Abstract
Key message
This report shows detailed characterization of LOX gene family in sorghum and provides new insight of sorghum LOX genes in genetic structure and their roles in plant response to infestation by sugarcane aphids.
Abstract
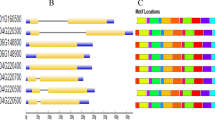
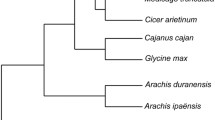
Lipoxygenases (LOXs) are monomeric, nonheme iron-containing dioxygenases that initiate the fatty acid oxidation pathway creating oxylipins and plant hormone jasmonate both have a key role in plant development and defense. To date, a comprehensive and systematic analysis of sorghum LOXs is still deficient. Thus, we performed a genome-wide analysis of the sorghum LOXs genome and identified nine LOXs genes. Detailed examination of protein sequences and phylogenetic analysis categorized the sorghum LOXs into two subclasses, 9-LOXs (SbLOX1, SbLOX3, SbLOX4, SbLOXm, and SbLOXo), 13-LOXs (SbLOX9, SbLOX5, and SbLOX2), and the unclassified SbLOX8. This classification was further supported by sequence similarity/identity matrix and subcellular localization analysis. The lipoxygenase domains, motifs, and vital amino acids were highly conserved in all sorghum LOX genes. In silico analysis of the promoter region of SbLOXs identified different hormones responsive cis-elements. Furthermore, to explore the roles of sorghum LOXs during sugarcane aphid feeding and exogenous MeJA application, expression analysis was conducted for all the eight LOXs in resistant (Tx2783) and susceptible (Tx7000) sorghum lines, respectively. As detailed in this report, the data generated from both genome-wide identification and expression analysis of lipoxygenase genes suggest the putative functions of two 13-LOXs (SbLOX9 and SbLOX5) and three 9-LOXs (SbLOX1, SbLOX3, and SbLOXo) in biosynthesis of jasmonic acid, green leaf volatiles and death acids, and all of them are involved in defense-related functions in plants. Furthermore, this report represents the first genome-wide analysis of the LOX gene family in sorghum, which will facilitate future studies to characterize the roles of each individual LOXs gene in aphid resistance and defense responses to other stresses.

Modified from Schiller et al. (2015) and Woldemariam et al. (2017)






Similar content being viewed by others
References
Allmann S, Halitschke R, Schuurink RC, Baldwin IT (2010) Oxylipin channelling in Nicotiana attenuata: lipoxygenase 2 supplies substrates for green leaf volatile production. Plant, Cell Environ 33:2028–2040
Bailly C, Bogatek-Leszczynska R, Côme D, Corbineau F (2002) Changes in activities of antioxidant enzymes and lipoxygenase during growth of sunflower seedlings from seeds of different vigour Seed Science Research 12:47
Bannenberg G, Martínez M, Hamberg M, Castresana C (2009) Diversity of the enzymatic activity in the lipoxygenase gene family of Arabidopsis thaliana. Lipids 44:85
Barry CS, Giovannoni JJ (2007) Ethylene and fruit ripening. J Plant Growth Regul 26:143
Bell E, Creelman RA, Mullet JE (1995) A chloroplast lipoxygenase is required for wound-induced jasmonic acid accumulation in Arabidopsis. Proc Natl Acad Sci 92:8675–8679
Bowling RD et al (2016) Sugarcane aphid (Hemiptera: Aphididae): a new pest on sorghum in North America. Journal of Integrated Pest Management 7:12
Brash AR (1999) Lipoxygenases: occurrence, functions, catalysis, and acquisition of substrate. J Biol Chem 274:23679–23682
Caldelari D, Wang G, Farmer EE, Dong X (2011) Arabidopsis lox3 lox4 double mutants are male sterile and defective in global proliferative arrest. Plant Mol Biol 75:25–33
Chang J, Duan X, Cui J, Xue W, Zhang Q (2012) Differential molecular responses of aphid-sensitive and aphid-resistant sorghum lines to aphid infestation Arthropod-Plant. Interactions 6:113–120
Chen Z, Chen X, Yan H, Li W, Li Y, Cai R, **ang Y (2015) The lipoxygenase gene family in poplar: identification, classification, and expression in response to MeJA treatment. PLoS ONE 10:e0125526
Christensen SA et al. (2015) Maize death acids, 9-lipoxygenase–derived cyclopente (a) nones, display activity as cytotoxic phytoalexins and transcriptional mediators Proceedings of the National Academy of Sciences 112:11407–11412
Christensen SA et al (2013) The maize lipoxygenase, Zm LOX 10, mediates green leaf volatile, jasmonate and herbivore-induced plant volatile production for defense against insect attack. Plant J 74:59–73
Christensen SA, Huffaker A, Hunter CT, Alborn HT, Schmelz EA (2016) A maize death acid, 10-oxo-11-phytoenoic acid, is the predominant cyclopentenone signal present during multiple stress and developmental conditions. Plant Signaling Behavior 11:e1120395
Constantino N et al (2013) Root-expressed maize lipoxygenase 3 negatively regulates induced systemic resistance to Colletotrichum graminicola in shoots. Frontiers in Plant Science 4:510
Crooks GE, Hon G, Chandonia J-M, Brenner SE (2004) WebLogo: a sequence logo generator. Genome Res 14:1188–1190
Edgar RC (2004) MUSCLE: a multiple sequence alignment method with reduced time and space complexity. BMC Bioinformatics 5:113
Ellis C, Karafyllidis I, Turner JG (2002) Constitutive activation of jasmonate signaling in an Arabidopsis mutant correlates with enhanced resistance to Erysiphe cichoracearum. Pseudomonas syringae, and Myzus persicae Molecular Plant-Microbe Interactions 15:1025–1030
Emanuelsson O, Nielsen H, Heijne GV (1999) ChloroP, a neural network-based method for predicting chloroplast transit peptides and their cleavage sites. Protein Sci 8:978–984
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Feussner I, Wasternack C (2002) The lipoxygenase pathway. Annu Rev Plant Biol 53:275–297
Glauser G, Dubugnon L, Mousavi SA, Rudaz S, Wolfender J-L, Farmer EE (2009) Velocity estimates for signal propagation leading to systemic jasmonic acid accumulation in wounded Arabidopsis. J Biol Chem 284:34506–34513
Goldman N, Yang Z (1994) A codon-based model of nucleotide substitution for protein-coding DNA sequences. Mol Biol Evol 11:725–736
Harwood JL (2000) Eicosanoids and related compounds in plants and animals. In: AF Rowley, H. Kuhn and T. Schewe (Ed.). London, UK: Portland Press Ltd, The New Phytologist. 146:23–25
Heitz T, Bergey DR, Ryan CA (1997) A gene encoding a chloroplast-targeted lipoxygenase in tomato leaves is transiently induced by wounding, systemin, and methyl jasmonate. Plant Physiol 114:1085–1093
Hu B, ** J, Guo A-Y, Zhang H, Luo J, Gao G (2015) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31:1296–1297
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549
Lescot M et al (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327
Li H, Huang Y (2017) Expression of brown-midrib in a spontaneous sorghum mutant is linked to a 5′-UTR deletion in lignin biosynthesis gene SbCAD2. Scientific Reports 7:1–10
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408
Ma F, Yang X, Shi Z, Miao X (2020) Novel crosstalk between ethylene-and jasmonic acid-pathway responses to a piercing–sucking insect in rice. New Phytol 225:474–487
Melan MA, Dong X, Endara ME, Davis KR, Ausubel FM, Peterman TK (1993) An Arabidopsis thaliana lipoxygenase gene can be induced by pathogens, abscisic acid, and methyl jasmonate. Plant Physiol 101:441–450
Mewis I, Tokuhisa JG, Schultz JC, Appel HM, Ulrichs C, Gershenzon J (2006) Gene expression and glucosinolate accumulation in Arabidopsis thaliana in response to generalist and specialist herbivores of different feeding guilds and the role of defense signaling pathways. Phytochemistry 67:2450–2462
Moran PJ, Thompson GA (2001) Molecular responses to aphid feeding in Arabidopsis in relation to plant defense pathways. Plant Physiol 125:1074–1085
Nalam VJ, Keeretaweep J, Sarowar S, Shah J (2012) Root-derived oxylipins promote green peach aphid performance on Arabidopsis foliage. Plant Cell 24:1643–1653
Nalam VJ, Keereetaweep J, Shah J (2013) The green peach aphid, Myzus persicae, acquires a LIPOXYGENASE5-derived oxylipin from Arabidopsis thaliana, which promotes colonization of the host plant. Plant Signaling Behavior 8:e22735
Ogunola OF et al (2017) Characterization of the maize lipoxygenase gene family in relation to aflatoxin accumulation resistance. PLoS ONE 12:e0181265
Padilla MN, Hernández ML, Sanz C, Martinez-Rivas JM (2009) Functional characterization of two 13-lipoxygenase genes from olive fruit in relation to the biosynthesis of volatile compounds of virgin olive oil. Journal of Agricultural and Food Chemistry 57:9097–9107
Padilla MN, Hernández ML, Sanz C, Martínez-Rivas JM (2012) Molecular cloning, functional characterization and transcriptional regulation of a 9-lipoxygenase gene from olive. Phytochemistry 74:58–68
Quevillon E, Silventoinen V, Pillai S, Harte N, Mulder N, Apweiler R, Lopez R (2005) InterProScan: protein domains identifier. Nucleic Acids Res 33:W116–W120
Reymond P, Weber H, Damond M, Farmer EE (2000) Differential gene expression in response to mechanical wounding and insect feeding in Arabidopsis. Plant Cell 12:707–719
Royo J, Vancanneyt G, Pérez AG, Sanz C, Störmann K, Rosahl S, Sánchez-Serrano JJ (1996) Characterization of three potato lipoxygenases with distinct enzymatic activities and different organ-specific and wound-regulated expression patterns. J Biol Chem 271:21012–21019
Sarde SJ, Kumar A, Remme RN, Dicke M (2018) Genome-wide identification, classification and expression of lipoxygenase gene family in pepper. Plant Mol Biol 98:375–387
Schommer C et al (2008) Control of jasmonate biosynthesis and senescence by miR319 targets. PLoS Biol 6:e230
Sharp AJ et al (2005) Segmental duplications and copy-number variation in the human genome. The American Journal of Human Genetics 77:78–88
Shen J et al (2014) A 13-lipoxygenase, TomloxC, is essential for synthesis of C5 flavour volatiles in tomato. J Exp Bot 65:419–428
Shivaji R et al (2010) Plants on constant alert: elevated levels of jasmonic acid and jasmonate-induced transcripts in caterpillar-resistant maize. J Chem Ecol 36:179–191
Siedow JN (1991) Plant lipoxygenase: structure and function. Annu Rev Plant Biol 42:145–188
Singh B, Padmaja P, Seetharama N (2004) Biology and management of the sugarcane aphid, Melanaphis sacchari (Zehntner)(Homoptera: Aphididae), in sorghum: a review. Crop Protection 23:739–755
Song H, Wang P, Li C, Han S, Lopez-Baltazar J, Zhang X, Wang X (2016) Identification of lipoxygenase (LOX) genes from legumes and their responses in wild type and cultivated peanut upon Aspergillus flavus infection. Scientific reports 6:35245
Steczko J, Donoho GP, Clemens JC, Dixon JE, Axelrod B (1992) Conserved histidine residues in soybean lipoxygenase: functional consequences of their replacement. Biochemistry 31:4053–4057
Team RC (2013) R: A language and environment for statistical computing. Austria, Vienna
Thaler JS, Humphrey PT, Whiteman NK (2012) Evolution of jasmonate and salicylate signal crosstalk. Trends Plant Sci 17:260–270
Tzin V et al (2015) Dynamic maize responses to aphid feeding are revealed by a time series of transcriptomic and metabolomic assays. Plant Physiol 169:1727–1743
ul Hassan MN, Zainal Z, Ismail I, (2015) Green leaf volatiles: biosynthesis, biological functions and their applications in biotechnology. Plant Biotechnol J 13:727–739
Umate P (2011) Genome-wide analysis of lipoxygenase gene family in Arabidopsis and rice. Plant Signaling Behavior 6:335–338
Upadhyay RK, Mattoo AK (2018) Genome-wide identification of tomato (Solanum lycopersicum L.) lipoxygenases coupled with expression profiles during plant development and in response to methyl-jasmonate and wounding. J Plant Physiol 231:318–328
USDA N (2017) Quick stats USDA National Agriculural Statistics Service, Washington, DC https://quickstats.nass.usda.gov/. Accessed 3 May 2019
Wang JW, Wu JY (2005) Nitric oxide is involved in methyl jasmonate-induced defense responses and secondary metabolism activities of Taxus cells. Plant Cell Physiol 46:923–930
Wasternack C, Parthier B (1997) Jasmonate-signalled plant gene expression. Trends Plant Sci 2:302–307
Woldemariam MG, Ahern K, Jander G, Tzin V (2018) A role for 9-lipoxygenases in maize defense against insect herbivory. Plant Signaling Behavior 13:4709–4723
Yan L et al (2013) Role of tomato lipoxygenase D in wound-induced jasmonate biosynthesis and plant immunity to insect herbivores. PLoS Genet 9:e1003964
Yang F et al (2015) Analysis of key genes of jasmonic acid mediated signal pathway for defense against insect damages by comparative transcriptome sequencing. Scientific Reports 5:16500
Yu C-S, Cheng C-W, Su W-C, Chang K-C, Huang S-W, Hwang J-K, Lu C-H (2014) CELLO2GO: a web server for protein subCELlular LOcalization prediction with functional gene ontology annotation. PLoS ONE 9:e99368
Zarate SI, Kempema LA, Walling LL (2007) Silverleaf whitefly induces salicylic acid defenses and suppresses effectual jasmonic acid defenses. Plant Physiol 143:866–875
Zhang B, Chen K, Bowen J, Allan A, Espley R, Karunairetnam S, Ferguson I (2006) Differential expression within the LOX gene family in ripening kiwifruit. J Exp Bot 57:3825–3836
Zhang P-J, He Y-C, Zhao C, Ye Z-H, Yu X-P (2018) Jasmonic acid-dependent defenses play a key role in defending tomato against Bemisia tabaci nymphs, but not adults. Frontiers in plant science 9:1065
Zhou G, Qi J, Ren N, Cheng J, Erb M, Mao B, Lou Y (2009) Silencing OsHI-LOX makes rice more susceptible to chewing herbivores, but enhances resistance to a phloem feeder. Plant J 60:638–648
Zhou G et al (2014) The 9-lipoxygenase Osr9-LOX1 interacts with the 13-lipoxygenase-mediated pathway to regulate resistance to chewing and piercing-sucking herbivores in rice. Physiol Plant 152:59–69
Zhu-Salzman K, Salzman RA, Ahn J-E, Koiwa H (2004) Transcriptional regulation of sorghum defense determinants against a phloem-feeding aphid. Plant Physiol 134:420–431
Zuckerkandl E, Pauling L (1965) Evolutionary divergence and convergence in proteins. In: Bryson V, Vogel HJ (eds) Evolving genes and proteins. Elsevier, Amsterdam, pp 97–166
Acknowledgements
We thank Ms. Angela L. Phillips for her excellent technical assistance. The U.S. Department of Agriculture, Agricultural Research Service, is an equal opportunity provider and employer and all agency services are available without discrimination. Mention of a trademark or proprietary product does not constitute a guarantee or warranty of a product by the U.S. Department of Agriculture and does not imply its approval to the exclusion of other products that may also be suitable.
Funding
This work was partly supported by funding from USDA-ARS CRIS project, Grant Number: 3072–21000-009-00D (YH).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiment: KS, SP and YH. Performed the experiments: KS and SP. Analyzed the data: KS and YH. Wrote the manuscript: KS and YH.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shrestha, K., Pant, S. & Huang, Y. Genome-wide identification and classification of Lipoxygenase gene family and their roles in sorghum-aphid interaction. Plant Mol Biol 105, 527–541 (2021). https://doi.org/10.1007/s11103-020-01107-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-01107-7