Abstract
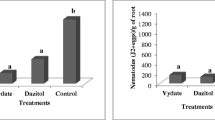
Meloidogyne graminicola is responsible for large yield losses in flooded rice, a problem that is mainly associated with the limited efficacy of nematode management tactics. This study aimed to assess the efficiency of different products applied by seed treatment in controlling M. graminicola in flooded rice grown from polymer-coated and uncoated seeds. Greenhouse experiments were designed to investigate treatment effects on nematode reproduction, root penetration, and plant vegetative parameters, and in vitro assays were used to assess treatment effects on egg hatching and juvenile mortality. Treatments were as follows: Wollastonite (calcium silicate), Agrosilicon (calcium and magnesium silicate), NemOut™ (Bacillus licheniformis + Bacillus subtilis + Trichoderma longibrachiatum), CompostAid® (Lactobacillus plantarum + B. subtilis + Enterococcus faecium), NemaControl® + StimuControl® (Bacillus amyloliquefaciens + Trichoderma harzianum), Nim-I-Go® (Azadirachta indica oil), Avicta 500 FS (abamectin), and CropStar® (thiodicarb + imidacloprid). Inoculated and uninoculated controls were also included. Avicta 500 FS, CropStar®, NemOut™, and CompostAid® were the most effective in reducing nematode penetration and number of nematodes per gram of root. The best in vitro results were obtained with Avicta 500 FS, CropStar®, NemOut™, and CompostAid®. Only Wollastonite showed no in vitro nematicidal activity. Most treatments also improved vegetative growth. It was concluded that treatment of rice seeds with chemical or biological products is effective in controlling M. graminicola.
Similar content being viewed by others
References
Adam, M., Heuer, H., & Hallmann, J. (2014). Bacterial antagonists of fungal pathogens also control root-knot nematodes by induced systemic resistance of tomato plants. PLoS One, 9, 1–8.
Adegbite, A. (2006). Root extracts of plants to control root-knot nematode on edible soybean. Journal of Vegetable Science, 12, 5–12.
Alves, G. C. S., Santos, J. M., Soares, P. L. M., Jesus, F. G., Almeida, E. J., & Thuler, R. T. (2011). Avaliação in vitro do efeito de rizobactérias sobre Meloidogyne incognita. M. javanica e Pratylenchus zeae. Arquivo do Instituto Biológico, 78(4), 557–564.
Amarasinghe, L. D. (2007). Study on approaches to integrated control of Meloidogyne graminicola in rice. Journal of Sciences of the University of Kelaniya, 3, 29–46.
Anter, A. A., Amin, A. W., Ashoub, A. H., & El-Nuby, A. S. (2014). Evaluation of rhizobacteria as resistance inducers or bio-control agents for the control of Meloidogyne incognita in tomato. Pakistan Journal of Nematology, 32(2), 211–221.
Bessi, R., Sujimoto, F. R., & Inomoto, M. M. (2010). Seed treatment affects Meloidogyne incognita penetration, colonization and reproduction on cotton. Ciência Rural, 10(6), 1428–1430.
Boneti, J. I. S., & Ferraz, S. (1981). Modificações do método de Hussey e Barker para extração de ovos de Meloidogyne exigua em raízes de cafeeiro. Fitopatologia Brasileira, 6(3), 553.
Bortolini, G. L., Araujo, D. V., Zavislak, F. D., Junior, J. R., & Krause, W. (2013). Controle de Pratylenchus brachyurus via tratamento de semente de soja. Enciclopédia Biosfera, 9(17), 818–830.
Bouman, B. A. M., Barker, R., Humphreys, E., Tuong, T. P., Atlin, G. N., Bennett, J., et al. (2007). Rice: Feeding the billions. In the comprehensive assessment of water in agriculture. IRRI, 1, 1–40.
Bouman, B. A. M., Humphreys, E., Tuong, T. P., & Barker, R. (2006). Rice and water. Advances in Agronomy, 92, 187–237.
Brodeur, J. (2012). Host specificity in biological control: Insights from opportunistic pathogens. Evolutionary Applications, 5(5), 470–480.
Byrd Jr., D. W., Kirkpatrick, J., & Barker, K. R. (1983). An improved technique for clearing and staining plant tissues for detection of nematodes. Journal of Nematology, 15(1), 142–143.
Cabrera, J. A., Kiewnick, S., Grimm, C., Dababat, A. A., & Sikora, R. A. (2009). Effective concentration and range of activity of abamectin as seed treatment against root-knot nematodes in tomato under glasshouse conditions. Nematology, 11, 909–915.
Carneiro, R. M. D. G., & Almeida, M. R. A. (2001). Técnica de eletroforese usada no estudo de enzimas dos nematoides de galhas para identificação de espécies. Nematologia Brasileira, 25, 555–560.
Chen, S. Y., Dickson, D. W., & Mitchell, D. J. (2000). Viability of Heterodera glycines exposed to fungal filtrates. Journal of Nematology, 32, 190–197.
Conab-Companhia Nacional de Abastecimento. (2018). Perspectivas para a agropecuária/ Companhia Nacional de Abastecimento, 4, 19–30.
Dongre, M., & Simon, S. (2013). Efficacy of certain botanical extracts in the management of Meloidogyne graminicola of rice. International Journal of Agricultural Science and Research, 3(3), 91–98.
Dutta, T. K., Ganguly, A. K., & Gaur, H. S. (2012). Global status of rice root-knot nematode, Meloidogyne graminicola. African Journal of Microbiology Research, 6(31), 6016–6021.
Ferreira, D. F. (2008). SISVAR: um programa para análises e ensino de estatística. Revista Symposium, 6, 36–41.
Gravanis, F. T., Vagelas, L., Leontopoulos, S., & Natsiopoulos, D. (2011). Nematicidal effects of Azadirachta indica seed extract on Meloidogyne spp. Journal of Agricultural Science and Technology, 1, 136–141.
Grisp - Global Rice Science Partnership. (2013). Rice almanac, 4th edition. Los Baños (Philippines). International Rice Research Institute, 283, 11–14.
Harman, G. E., Howell, C. R., Viterbo, A., Chet, I., & Lorito, M. (2004). Trichoderma species-opportunistic, avirulent plant symbionts. Nature reviews-Microbiology, 2(1), 43–56.
Hussey, R. S., & Baker, K. R. (1973). A comparison of methods of collecting inocula of Meloidogyne species, including a new technique. Plant Disease Report, 57, 1025–1028.
Jain, R. K., Khan, M. R., & Kumar, V. (2012). Rice root-knot nematode (Meloidogyne graminicola) infestation in rice. Archives of Phytopathology and Plant Protection, 45(6), 635–645.
Jansson, RK., Dybas, RA. (1998). Avermectins: Biochemical mode of action, biological activity and agricultural importance. In: ISHAAYA, I.; DEGHEELE, D. insecticides with novel modes of action: Mechanisms and application, Springer, 152-167.
Javeed, M. T., Alhazmi, A., & Molan, Y. Y. (2016). Antagonistic effects of some indigenous isolates of Trichoderma spp. against Meloidogyne javanica. Pakistan Journal of Nematology, 34(2), 183–191.
Joo, SB., Kumar, VJR., Ahmad, RI., Kim, BC., Park, W., Park, SD.; Se-Eun Kim,, et. al., (2012). Bacterial mixture from greenhouse soil as a biocontrol agent against root-knot nematode, Meloidogyne incognita, on oriental melon. Journal of Microbiology and Biotechnology, 22(1):114–117.
Khalil, M. S. (2013). Abamectin and azadirachtin as eco-friendly promising biorational tools in integrated nematodes management programs. Journal Plant Pathology Microbiology, 4, 4–174.
Khalil, M. S. H., Alamm, A. F. G., & Barakat, A. S. T. (2012). Nematicidal activity of some biopesticide agents and microorganisms against root-knot nematode on tomato plants under greenhouse conditions. Journal of Plant Protection Research, 52, 47–52.
Khalili, E., Sadravi, M., Naeimi, S., & Khosravi, V. (2012). Biological control of rice brown spot with native isolates of three Trichoderma species. Brazilian Journal of Microbiology, 43(1), 297–305.
Khan, M. R., & Anwer, A. (2011). Occurrence of rice root-knot nematode and yield loss assessment in Aligarh and Hathras districts of Uttar Pradesh, India. Indian Journal of Nematology, 41(1), 34–40.
Khan, M. R., Solanki, R. D., Bohra, B., & Vyas, B. N. (2012a). Evaluation of Achook (Azadirachtin 1500 ppm) against root knot nematode (Meloidogyne incognita) infecting okra. South Asian Journal of Experimental Biology, 2, 149–156.
Khan, M. R., Zaidi, B., & Haque, Z. (2012b). Nematicides control rice root-knot, caused by Meloidogyne graminicola. Phytopathologia Mediterranea, 51(2), 298–306.
Kubo, R. K., Machado, A. C. Z., & Oliveira, C. M. G. (2012). Efeito do tratamento de semente no controle de Rotylenchulus reniformis em dois cultivares de algodão. Arquivo do Instituto Biologico, 79(2), 239–245.
Le, H. T. T., Padghamb, J. L., & Sikora, R. A. (2009). Biological control of the rice root-knot nematode Meloidogyne graminicola on rice, using endophytic and rhizosphere fungi. International Journal of Pest Management, 55(1), 31–36.
Lian, L. H., Tian, B. Y., **ong, R., Zhu, M. Z., Xu, J., & Zhang, K. Q. (2007). Proteases from Bacillus: A new insight into the mechanism of action for rhizobacterial suppression of nematode populations. Letters in Applied Microbiology, 45, 262–269.
Ludwig, J., Moura, A. B., & Gomes, C. B. (2013). Potencial da microbiolização de sementes de arroz com rizobactérias para o biocontrole do nematoide das galhas. Tropical Plant Pathology, 38, 1–5.
MAPA (2020) Ministério da Agricultura, Pecuária e Abastecimento. Available at <http://extranet.agricultura.gov.br/agrofit_cons/principal_agrofit_cons>. Accessed 19 Nov 2020.
Mattei, D., & Dias-Arieira, C. R. (2015). Different sources of silicon in the embryonic development and in the hatching of Meloidogyne javanica. African Journal of Agricultural Research, 10(52), 4814–4819.
Mattei, D., Dias-Arieira, C. R., Lopes, A. P. M., & Miamoto, A. (2017). Influence of Rocksil, Silifort and Wollastonite on penetration and development of Meloidogyne javanica in Poaceae and Fabaceae. Journal of Phytopathology, 165, 91–97.
Mohanty, K. C., Mahapatra, S. N., & Swain, S. C. (2000). Efficacy of certain chemicals as seed treatment against Meloidogyne graminicola on rice. Indian Journal Nematology, 30(2), 225–264.
Ntalli, N., Oplos, C., Michailidis, M., Thanasenaris, A., Kontea, D., Caboni, P., Tsiropoulos, N. T., Menkissoglu-Spiroudi, U., & Adamski, Z. (2016). Strong synergistic activity and egg hatch inhibition by (E,E)-2,4-decadienal and (E)-2-decenal in Meloidogyne species. Journal of Pest Science, 89, 565–579.
Oliveira, D. F., Santos-Junior, H., Nunes, A. S., Campos, V. P., Pinho, R. S. C., & Gajo, G. C. (2014). Purification and identification of metabolites produced by Bacillus cereus and B. subtilis active against Meloidogyne exigua, and their in silico interaction with a putative phosphoribosyl transferase from M. incognita. Anais da Academia Brasileira de Ciências, 86, 525–538.
Opperman, C. H., & Chang, S. (1990). Plant-parasitic nematode acetylcholinesterase inhibition by carbamate and organophosphate nematicides. Journal of Nematology, 22, 481–488.
Padgham, J. L., & Sikora, R. A. (2007). Biological control potential and modes of action of Bacillus megaterium against Meloidogyne graminicola on rice. Crop Protection, 26, 971–977.
Pankaj, K., Bansal, R. K., & Nandal, S. N. (2010). Biocontrol of Meloidogyne graminicola using rhizobacteria on rice seedlings. Nematologia Mediterranea, 38, 115–119.
Pozza, A. A. A., Alves, E., Pozza, E. A., Carvalho, J. G., Montanari, M., Guimarães, P. T. G., et al. (2004). O silício no controle de cercosporiose em três variedades de café. Fitopatologia Brasileira, 29, 373–402.
Prasad, J. S., Vishakanta, & Gubbaiah. (2006). Outbreak of root-knot nematode (Meloidogyne graminicola) disease in rice and farmers perceptions. Indian Journal of Nematology, 36(1), 85–88.
Priya, S. (2015). Biomanagement of rice root knot nematode, Meloidogyne graminicola Golden and Brichfield in aerobic rice. International Journal in Management and Social Science, 3, 591–598.
Putter, I., Connell, J. G. M., Preiser, F. A., Haidri, A. A., Ristich, S. S., & Dybas, R. A. (1981). Avermectins: Novel insecticides, acaricides and nematicides from a soil microorganism. Experientia, 37, 963–964.
Qiao, K., Liu, X., Wang, H., **a, X., Ji, X., & Wang, K. (2012). Effect of abamectin on root-knot nematodes and tomato yield. Pest Management Science, 68(6), 853–857.
Ribeiro, L. M., Campos, H. D., Ribeiro, G. C., Neves, D. L., & Dias-Arieira, C. R. (2012). Effect of treating cotton seeds on the population dynamics of Pratylenchus brachyurus under water stress conditions. Nematropica, 42(1), 84–90.
Santana, M. V., Ferreira, B. S., Silva, J. O., Freire, L. L., Barcellos, L. C., & Rocha, M. R. (2016). Modos de aplicação de nematicidas para o controle do nematoide-das-galhas Meloidogyne javanica em soja. Multi-Science Journal, 1(4), 114–118.
Sharma, R. D. (2001). Pathogenicity and reproduction of Meloidogyne javanica on yellow passion fruit hibrids. Nematologia Brasileira, 25, 247–249.
Sharon, E., Chet, I., & Spiegel, Y. (2011). Trichoderma as a biological control agent. Biological Control, 11, 183–201.
Siahpoush, S. (2011). Change of some defense compounds of cucumber treated with Bacillus cereus and salicylic acid against Meloidogyne javanica. African Journal of Plant Science, 5, 829–834.
Silva, R. V., Oliveira, R. D. L., Nascimento, K. J. T., & Rodrigues, F. A. (2010). Biochemical responses of coffee resistance against Meloidogyne exigua mediated by silicon. Plant Pathology, 59, 586–593.
Soriano, I. R. S., & Reversat, G. (2003). Management of Meloidogyne graminicola and yield of upland rice in South-Luzon, Philippines. Nematology, 5, 879–884.
Spiegel, Y., Sharon, E., & Chet, I. (2005). Mechanisms and improved biocontrol of the root-knot nematodes by Trichoderma spp. Acta Horticulturae, 698, 225–228.
Steffen, R. B., Antoniolli, Z. I., Steffen, G. P. K., Jacques, R. J. S., & Echkardt, D. P. (2011). Efeito da abamectina e carbofuran no controle de danos causados por Meloidogyne graminicola em plantas de arroz irrigado. Revista da FZVA, 18(2), 56–69.
Tian, B., Yang, J., & Zhang, K. Q. (2007). Bacteria used in the biological control of plant-parasitic nematodes: Populations, mechanisms of action, and future prospects. FEMS Microbiology Ecology, 61, 197–213.
Tunes, L. V. M., Fonseca, D. A. R., Meneghello, G. E., Reis, B. B., Brasil, V. D., Rufino, C. A., et al. (2014). Qualidade fisiológica, sanitária e enzimática de sementes de arroz irrigado recobertas com silício. Revista Ceres, 61(5), 675–685.
Zheng, Z., Zheng, J., Zhang, Z., Donghai, P., & Sun, M. (2016). Nematicidal spore-forming bacilli share similar virulence factors and mechanisms. Scientific Reports, 6, 1–9.
Acknowledgements
The authors thank the Brazilian National Council for Scientific and Technological Development (CNPq) for providing a doctoral scholarship to MRCS and a research productivity grant to CRDA. We also thank the Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES) for the scholarship awarded to APML.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Soares, M.R.C., Dias-Arieira, C.R. & Lopes, A.P.M. Seed treatments for control of Meloidogyne graminicola in flooded rice. Eur J Plant Pathol 160, 901–915 (2021). https://doi.org/10.1007/s10658-021-02294-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-021-02294-9