rea G (2014) Structure/function/dynamics of photosystem II plastoquinone binding sites. Curr Protein Pept Sci 15(4):285–295
Lavergne J, Junge W (1993) Proton release during the redox cycle of the water oxidase. Photosynth Res 38(3):279–296. https://doi.org/10.1007/bf00046752
Lazár D, Jablonský J (2009) On the approaches applied in formulation of a kinetic model of photosystem II: different approaches lead to different simulations of the chlorophyll a fluorescence transients. J Theor Biol 257(2):260–269
Mani K, Zournas A, Dismukes GC (2022) Bridging the gap between Kok-type and kinetic models of photosynthetic electron transport within Photosystem II. Photosynth Res 151(1):83–102. https://doi.org/10.1007/s11120-021-00868-6
Manoj KM, Gideon DA, Parashar A, Nirusimhan V, Annadurai P, Jacob VD, Manekkathodi A (2021) Validating the predictions of murburn model for oxygenic photosynthesis: Analyses of ligand-binding to protein complexes and cross-system comparisons. J Biomol Struct Dyn. https://doi.org/10.1080/07391102.2021.1953607
Meunier PC (1993) Oxygen evolution by photosystem II: the contribution of backward transitions to the anomalous behaviour of double-hits revealed by a new analysis method. Photosynth Res 36(2):111–118
Meunier P, Burnap R (1996) Improved 5-step modeling of the Photosystem II S-state mechanism in cyanobacteria. Photosynth Res 47(1):61–76
Oja V, Laisk A (2020) Time-and reduction-dependent rise of photosystem II fluorescence during microseconds-long inductions in leaves. Photosynth Res 145(3):209–225
Pham LV, Messinger J (2014) Electrochemically produced hydrogen peroxide affects Joliot-type oxygen-evolution measurements of photosystem II. Biochim et Biophys Acta (BBA)-Bioenerg 1837(9):1411–1416. https://doi.org/10.1016/j.bbabio.2014.01.013
Pham LV, Messinger J (2016) Probing S-state advancements and recombination pathways in photosystem II with a global fit program for flash-induced oxygen evolution pattern. Biochim et Biophys Acta (BBA)-Bioenerg 1857(6):848–859. https://doi.org/10.1016/j.bbabio.2016.03.013
Pham LV, Janna Olmos JD, Chernev P, Kargul J, Messinger J (2019) Unequal misses during the flash-induced advancement of photosystem II: effects of the S state and acceptor side cycles. Photosynth Res 139(1):93–106
Prasil O, Kolber Z, Berry J, Falkowski P (1996) Cyclic electron flow around photosystem II in vivo. Photosynth Res 48(3):395–410
Rappaport F, Guergova-Kuras M, Nixon PJ, Diner BA, Lavergne J (2002) Kinetics and pathways of charge recombination in photosystem II. Biochemistry 41(26):8518–8527
Robinson HH, Crofts AR (1983) Kinetics of the oxidation—reduction reactions of the photosystem II quinone acceptor complex, and the pathway for deactivation. FEBS Lett 153(1):221–226
Satoh K, Koike H, Ichimura T, Katoh S (1992) Binding affinities of benzoquinones to the QB site of Photosystem II in Synechococcus oxygen-evolving preparation. Biochim et Biophys Acta (BBA)-Bioenerg 1102(1):45–52. https://doi.org/10.1016/0005-2728(92)90063-8
Schatz GH, Brock H, Holzwarth AR (1988) Kinetic and energetic model for the primary processes in photosystem II. Biophys J 54(3):397–405. https://doi.org/10.1016/S0006-3495(88)82973-4
Shinkarev V (1996) Binary oscillations in the Kok model of oxygen evolution in oxygenic photosynthesis. Photosynth Res 48(3):411–417. https://doi.org/10.1007/BF00029473
Shinkarev VP (2003) Oxygen evolution in photosynthesis: simple analytical solution for the Kok model. Biophys J 85(1):435–441
Shinkarev V (2005a) Flash-induced oxygen evolution in photosynthesis: simple solution for the extended s-state model that includes misses, double hits, inactivation, and backwards transitions. Biophys J 88:412–421
Shinkarev VP (2005b) Flash-induced oxygen evolution and other oscillation processes in Photosystem II. In: Wydrzynski T, Satoh K (eds) Photosystem II: The Water/Plastoquinone Oxido-Reductase In Photosynthesis. Klewer Academic Publishers, The Netherlands
Shinkarev VP, Wraight CA (1993a) Kinetic factors in the bicycle model of oxygen evolution by Photosystem II. Photosynth Res 38(3):315–321. https://doi.org/10.1007/BF00046756
Shinkarev VP, Wraight CA (1993b) Oxygen evolution in photosynthesis: from unicycle to bicycle. Proc Natl Acad Sci USA 90(5):1834–1838
Shinopoulos KE, Brudvig GW (2012) Cytochrome b 559 and cyclic electron transfer within photosystem II. Biochim et Biophys Acta (BBA)-Bioenerg 1817(1):66–75
Steffen M (1990) A simple method for monotonic interpolation in one dimension. Astron Astrophys 239:443
Swarztrauber PN (1982) Vectorizing the FFTs. In: Rodrigue G (ed) Parallel Computations". Academic Press, Cambridge
Takagi D, Ifuku K, Nishimura T, Miyake C (2019) Antimycin A inhibits cytochrome b559-mediated cyclic electron flow within photosystem II. Photosynth Res 139(1):487–498
Vass I (2012) Molecular mechanisms of photodamage in the Photosystem II complex. Biochim et Biophys Acta (BBA)-Bioenerg 1817(1):209–217
Vass I, Cser K (2009) Janus-faced charge recombinations in photosystem II photoinhibition. Trends Plant Sci 14(4):200–205
Vinyard DJ, Zachary CE, Ananyev G (1827) Dismukes GC (2013) Thermodynamically accurate modeling of the catalytic cycle of photosynthetic oxygen evolution: a mathematical solution to asymmetric Markov chains. Biochim Biophys Acta 7:861–868. https://doi.org/10.1016/j.bbabio.2013.04.008
Wraight C (1979) Electron acceptors of bacterial photosynthetic reaction centers II. H+ binding coupled to secondary electron transfer in the quinone acceptor complex. Biochim et Biophys Acta (BBA)-Bioenerg 548(2):309–327
Yadav DK, Prasad A, Kruk J, Pospíšil P (2014) Evidence for the involvement of loosely bound plastosemiquinones in superoxide anion radical production in photosystem II. PLoS ONE 9(12):e115466. https://doi.org/10.1371/journal.pone.0115466
Yanykin DV, Khorobrykh AA, Khorobrykh SA, Klimov VV (2010) Photoconsumption of molecular oxygen on both donor and acceptor sides of photosystem II in Mn-depleted subchloroplast membrane fragments. Biochim et Biophys Acta (BBA)-Bioenerg 1797(4):516–523
Yanykin D, Khorobrykh A, Terentyev V, Klimov V (2017) Two pathways of photoproduction of organic hydroperoxides on the donor side of photosystem 2 in subchloroplast membrane fragments. Photosynth Res 133(1):129–138
Yao M, Liu Y, Fei L, Zhou Y, Wang F, Chen J (2018) Self-adaptable quinone–quinol exchange mechanism of photosystem II. J Phys Chem B 122(46):10478–10489
Zhu X-G, Baker NR, Ort DR, Long SP (2005) Chlorophyll a fluorescence induction kinetics in leaves predicted from a model describing each discrete step of excitation energy and electron transfer associated with photosystem II. Planta 223(1):114–133
Acknowledgements
This work was funded by the Department of Energy, Basic Energy Sciences, Grant DE-FG02-10ER16195 (to G.C.D. and G.A.) and the NSF Science and Technology BioXFEL center award 1231306 (to P.F.).
Funding
Basic Energy Sciences, DE-FG02-10ER16195,G. Charles Dismukes,BioXFEL Science and Technology Center, 1231306, Petra Fromme
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is dedicated to the memory of Professor Kenneth Sauer who made important contributions to understanding of the photosystems and the Z-scheme.
Also, we had a dedication to another friend and mentor to the authors, Prof. Ron Pace, who also passed away during the production of this paper and who served as an inspiration to the work. The dedication read. This work is dedicated to the memory of Professor Ronald Pace whose dogged questioning and insightful ideas inspired us to ask why, to delve deeper, and to always treat historical interpretations as conditional models.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gates, C., Ananyev, G., Roy-Chowdhury, S. et al. Regulation of light energy conversion between linear and cyclic electron flow within photosystem II controlled by the plastoquinone/quinol redox poise. Photosynth Res 156, 113–128 (2023). https://doi.org/10.1007/s11120-022-00985-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-022-00985-w