Abstract
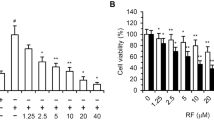
Whether allicin can suppress the angiogenesis via inhibiting the activity of vascular endothelial cells (VECs) in preventing epidural hypertrophic scars remains unknown. VECs were treated by allicin at a gradient of concentrations. Cell activity was measured by CCK-8 assay, scratch assay and flow cytometry. Reverse-transcription PCR and Western Blot were used to measure the expression levels of relevant genes and proteins. After treated with allicin at concentrations of 0, 25, 50 and 100 mg/L, the viability of VECs significantly decreased at 24 h (p < 0.001*) and 48 h (p < 0.001*), and migration rate significantly decreased in scratch assay (p = 0.017*) and in Transwell assay (p = 0.021*). As the concentrations of allicin increased, the apoptosis rate of VECs rose up (p = 0.018*). There was no significant difference on cell numbers at S phase (p = 0.25), but cell numbers at G1 phase decreased (p = 0.039*) and at G2 phase increased (p = 0.047*). With the increase of allicin concentrations, the ability of tube formation for VECs significantly decreased (p < 0.001*). Comparing with control group, the expression of PCNA and BCL-2 decreased (p < 0.001*), while the expression of BAX increased significantly (p < 0.001*). Regarding to JAK2/STAT3 pathway, the expression levels of JAK3 and STAT3 decreased significantly with the increase of allicin concentrations (p < 0.001*). Allicin can suppress the activity of VECs probably by regulating JAK2/STAT3 pathway.



Similar content being viewed by others
Data Availability
All data and material are available.
Code availability
The statistics were performed using SPSS version 24 (IBM Corp., USA).
References
Korntner S, Lehner C, Gehwolf R, Wagner A, Grutz M, Kunkel N, Tempfer H, Traweger A (2019) Limiting angiogenesis to modulate scar formation. Adv Drug Deliv Rev 146:170–189. https://doi.org/10.1016/j.addr.2018.02.010
Bosscher HA, Heavner JE (2010) Incidence and severity of epidural fibrosis after back surgery: an endoscopic study. Pain practice : The Official Journal of World Institute of Pain 10(1):18–24. https://doi.org/10.1111/j.1533-2500.2009.00311.x
Coentro JQ, Pugliese E, Hanley G, Raghunath M, Zeugolis DI (2019) Current and upcoming therapies to modulate skin scarring and fibrosis. Adv Drug Deliv Rev 146:37–59. https://doi.org/10.1016/j.addr.2018.08.009
Song Y, Yu Z, Song B, Guo S, Lei L, Ma X, Su Y (2018) Usnic acid inhibits hypertrophic scarring in a rabbit ear model by suppressing scar tissue angiogenesis. Biomed Pharmacother 108:524–530. https://doi.org/10.1016/j.biopha.2018.06.176
Borlinghaus J, Albrecht F, Gruhlke MC, Nwachukwu ID, Slusarenko AJ (2014) Allicin: chemistry and biological properties. Molecules 19(8):12591–12618. https://doi.org/10.3390/molecules190812591
Sun HH, Wang JC, Feng XM, Zhu SL, Cai J (2020) Allicin inhibits proliferation and promotes apoptosis of human epidural scar fibroblasts. World Neurosurgery 136:e460–e468. https://doi.org/10.1016/j.wneu.2020.01.031
D'Argenio G, Mazzone G, Ribecco MT, Lembo V, Vitaglione P, Guarino M, Morisco F, Napolitano M, Fogliano V, Caporaso N (2013) Garlic extract attenuating rat liver fibrosis by inhibiting TGF-beta1. Clin Nutr 32(2):252–258. https://doi.org/10.1016/j.clnu.2012.07.001
Liu C, Cao F, Tang QZ, Yan L, Dong YG, Zhu LH, Wang L, Bian ZY, Li H (2010) Allicin protects against cardiac hypertrophy and fibrosis via attenuating reactive oxygen species-dependent signaling pathways. J Nutriti Biochem 21(12):1238–1250. https://doi.org/10.1016/j.jnutbio.2009.11.001
Guyer RD, Patterson M, Ohnmeiss DD (2006) Failed back surgery syndrome: diagnostic evaluation. J Am Academy Orthopaedic Surg 14(9):534–543. https://doi.org/10.5435/00124635-200609000-00003
Ozkan U, Osun A, Samancioglu A, Ercan S, Firat U, Kemaloglu S (2014) The effect of bevacizumab and 5-Fluorouracil combination on epidural fibrosis in a rat laminectomy model. European Review for Medical and Pharmacological Sciences 18(1):95–100
Wang H, Sun W, Fu D, Shen Y, Chen YY, Wang LL (2018) Update on biomaterials for prevention of epidural adhesion after lumbar laminectomy. J Orthopaedic Trans 13:41–49. https://doi.org/10.1016/j.jot.2018.02.001
Wang W, Du Z, Nimiya Y, Sukamtoh E, Kim D, Zhang G (2016) Allicin inhibits lymphangiogenesis through suppressing activation of vascular endothelial growth factor (VEGF) receptor. J Nutritional Biochem 29:83–89. https://doi.org/10.1016/j.jnutbio.2015.11.004
Sela U, Brill A, Kalchenko V, Dashevsky O, Hershkoviz R (2008) Allicin inhibits blood vessel growth and downregulates Akt phosphorylation and actin polymerization. Nutr Cancer 60(3):412–420. https://doi.org/10.1080/01635580701733083
Wietecha MS, DiPietro LA (2013) Therapeutic approaches to the regulation of wound angiogenesis. Advances in Wound Care 2(3):81–86. https://doi.org/10.1089/wound.2011.0348
Shi P, Cao Y, Gao J, Fu B, Ren J, Ba L, Song C, Qi H, Huang W, Guan X, Sun H (2018) Allicin improves the function of cardiac microvascular endothelial cells by increasing PECAM-1 in rats with cardiac hypertrophy. Phytomedicine 51:241–254. https://doi.org/10.1016/j.phymed.2018.10.021
Staton CA, Stribbling SM, Tazzyman S, Hughes R, Brown NJ, Lewis CE (2004) Current methods for assaying angiogenesis in vitro and in vivo. Int J Exp Pathol 85(5):233–248. https://doi.org/10.1111/j.0959-9673.2004.00396.x
Gruhlke MC, Nicco C, Batteux F, Slusarenko AJ (2016) The Effects of Allicin, a Reactive Sulfur Species from Garlic, on a Selection of Mammalian Cell Lines. Antioxidants 6(1):1. https://doi.org/10.3390/antiox6010001
Chen X, Pang S, Lin J, **a J, Wang Y (2016) Allicin prevents oxidized low-density lipoprotein-induced endothelial cell injury by inhibiting apoptosis and oxidative stress pathway. BMC Complementary and Alternative Medicine 16:133. https://doi.org/10.1186/s12906-016-1126-9
Chen S, Tang Y, Qian Y, Chen R, Zhang L, Wo L, Chai H (2014) Allicin prevents H(2)O(2)-induced apoptosis of HUVECs by inhibiting an oxidative stress pathway. BMC Complementary and Alternative Medicine 14:321. https://doi.org/10.1186/1472-6882-14-321
Wang SL, Liu DS, Liang ES, Gao YH, Cui Y, Liu YZ, Gao W (2015) Protective effect of allicin on high glucose/hypoxia-induced aortic endothelial cells via reduction of oxidative stress. Experimental and Therapeutic Medicine 10(4):1394–1400. https://doi.org/10.3892/etm.2015.2708
Busch C, Jacob C, Anwar A, Burkholz T, Aicha Ba L, Cerella C, Diederich M, Brandt W, Wessjohann L, Montenarh M (2010) Diallylpolysulfides induce growth arrest and apoptosis. Int J Oncol 36(3):743–749. https://doi.org/10.3892/ijo_00000550
Kelkel M, Cerella C, Mack F, Schneider T, Jacob C, Schumacher M, Dicato M, Diederich M (2012) ROS-independent JNK activation and multisite phosphorylation of Bcl-2 link diallyl tetrasulfide-induced mitotic arrest to apoptosis. Carcinogenesis 33(11):2162–2171. https://doi.org/10.1093/carcin/bgs240
Hall PA, Levison DA, Woods AL, Yu CC, Kellock DB, Watkins JA, Barnes DM, Gillett CE, Camplejohn R, Dover R et al (1990) Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: an index of cell proliferation with evidence of deregulated expression in some neoplasms. J Pathol 162(4):285–294. https://doi.org/10.1002/path.1711620403
Prelich G, Tan CK, Kostura M, Mathews MB, So AG, Downey KM, Stillman B (1987) Functional identity of proliferating cell nuclear antigen and a DNA polymerase-delta auxiliary protein. Nature 326(6112):517–520. https://doi.org/10.1038/326517a0
Saeedi Borujeni MJ, Hami J, Haghir H, Rastin M, Sazegar G (2016) Evaluation of Bax and Bcl-2 Proteins Expression in the Rat Hippocampus due to Childhood Febrile Seizure. Iranian Journal of Child Neurology 10(1):53–60
Renault TT, Dejean LM, Manon S (2017) A brewing understanding of the regulation of Bax function by Bcl-xL and Bcl-2. Mech Ageing Dev 161(Pt B):201–210. https://doi.org/10.1016/j.mad.2016.04.007
Kang JW, Lee SM (2012) Melatonin inhibits type 1 interferon signaling of toll-like receptor 4 via heme oxygenase-1 induction in hepatic ischemia/reperfusion. J Pineal Res 53(1):67–76. https://doi.org/10.1111/j.1600-079X.2012.00972.x
Duan W, Yang Y, Yan J, Yu S, Liu J, Zhou J, Zhang J, ** Z, Yi D (2012) The effects of curcumin post-treatment against myocardial ischemia and reperfusion by activation of the JAK2/STAT3 signaling pathway. Basic Res Cardiol 107(3):263. https://doi.org/10.1007/s00395-012-0263-7
Chen H, Zhu B, Zhao L, Liu Y, Zhao F, Feng J, ** Y, Sun J, Geng R, Wei Y (2018) Allicin Inhibits Proliferation and Invasion in Vitro and in Vivo via SHP-1-Mediated STAT3 Signaling in Cholangiocarcinoma. Cell Physiol Biochem 47(2):641–653. https://doi.org/10.1159/000490019
Acknowledgements
This study was funded by Jiangsu Provincial Health and Family Planning Commission (CXTDB2017004)
Funding
This study was funded by Jiangsu Provincial Health and Family Planning Commission (CXTDB2017004).
Author information
Authors and Affiliations
Contributions
H-HS cell culture and experiments, data analysis and statistics, manuscript drafting, the polish of language. X-MF cell culture and experiments, verification of statistics, manuscript revision and format setting. J-CW study design, supervision, experimental technique guidance, the polish of language. JC study design, software and verification of statistics, the polish of language.
Corresponding authors
Ethics declarations
Conflicts of interest
All authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, HH., Feng, XM., Wang, JC. et al. Allicin can suppress the activity of vascular endothelial cells probably by regulating JAK2/STAT3 pathway. Mol Cell Biochem 476, 435–441 (2021). https://doi.org/10.1007/s11010-020-03919-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-020-03919-z