Abstract
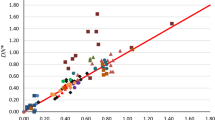
Reichardt’s normalized \(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)\) parameter for solvent polarity has been analyzed in terms of properties of solvent molecules estimated from quantum–mechanical calculations of isolated solvent molecules. The analyses show that \(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)\) has a strong dependence on the partial charge on the most positive hydrogen atom in the solvent molecule, reflecting hydrogen bonding at the pendant oxygen atom of the betaine dye used to define the ET(30) scale. There are smaller, and roughly equal, dependences on the dipole moments and quadrupolar amplitudes of the solvent molecules and an inverse dependence on the solvent polarizability. These three dependences reflect the solvent polarity, that is, the ability to stabilize charge through longer-range interactions. The reason for the inverse dependence on the solvent polarizability is unclear, but a similar dependence was found previously in the analysis of the Kamlet, Abboud and Taft π* scale. The resulting equation for \(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)\) reproduces the experimental values for around 160 solvents, representing most classes of organic solvents, with a standard deviation of around 0.07 {\(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)\) values range from 0 to 1}. The Kamlet and Taft α scale of hydrogen bond donor acidities, which is, in effect, derived from the differences between the \(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)\) and π* values for a solvent, is discussed. The results of the present analyses of \(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)\) and earlier analyses of π* indicate that, while the α values capture the effect of solvent hydrogen bond donor acidity, it also contains residual dependences on other molecular properties. These residual dependences result from the differences in the dependences of the \(E_{{\text{T}}}^{{\text{N}}} \left({30} \right)\) and π* on solvent properties.


Similar content being viewed by others
Notes
The quadrupolar amplitude is calculated as \(A = \sqrt {\sum {q_{ij} q_{ij} } } {\text{ with }}i = (x,y,z){\text{ and }}j = (x,y,z),\) where the qij are the components of the traceless quadrupole.
The differences quoted refer to values calculated using density functional theory and charges derived using the CM5 charge model; those from other calculations and charge models are similar. The differences are given as \(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)_{{{\text{calc}}}} - E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)_{{\exp}}\).
Complex charge distributions, such as those of polyatomic molecules, are commonly represented by a series of superimposed point objects. The first is a point charge (the net charge), which is a scalar quantity, the second is the dipole, which is a vector and the third is the quadrupole, which is a tensor. Just as the dipole has no net charge, the quadrupole has no net moment. The dipole moment and quadrupolar amplitude are used here simply as quantitative measures of the scale of charge centers imbedded in the bulk solvent, the “intensity” of embedded charges. The simplest way to see the necessity for both the dipolar and quadrupolar contributions is to consider CO2, which, despite having partial charges on the O and C atoms, has a zero dipole moment, but a non-zero quadrupolar amplitude.
References
Kamlet, M.J., Taft, R.W.: The solvatochromic comparison method. I. The β-scale of solvent hydrogen-bond acceptor (HBA) basicities. J. Am. Chem. Soc. 98, 377–383 (1976)
Taft, R.W., Kamlet, M.J.: The solvatochromic comparison method 2 The α-scale of solvent hydrogen-bond donor (HBD) acidities. J. Am. Chem. Soc. 98, 2886–2894 (1976)
Kamlet, M.J., Abboud, J.L., Taft, R.W.: The solvatochromic comparison method. 6. The π* scale of solvent polarities. J. Am. Chem. Soc. 99, 6027–6038 (1977)
Taft, R.W., Abboud, J.L., Kamlet, M.J.: Linear solvation energy relationships. 12. The dδ term in thhe solvatochromic equations. J. Am. Chem. Soc. 103, 1080–1086 (1981)
Abraham M.H.: Hydrogen bonding. Part 7. A scale of solute hydrogen-bond acidity based on log k values for complexation in tetrachloromethane. J. Chem. Soc., Perkin Trans. 699–711 (1989).
Abraham M.H.: Hydrogen bonding. Part 10. A scale of solute hydrogen-bond basicity using log k values for complexation in tetrachloromethane. J. Chem. Soc., Perkin Trans. 521–529 (1990)
Abraham M.H.: Scales of solute hydrogen-bonding: their construction and application to physicochemical and biochemical processes. Chem. Soc. Rev. 73–83 (1993)
Dimroth, K., Reichardt, C., Siepmann, T., Bohlmann, F.: Über Pyridinium-N-phenol-Betaine und ihre Verwendung zur Charakterisierung der Polarität von Lösungsmitteln. Justus Liebigs Ann. Chem. 661, 1–37 (1963)
Gutmann, V.: Empirical parameters for donor and acceptor properties of solvents. Electrochim. Acta 21, 661–670 (1976)
Kosower, E.M.: The effect of solvent on spectra. I. A new empirical measure of solvent polarity: Z-values. J. Am. Chem. Soc. 80, 3253–3260 (1958)
Gritzner, G.: The softness parameter (SP), a measure of the soft donor properties of solvents. Z. Phys. Chem. 158, 99–107 (1988)
Marcus, Y.: The properties of organic liquids that are relevant to their use as solvating solvents. Chem. Soc. Rev. 22, 409–416 (1993)
Reichardt, C.: Solvatochromic dyes as solvent polarity indicators. Chem. Rev. 94, 2319–2358 (1994)
Machado, V.G., Stock, R.I., Reichardt, C.: Pyridinium-N-phenolate betaine dyes. Chem. Rev. 114, 10429–10475 (2014)
Katritzky, A.R., Fara, D.C., Yang, H., Tamm, K., Tamm, T., Karelson, M.: Quantitative measures of solvent polarity. Chem. Rev. 104, 175–198 (2004)
Waghorne, W.E., O’Farrell, C.: Solvent basicity, a study of Kamlet-Taft β and Gutmann DN values using computationally derived molecular properties. J. Solution Chem. 47, 1609–1625 (2018)
Waghorne W.E.: A study of the Kamlet–Taft β and π* scales of solvent basicity and polarity using computationally derived molecular properties. J. Solution Chem. (submitted 27 August, 2019)
Devereux, M., Popelier, P.L.A., McLay, I.M.: A refined model for prediction of hydrogen bond acidity and basicity parameters from quantum mechanical descriptors. Phys. Chem. Chem. Phys. 11, 1595–1603 (2009)
Marcus, Y.: Properties of Solvents. Wiley, Chichester, New York (1998)
Reichardt, C., Eschner, M., Schäfer, G.: Synthesis and UV-visible spectroscopic properties of new ‘fluorophilic’ fluorine- and perfluoroalkyl-substituted solvatochromic pyridinium N-phenolate betaine dyes. J. Phys. Org. Chem. 14, 737–751 (2001)
Frisch M.J., Trucks G.W., Schlegel H.B., Scuseria G.E., Robb M.A., Cheeseman J.R., Scalmani G., Barone V., Mennucci, B., Petersson G.A., Nakatsuji H., Caricato M., Li X., Hratchian H.P., Izmaylov A.F., Bloino J., Zheng G., Sonnenberg J.L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J.A., Peralta J.E., Ogliaro F., Bearpark M., Heyd J.J., Brothers E., Kudin K.N., Staroverov V.N., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J.C., Iyengar S.S., Tomasi J., Cossi M., rega N., Millam J.M., Klene M., Knox J.E., Cross J.B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A.J., Cammi R., Pomelli C., Ochterski J.W., Martin E.L., Morokuma K., Zakrzewski V.G., Voth G.A., Salvador P., Dannenberg J.J., Dapprich S., Daniels A.D., Farkas O., Foresman J.B., Ortiz J.V., Cioslowski J., Fox D.J., Gaussian 09 ed., Gaussian, Inc.: Wallingford CT (2009).
Hirshfeld, F.L.: Bonded-atom fragments for describing molecular charge densities. Theor. Chim. Acta 44, 129–138 (1977)
Ritchie, J.P.: Electron density distribution analysis for nitromethane, nitromethide, and nitramide. J. Am. Chem. Soc. 107, 1829–1837 (1985)
Ritchie, J.P., Bachrach, S.M.: Some methods and applications of electron density distribution analysis. J. Comp. Chem. 8, 499–509 (1987)
Foster, J.P., Weinhold, F.: Natural hybrid orbitals. J. Am. Chem. Soc. 102, 7211–7218 (1980)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Waghorne, W.E. A Study of the Reichardt \(E_{{\text{T}}}^{{\text{N}}} \left( {30} \right)\) Parameter using Solvent Molecular Properties Derived from Computation Chemistry and Consideration of the Kamlet and Taft α Scale of Solvent Hydrogen Bond Donor Acidities. J Solution Chem 49, 1360–1372 (2020). https://doi.org/10.1007/s10953-020-01002-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-020-01002-1