Abstract
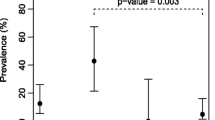
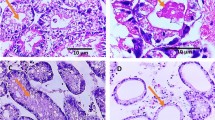
Cryptosporidium spp. has been recognized as an important pathogen. As bivalve mollusks are noted as potential sources of several pathogens due to their consumption as foodstuffs, the aim of this study was to investigate the occurrence of Cryptosporidium spp. oocysts in Perna perna mussels and in seawater samples from a mussel farm in Southeastern Brazil, where mussels are grown directly in the sea, attached to ropes. Oocysts were observed by microscopy and confirmed by an enzyme-linked immunosorbent assay. Oocysts were present in mussel gills and GI tracts, as well as in the seawater. Of the 100 females, 10% and 11% showed contaminated GI tracts and gills, respectively, while this rate was lower in males, at 5% and 8.9%. Oocysts were present in higher amounts in the GI tract compared to gills and water. Contamination of the study area is apparent, leading to public health risks. More in-depth studies are needed, including molecular investigations, to identify Cryptosporidium species in mussels, as well as the implementation of monitoring actions in animals destined for human consumption.

Similar content being viewed by others
Change history
14 February 2018
The original version of this article unfortunately contained a mistake.
References
Cacciò S, Pinter E, Fantini R, Mezzaroma I, Pozio E (2002) Human infection with Cryptosporidium felis: case report and literature review. Emerg Infect Dis 8:85–86
Cardoso TSF, Santos IRS, Berto BP, Cardoso SV, Mesquita EFM, Lopes CWG (2012) Correlation between biometric and parasitologic data of mussels (Perna perna) from Ponta do Tinguí, Mangaratiba, RJ. Brazil Revista Brasileira de Medicina Veterinária 34:9–13
Choi N, Kfouri C, Kweon HJ, You EC, Chagnon FJF, Harleman DRF (2003) Potable water and wastewater infrastructure planning for the city of Paraty, Brazil Global Water at MIT 264
Coelho C, Heinert AP, Simões CM, Barardi CR (2003) Hepatitis A virus detection in oysters (Crassostrea gigas) in Santa Catarina State, Brazil, by reverse transcription-polymerase chain reaction. J Food Prot 66:507–511
Da Fonseca IP, Ramos PS, Ruano FA, Duarte AP, Costa JC, Almeida AC, Falca ML, Fazendeiro MI (2006) Efficacy of commercial cleansing procedures in eliminating Cryptosporidium parvum oocysts from bivalves. J Eukaryot Microbiol 53:S49-S51
Fayer R, Speer CA, Dubey JP (1997) The general biology of Cryptosporidium. Cryptosporidium and Cryptosporidiosis. FL CRC Press, Boca Raton, pp 1–42
Fayer R, Graczyk TK, Lewis EJ, Trout JM, Farley CA (1998) Survival of infectious Cryptosporidium parvum oocysts in seawater and eastern oysters (Crassostrea virginica) in the Chesapeake Bay. Appl Environ Microbiol 64:1070–1074
Fayer R, Morgan U, Upton SJ (2000) Epidemiology of Cryptosporidium: transmission, detection and identification International. J Parasitol 30:1305–1322
Fayer R, Trout JM, Lewis EJ, Santin M, Zhou L, Lal AA, **ao L (2003) Contamination of Atlantic coast commercial shell-fish with Cryptosporidium. Parasitol Res 89:141–145
Fayer R, Dubey JP, Lindsay DS (2004) Zoonotic protozoa: from land to sea. Trends Parasitol 20:531–536
Freire-Santos F, Oteiza-López AM, Vergara-Castiblanco CA, Ares-Mazás ME (2000) Study of the combined influence of environmental factors on viability of Cryptosporidium parvum oocysts in water evaluated by fluorogenic vital dyes and excystation techniques. Vet Parasitol 89:253–259
Girdwood RWA, Smith HV (1999) Cryptosporidium. In: Encyclopaedia of food microbiology. London Academic Press, pp 487–497
Giangaspero A, Molini U, Iorio R, Traversa D, Paoletti B, Giansante C (2005) Cryptosporidium parvum oocysts in sea-water clams (Chameleagallina) in Italy. Prev Vet Med 69:203–312
Gomez-Bautista M, Ortega-Mora LM, Tabares E, Lopez-Rodas V, Costas E (2000) Detection of infectious Cryptosporidium parvum oocysts in mussels (Mytilus galloprovincialis) and cockles (Cerastoderma edule). Appl Environ Microbiol 66:1866–1870
Gómez-Couso H, Freire-Santos F, Martínez-Urtaza J, Ares-Mazás ME (2003) Contamination of bivalve molluscs by Cryptosporidium oocysts: the need for new quality control standards. Int J Food Microbiol 87:97–105
Graczyk TK, Schwab KJ (2000) Food-borne infections vectored by molluscan shellfish. Curr Gastroenterol Rep 2:305–309
Graczyk TK, Fayer R, Lewis EJ, Trout JM, Farley CA (1999) Cryptosporidium oocysts in Bent mussels (Ischadium recurvum) in the Chesapeake Bay. Parasitol Res 85:518–521
Graczyk TK, Marcogliese DJ, Delafontaine Y, Dasilva AJ, Mhagami-Ruwende B, Pieniazek NJ (2001) Cryptosporidium parvum oocysts in zebra mussels (Dreissena polymorpha): evidence from the St Lawrence River. Parasitol Res 87:231–234
Graczyk TK, Conn DB, Lucy F, Minchin D, Tamang L, Moura L, Da Silva A (2004) Human waterborne parasites in zebra mussels (Dreissena polymorpha) from the Shannon river drainage area, Ireland. Parasitol Res 93:385–391
Griffiths JK (1998) Human cryptosporidiosis: epidemiology, transmission, treatment and diagnosis. Adv Parasitol 40:37–85
Henriksen SA, Pohlenz JF (1981) Staining of Cryptosporidia by a modified Ziehl-Neelsen technique. Acta Vet Scand 22:594–596
Jenkins MC, Trout JM, Abrahamsen MS, Lancto CA, Higgins J, Fayer R (2000) Estimating viability of Cryptosporidium parvum oocysts using reverse transcriptase-polymerase chain reaction (RT-PCR) directed at mRNA encoding amyloglucosidase. J Microbiol Methods 43:97–106
Leal DAG, Pereira MA, Franco RMB, Branco N, Cantusio-Neto R (2008) First report of Cryptosporidium spp. oocysts in oysters (Crassostrea rhizophorae) and cockles (Tivela mactroides) in Brazil. Braz J Water Health 6:527–532
Lee JR, Younger AD (2002) Develo** microbiological risk assessment for shellfish purification. Int Biodeterior Biodegrad 50:177–183
Lee CY, Panicker G, Bej AK (2003) Detection of pathogenic bacteria in shellfish using multiplex PCR followed by CovaLinkTM NH microwell plate sandwich hybridization. J Microbiol Methods 53:199–209
Li X, Guyot K, Dei-Cas E, Mallard JP, Ballet JJ, Brasseur P (2006) Cryptosporidium oocysts in mussels (Mytilus edulis) from Normandy (France). Int J Food Microbiol 108:321–325
Lowery CJ, Nugent P, Moore JE, Millar BC, ** of Cryptosporidium parvum recovered from a recreational river source and an associated mussel (Mytilus edulis) bed in Northern Ireland. Epidemiol Infect 127:545–553
MacRae M, Hamilton C, Strachan NJC, Wright S, Ogden ID (2005) The detection of Cryptosporidium parvum and Escherichia coli O157 in UK bivalve shellfish. J Microbiol Methods 60:395–401
McOliver CC (2009) Recreational water contact as a risk factor for Cryptosporidium exposure among persons with HIV/AIDS in Baltimore, MD. The Johns Hopkins University, Baltimore, p 234
Meireles MV (2010) Cryptosporidium infection in Brazil: implications for veterinary medicine and public health Revista. Brasileira de Parasitologia Veterinária 19:197–204
Melo PC, Teodisio J, Reis J, Duarte A, Costa JC, Fonseca IP (2006) Cryptosporidium spp. in freshwater bivalves in Portugal. J Eukaryot Microbiol 53:S28-S29
National Aquaculture Sector Overview (2004) Brazil. National Aquaculture Sector Overview Fact Sheets. Text by Suplicy, F.M. In: FAO Fisheries and Aquaculture Department [online]. Rome
Oliveira GFM, Lima MF, Bomfim TCB (2014) Perna perna mussels as bioindicators of aquatic contamination by Cryptosporidium spp. Acta Scientiae Veterinariae 42:1–6
Pedraza-Díaz S, Amar C, McLauchlin J (2000) The identification and characterization of an unusual genotype of Cryptosporidium from human faeces as Cryptosporidium meleagridis. FEMS Microbiol Lett 189:189–194
Pedraza-Díaz S, Amar C, Iversen AM, Stanley PJ, McLauchlin J (2001) Unusual Cryptosporidium species recovered from human faeces: first description of Cryptosporidium felis and Cryptosporidium ‘‘dog type’’ from patients in England. J Med Microbiol 50:293–296
Ritchie LS (1948) An ether sedimentation technique for routine stool examination. Bull US Army Med Dep 8:326–334
Robertson LJ, Gjerde B (2000) Isolation and enumeration of Giardia cysts, Cryptosporidium oocysts, and Ascaris eggs from fruits and vegetables. J Food Prot 63:775–877
Rose JB (1997) Environmental ecology of Cryptosporidium and public health implications. Annu Rev Public Health 18:135–161
Schets FM, Harold HJM, Berg VD, Engels GB, Lodder WJ, Husman AMR (2007) Cryptosporidium and Giardia in commercial and non-commercial oysters (Crassostrea gigas) and water from the Oosterschelde, The Netherlands. Int J Food Microbiol 113:189–194
Skinner LF, Barboza DF, Rocha RM (2016) Rapid assessment survey of introduced ascidians in a region with many marinas in the Southwest Atlantic Ocean, Brazil. Manag Biol Invasions 7:13–20
Smith HV, Rose JB (1998) Waterborne cryptosporidiosis: current status. Parasitol Today 14:14–22
Smith HV, McDiarmid A, Smith AL, Hinson AR, Gilmour RA (1989) An analysis of staining methods for the detection of Cryptosporidium spp. oocysts in water-related samples. Parasitol Res 3:323–327
Solo-Gabriele H, Neumeister M (1996) U.S. outbreaks of Cryptosporidiosis. J Am Water Works Assoc 88:76–86
Srisuphanunt M, Saksirisampant W, Karanis P (2009) Detection of cryptosporidium oocysts in green mussels (Perna viridis) from shell-fish markets of Thailand. Parasite 16:235–239
Suplicy FM (2008) Legal aspects and governmental actions for the development of mollusc farming in Brazil. In: FAO Actas de Pesca y Acuicultura, Rome FAO, pp 205–208
Ugwoke EV, Umoh JU, Okolocha EC, Lawai IA (2013) Cryptosporidium oocysts in Anodonta sp. (bivalve mollusc) as indicators of pollution of Tiga Lake ecosystem in Kano State, Nigeria. J Parasitol Vector Biol 5:77–82
Author information
Authors and Affiliations
Corresponding author
Additional information
A correction to this article is available online at https://doi.org/10.1007/s00128-018-2284-x.
Rights and permissions
About this article
Cite this article
Santos, A.L., de Oliveira, L.T.F., Souza, A.L.A. et al. Cryptosporidium spp. Contamination in Perna perna Mussels Destined for Human Consumption in Southeastern Rio de Janeiro, Brazil. Bull Environ Contam Toxicol 100, 240–244 (2018). https://doi.org/10.1007/s00128-017-2223-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-017-2223-2