Abstract
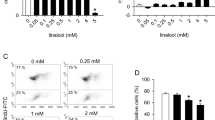
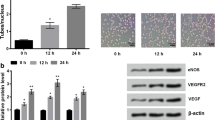
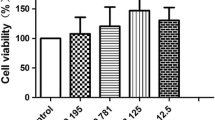
Endothelial cells play essential roles in angiogenesis. Heat shock protein A12B (HSPA12B), a novel member of the multigene Hsp70 family, expresses specifically in endothelial cells. Alpha-lipoic acid (LA) has been used for the treatment of human diabetic complications for more than 20 years. However, little is known whether LA impacts endothelial proliferation and migration. To address these questions, primary human umbilical vein endothelial cells (HUVECs) were isolated and treated with LA. We found that LA reduced viable HUVECs but not caused LDH leakage and nuclear condensation, suggesting an inhibitory effect of LA on HUVEC proliferation. We also noticed that LA impeded wound closure of HUVEC monolayers. The expressions of C-Myc, VEGF, and eNOS and phosphorylation of focal adhesion kinase were reduced by LA. Moreover, LA decreased the expression of heat shock protein A12B (HSPA12B). Notably, overexpression of HSPA12B in endothelial cells prevented the LA-induced loss of VEGF. More importantly, HSPA12B overexpression attenuated the LA-induced inhibition of endothelial proliferation and migration. Collectively, the results demonstrated that LA inhibited proliferative and migratory abilities in human vascular endothelial cells through the downregulation of the HSPA12B/VEGF signaling axis. The data suggest that besides the treatment in diabetic complications, LA might represent a viable therapeutic potential for human diseases that involve high angiogenic activities such as cancers.










Similar content being viewed by others
References
Bretones G, Delgado MD, Leon J (2015) Myc and cell cycle control. Biochim Biophys Acta 1849:506–516. https://doi.org/10.1016/j.bbagrm.2014.03.013
Budhiraja G, Sahu N, Subramanian A (2018) Low-intensity ultrasound upregulates the expression of Cyclin-D1 and promotes cellular proliferation in human mesenchymal stem cells. Biotechnol J 13:e1700382. https://doi.org/10.1002/biot.201700382
Coletta C et al (2015) Regulation of vascular tone, angiogenesis and cellular bioenergetics by the 3-mercaptopyruvate sulfurtransferase/H2S pathway: functional impairment by hyperglycemia and restoration by DL-α-lipoic acid. Mol Med 21:1–14. https://doi.org/10.2119/molmed.2015.00035
Dworacka M, Iskakova S, Krzyzagorska E, Wesolowska A, Kurmambayev Y, Dworacki G (2015) Alpha-lipoic acid modifies circulating angiogenic factors in patients with type 2 diabetes mellitus. Diabetes Res Clin Pract 107:273–279. https://doi.org/10.1016/j.diabres.2014.11.005
Fassina G, Vene R, Morini M, Minghelli S, Benelli R, Noonan DM, Albini A (2004) Mechanisms of inhibition of tumor angiogenesis and vascular tumor growth by epigallocatechin-3-gallate. Clin Cancer Res 10:4865–4873. https://doi.org/10.1158/1078-0432.CCR-03-0672
Gau D, Veon W, Capasso TL, Bottcher R, Shroff S, Roman BL, Roy P (2017) Pharmacological intervention of MKL/SRF signaling by CCG-1423 impedes endothelial cell migration and angiogenesis. Angiogenesis 20:663–672. https://doi.org/10.1007/s10456-017-9560-y
Han Z, Truong QA, Park S, Breslow JL (2003) Two Hsp70 family members expressed in atherosclerotic lesions. Proc Natl Acad Sci U S A 100:1256–1261. https://doi.org/10.1073/pnas.252764399
Hellstrom M, Gerhardt H, Kalen M, Li X, Eriksson U, Wolburg H, Betsholtz C (2001) Lack of pericytes leads to endothelial hyperplasia and abnormal vascular morphogenesis. J Cell Biol 153:543–553
Hu G et al (2006) A novel endothelial-specific heat shock protein HspA12B is required in both zebrafish development and endothelial functions in vitro. J Cell Sci 119:4117–4126. https://doi.org/10.1242/jcs.03179
Kong Q et al (2016) HSPA12B attenuated acute myocardial ischemia/reperfusion injury via maintaining endothelial integrity in a PI3K/Akt/mTOR-dependent mechanism. Sci Rep 6:33636. https://doi.org/10.1038/srep33636
Lee SH, Lee YJ, Song CH, Ahn YK, Han HJ (2010) Role of FAK phosphorylation in hypoxia-induced hMSCS migration: involvement of VEGF as well as MAPKS and eNOS pathways. Am J Phys Cell Phys 298:C847–C856. https://doi.org/10.1152/ajpcell.00418.2009
Lee SG, Lee CG, Yun IH, Hur DY, Yang JW, Kim HW (2012) Effect of lipoic acid on expression of angiogenic factors in diabetic rat retina. Clin Exp Ophthalmol 40:e47–e57. https://doi.org/10.1111/j.1442-9071.2011.02695.x
Li J et al (2013) HSPA12B attenuates cardiac dysfunction and remodelling after myocardial infarction through an eNOS-dependent mechanism. Cardiovasc Res 99:674–684. https://doi.org/10.1093/cvr/cvt139
Luo W et al (2019) C-Myc inhibits myoblast differentiation and promotes myoblast proliferation and muscle fibre hypertrophy by regulating the expression of its target genes, miRNAs and lincRNAs. Cell Death Differ 26:426–442. https://doi.org/10.1038/s41418-018-0129-0
Ma R et al (2010) Dexamethasone attenuated bupivacaine-induced neuron injury in vitro through a threonine-serine protein kinase B-dependent mechanism. Neuroscience 167:329–342. https://doi.org/10.1016/j.neuroscience.2009.12.049
Ma H et al (2015) HSPA12B: a novel facilitator of lung tumor growth. Oncotarget 6:9924–9936. https://doi.org/10.18632/oncotarget.3533
Nebbioso M, Pranno F, Pescosolido N (2013) Lipoic acid in animal models and clinical use in diabetic retinopathy. Expert Opin Pharmacother 14:1829–1838. https://doi.org/10.1517/14656566.2013.813483
Nikitenko L, Boshoff C (2006) Endothelial cells and cancer. Handb Exp Pharmacol 307–334
Obrosova IG et al (2001) Antioxidants attenuate early up regulation of retinal vascular endothelial growth factor in streptozotocin-diabetic rats. Diabetologia 44:1102–1110. https://doi.org/10.1007/s001250100631
Okonkwo UA, DiPietro LA (2017) Diabetes and Wound Angiogenesis. Int J Mol Sci 18. https://doi.org/10.3390/ijms18071419
Ono K et al (2002) Periodate-treated, non-anticoagulant heparin-carrying polystyrene (NAC-HCPS) affects angiogenesis and inhibits subcutaneous induced tumour growth and metastasis to the lung. Br J Cancer 86:1803–1812. https://doi.org/10.1038/sj.bjc.6600307
Parente E, Colannino G, Picconi O, Monastra G (2017) Safety of oral alpha-lipoic acid treatment in pregnant women: a retrospective observational study. Eur Rev Med Pharmacol Sci 21:4219–4227
Pop-Busui R et al (2013) Effects of triple antioxidant therapy on measures of cardiovascular autonomic neuropathy and on myocardial blood flow in type 1 diabetes: a randomised controlled trial. Diabetologia 56:1835–1844. https://doi.org/10.1007/s00125-013-2942-9
Ramos-Garcia P, Gil-Montoya JA, Scully C, Ayen A, Gonzalez-Ruiz L, Navarro-Trivino FJ, Gonzalez-Moles MA (2017) An update on the implications of cyclin D1 in oral carcinogenesis. Oral Dis 23:897–912. https://doi.org/10.1111/odi.12620
Rochette L, Ghibu S, Muresan A, Vergely C (2015) Alpha-lipoic acid: molecular mechanisms and therapeutic potential in diabetes. Can J Physiol Pharmacol 93:1021–1027. https://doi.org/10.1139/cjpp-2014-0353
Saini J, Sharma PK (2018) Clinical, Prognostic and therapeutic significance of heat shock proteins in cancer. Curr Drug Targets 19:1478–1490. https://doi.org/10.2174/1389450118666170823121248
Steiner MK, Syrkina OL, Kolliputi N, Mark EJ, Hales CA, Waxman AB (2009) Interleukin-6 overexpression induces pulmonary hypertension. Circ Res 104:236–244. https://doi.org/10.1161/CIRCRESAHA.108.182014
Steinritz D et al (2010) Effect of N-acetyl cysteine and alpha-linolenic acid on sulfur mustard caused impairment of in vitro endothelial tube formation. Toxicol Sci 118:521–529. https://doi.org/10.1093/toxsci/kfq271
van de Mark K, Chen JS, Steliou K, Perrine SP, Faller DV (2003) Alpha-lipoic acid induces p27Kip-dependent cell cycle arrest in non-transformed cell lines and apoptosis in tumor cell lines. J Cell Physiol 194:325–340. https://doi.org/10.1002/jcp.10205
Vos MJ, Hageman J, Carra S, Kam**a HH (2008) Structural and functional diversities between members of the human HSPB, HSPH, HSPA, and DNAJ chaperone families. Biochemistry 47:7001–7011. https://doi.org/10.1021/bi800639z
Wu J, Li X, Huang L, Jiang S, Tu F, Zhang X, Ma H, Li R, Li C, Li Y, Ding Z, Liu L (2015) HSPA12B inhibits lipopolysaccharide-induced inflammatory response in human umbilical vein endothelial cells. J Cell Mol Med 19:544–554. https://doi.org/10.1111/jcmm.12464
Yao Y et al (2012) Alpha-lipoic acid increases tolerance of cardiomyoblasts to glucose/glucose oxidase-induced injury via ROS-dependent ERK1/2 activation. Biochim Biophys Acta 1823:920–929. https://doi.org/10.1016/j.bbamcr.2012.02.005
Yonenaga Y et al (2005) Absence of smooth muscle actin-positive pericyte coverage of tumor vessels correlates with hematogenous metastasis and prognosis of colorectal cancer patients. Oncology 69:159–166. https://doi.org/10.1159/000087840
Zhao LN et al (2017) MiR-383 inhibits proliferation, migration and angiogenesis of glioma-exposed endothelial cells in vitro via VEGF-mediated FAK and Src signaling pathways. Cell Signal 30:142–153. https://doi.org/10.1016/j.cellsig.2016.09.007
Zhao Y et al (2018) HSPA12B promotes functional recovery after ischaemic stroke through an eNOS-dependent mechanism. J Cell Mol Med 22:2252–2262. https://doi.org/10.1111/jcmm.13507
Ziegler D, Gries FA (1997) Alpha-lipoic acid in the treatment of diabetic peripheral and cardiac autonomic neuropathy. Diabetes 46(Suppl 2):S62–S66
Funding
This work was supported by the National Natural Science Foundation of China (81870234 and 81770854), by Jiangsu Province’s Outstanding Medical Academic Leader program (LJ201124), and a project funded by Collaborative Innovation Center for Cardiovascular Disease Translational Medicine.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The Ethical Board of the First Affiliated Hospital of Nan**g Medical University approved these studies (#2012-SR-153). All the human study procedures were followed in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975, as revised in 2000.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ni, Y., Wang, J., Wang, Z. et al. Alpha-lipoic acid inhibits proliferation and migration of human vascular endothelial cells through downregulating HSPA12B/VEGF signaling axis. Cell Stress and Chaperones 25, 455–466 (2020). https://doi.org/10.1007/s12192-020-01086-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-020-01086-0