Abstract
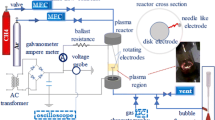
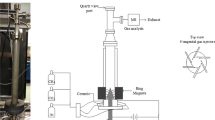
Hydrogen and solid carbon were produced through methane decomposition in a plasma reactor with a parallel set of screw type helix and rod-like electrodes. The novel configuration led to the 3-dimensional movement of plasma zone in the axial and angular directions as well as arc elongation. The effect of arc elongation and movement at various angular velocities of high voltage electrode was investigated on the reactor performance in terms of methane conversion, hydrogen yield, and energy yield. In addition, the influence of fluid flow direction was considered. Methane conversion of 47% and hydrogen production rate of 132.7 ml/min with the energy yield of 36.8 g/kWh were achieved at stable operating conditions. The deposition rate of carbon was 35 mg/min which had a graphite-like structure. A reaction pathway is proposed according to reaction path analysis in order to interpret the underlying plasma chemical process.















Similar content being viewed by others
References
Khoja AH, Tahir M, Amin NAS (2017) Dry reforming of methane using different dielectric materials and DBD plasma reactor configurations. Energy Convers Manag 144:262–274
Khalifeh O, Taghvaei H, Mosallanejad A, Rahimpour MR, Shariati A (2016) Extra pure hydrogen production through methane decomposition using nanosecond pulsed plasma and Pt–Re catalyst. Chem Eng J 294:132–145
Cho DL, Kim H-N, Lee M, Cho E (2015) Production of pure hydrogen from methane by low temperature plasma processing. Korean J Chem Eng 32(12):2519–2523. https://doi.org/10.1007/s11814-015-0107-x
Moshrefi MM, Rashidi F (2014) Hydrogen production from methane by DC spark discharge: effect of current and voltage. J Nat Gas Sci Eng 16:85–89
Mizeraczyk J, Jasiński M (2016) Plasma processing methods for hydrogen production. Eur Phys J Appl Phys 75(2):24702
Mizeraczyk J, Urashima K, Jasiński M, Dors M (2014) Hydrogen production from gaseous fuels by plasmas-a review. Int J Plasma Env Sci Technol 8(2):89–97
Ibrahim AA, Fakeeha AH, Al-Fatesh AS, Abasaeed AE, Khan WU (2015) Methane decomposition over iron catalyst for hydrogen production. Int J Hydrog Energy 40(24):7593–7600
Moshrefi MM, Rashidi F, Bozorgzadeh HR (2015) Use of a DC discharge in a plasma reactor with a rotating ground electrode for production of synthesis gas by partial oxidation of methane. Res Chem Intermed 41(9):5941–5959
Ghorbanzadeh A, Matin N (2005) Methane conversion to hydrogen and higher hydrocarbons by double pulsed glow discharge. Plasma Chem Plasma Process 25(1):19–29
Li D, Li X, Bai M, Tao X, Shang S, Dai X, Yin Y (2009) CO2 reforming of CH4 by atmospheric pressure glow discharge plasma: a high conversion ability. Int J Hydrog Energy 34(1):308–313
Aleknaviciute I, Karayiannis T, Collins M, Xanthos C (2013) Methane decomposition under a corona discharge to generate COx-free hydrogen. Energy 59:432–439
Horvath G, Zahoran M, Mason N, Matejcik S (2011) Methane decomposition leading to deposit formation in a DC positive CH4–N2 corona discharge. Plasma Chem Plasma Process 31(2):327–335
Kundu SK, Kennedy EM, Gaikwad VV, Molloy TS, Dlugogorski BZ (2012) Experimental investigation of alumina and quartz as dielectrics for a cylindrical double dielectric barrier discharge reactor in argon diluted methane plasma. Chem Eng J 180:178–189
Kim TK, Lee WG (2012) Reaction between methane and carbon dioxide to produce syngas in dielectric barrier discharge system. J Ind Eng Chem 18(5):1710–1714
Li XD, Zhang H, Yan SX, Yan JH, Du CM (2013) Hydrogen production from partial oxidation of methane using an AC rotating gliding arc reactor. IEEE Trans Plasma Sci 41(1):126–132
Zhang H, Du C, Wu A, Bo Z, Yan J, Li X (2014) Rotating gliding arc assisted methane decomposition in nitrogen for hydrogen production. Int J Hydrog Energy 39(24):12620–12635
Zhang H, Wang W, Li X, Han L, Yan M, Zhong Y, Tu X (2018) Plasma activation of methane for hydrogen production in a N2 rotating gliding arc warm plasma: a chemical kinetics study. Chem Eng J 345:67–78
Rahim I, Nomura S, Mukasa S, Toyota H (2015) Decomposition of methane hydrate for hydrogen production using microwave and radio frequency in-liquid plasma methods. Appl Therm Eng 90:120–126
Mizeraczyk J, Jasiński M, Nowakowska H, Dors M (2012) Studies of atmospheric-pressure microwave plasmas used for gas processing. Nukleonika 57:241–247
Chung W-C, Chang M-B (2016) Dry reforming of methane by combined spark discharge with a ferroelectric. Energy Convers Manag 124:305–314
Moshrefi MM, Rashidi F, Bozorgzadeh HR, Haghighi ME (2013) Dry reforming of methane by DC spark discharge with a rotating electrode. Plasma Chem Plasma Process 33(2):453–466
Li X-S, Lin C-K, Shi C, Xu Y, Wang Y-N, Zhu A-M (2008) Stable kilohertz spark discharges for high-efficiency conversion of methane to hydrogen and acetylene. J Phys D Appl Phys 41(17):175203
Lee DH, Song Y-H, Kim K-T, Lee J-O (2013) Comparative study of methane activation process by different plasma sources. Plasma Chem Plasma Process 33(4):647–661
Lee DH, Kim K-T, Cha MS, Song Y-H (2010) Plasma-controlled chemistry in plasma reforming of methane. Int J Hydrog Energy 35(20):10967–10976
Yang Y (2003) Direct non-oxidative methane conversion by non-thermal plasma: modeling study. Plasma Chem Plasma Process 23(2):327–346
Yang Y (2003) Direct non-oxidative methane conversion by non-thermal plasma: experimental study. Plasma Chem Plasma Process 23(2):283–296
Pristavita R, Mendoza-Gonzalez N-Y, Meunier J-L, Berk D (2010) Carbon blacks produced by thermal plasma: the influence of the reactor geometry on the product morphology. Plasma Chem Plasma Process 30(2):267–279
Pristavita R, Meunier J-L, Berk D (2011) Carbon nano-flakes produced by an inductively coupled thermal plasma system for catalyst applications. Plasma Chem Plasma Process 31(2):393–403
Okeke L, Störi H (1991) Plasma-chemical decomposition of methane during diamond synthesis. Plasma Chem Plasma Process 11(4):489–499
Lee H, Lee D-H, Song Y-H, Choi WC, Park Y-K, Kim DH (2015) Synergistic effect of non-thermal plasma–catalysis hybrid system on methane complete oxidation over Pd-based catalysts. Chem Eng J 259:761–770
Ogo S, Sekine Y (2017) Catalytic reaction assisted by plasma or electric field. Chem Rec 17(8):726–738
Moshrefi MM, Rashidi F (2018) Hydrogen production from methane decomposition in cold plasma reactor with rotating electrodes. Plasma Chem Plasma Process 38(3):503–515. https://doi.org/10.1007/s11090-018-9875-5
Janev R, Reiter D (2002) Collision processes of CH y and CH y + hydrocarbons with plasma electrons and protons. Phys Plasmas 9(9):4071–4081
Legrand J, Diamy A, Hrach R, Hrachova V (1998) Kinetics of reactions in CH4\N2 afterglow plasma: a simplified model. Vacuum 50(3–4):491–495
Morgan NN, ElSabbagh M (2017) Hydrogen production from methane through pulsed DC plasma. Plasma Chem Plasma Process 37(5):1375–1392
Kado S, Urasaki K, Sekine Y, Fujimoto K, Nozaki T, Okazaki K (2003) Reaction mechanism of methane activation using non-equilibrium pulsed discharge at room temperature. Fuel 82(18):2291–2297
Chiremba E, Zhang K, Kazak C, Akay G (2017) Direct nonoxidative conversion of methane to hydrogen and higher hydrocarbons by dielectric barrier discharge plasma with plasma catalysis promoters. AIChE J 63(10):4418–4429. https://doi.org/10.1002/aic.15769
Gao Y, Zhang S, Sun H, Wang R, Tu X, Shao T (2018) Highly efficient conversion of methane using microsecond and nanosecond pulsed spark discharges. Appl Energy 226:534–545. https://doi.org/10.1016/j.apenergy.2018.06.006
Khalifeh O, Mosallanejad A, Taghvaei H, Rahimpour MR, Shariati A (2016) Decomposition of methane to hydrogen using nanosecond pulsed plasma reactor with different active volumes, voltages and frequencies. Appl Energy 169:585–596
Moshrefi MM, Rashidi F, Bozorgzadeh HR, Zekordi SM (2012) Methane conversion to hydrogen and carbon black by DC-spark discharge. Plasma Chem Plasma Process 32(6):1157–1168
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kheirollahivash, M., Rashidi, F. & Moshrefi, M.M. Hydrogen Production from Methane Decomposition Using a Mobile and Elongating Arc Plasma Reactor. Plasma Chem Plasma Process 39, 445–459 (2019). https://doi.org/10.1007/s11090-018-9950-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-018-9950-y