Abstract
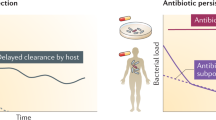
Antibiotic resistance is a massive problem in today’s world and a serious threat to human civilization. Apart from being genetically resistant to antibiotics, the other important mechanism by which pathogenic bacteria can evade antibiotics is multidrug tolerance through the formation of persisters. In contrast to antibiotic-resistant bacteria, persister cells require no heritable genetic changes for surviving antibiotic treatment but become drug tolerant due to transient growth arrest. Since most antibiotics only target active metabolic pathways in the growing cells, non-growing persisters can escape the bactericidal effects of antibiotics and resume growth once the antibiotic is withdrawn resulting in treatment failure. Here in this book chapter, we try to highlight different aspects of persisters starting from unique characteristics of persisters, mechanisms of persister formation, clinical relevance, different techniques to study persisters, and some promising treatment options. Finally, we emphasize how focused research in this particular field can have a big impact to eradicate antimicrobial resistance.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Adams KN et al (2011) Drug tolerance in replicating mycobacteria mediated by a macrophage-induced efflux mechanism. Cell 145:39–53
Allison KR, Brynildsen MP, Collins JJ (2011a) Heterogeneous bacterial persisters and engineering approaches to eliminate them. Curr Opin Microbiol 14:593–598
Allison KR, Brynildsen MP, Collins JJ (2011b) Metabolite-enabled eradication of bacterial persisters by aminoglycosides. Nature 473:216–220
Al-Seraih A et al (2017) Enterocin B3A-B3B produced by LAB collected from infant faeces: potential utilization in the food industry for Listeria monocytogenes biofilm management. Antonie Van Leeuwenhoek 110:205–219
Amato SM, Orman MA, Brynildsen MP (2013) Metabolic control of persister formation in Escherichia coli. Mol Cell 50:475–487
Amison RT et al (2020) The small quinolone derived compound HT61 enhances the effect of tobramycin against Pseudomonas aeruginosa in vitro and in vivo. Pulm Pharmacol Ther 61:101884
Arnion H et al (2017) Mechanistic insights into type I toxin antitoxin systems in Helicobacter pylori: the importance of mRNA folding in controlling toxin expression. Nucleic Acids Res 45:4782–4795
Balaban NQ, Merrin J, Chait R, Kowalik L, Leibler S (2004) Bacterial persistence as a phenotypic switch. Science 305:1622–1625
Balaban NQ et al (2019) Definitions and guidelines for research on antibiotic persistence. Nat Rev Microbiol 17:441–448
Barak Z, Gallant J, Lindsley D, Kwieciszewki B, Heidel D (1996) Enhanced ribosome frameshifting in stationary phase cells. J Mol Biol 263:140–148
Barraud N, Buson A, Jarolimek W, Rice SA (2013) Mannitol enhances antibiotic sensitivity of persister bacteria in Pseudomonas aeruginosa biofilms. PLoS One 8:e84220
Battesti A, Bouveret E (2006) Acyl carrier protein/SpoT interaction, the switch linking SpoT-dependent stress response to fatty acid metabolism. Mol Microbiol 62:1048–1063
Beaber JW, Hochhut B, Waldor MK (2004) SOS response promotes horizontal dissemination of antibiotic resistance genes. Nature 427:72–74
Berghoff BA, Hoekzema M, Aulbach L, Wagner EG (2017) Two regulatory RNA elements affect TisB-dependent depolarization and persister formation. Mol Microbiol 103:1020–1033
Bigger J (1944) Treatment of Staphylococcal infections with penicillin by intermittent sterilisation. The Lancet 244:497–500
Bougdour A, Cunning C, Baptiste PJ, Elliott T, Gottesman S (2008) Multiple pathways for regulation of sigmaS (RpoS) stability in Escherichia coli via the action of multiple anti-adaptors. Mol Microbiol 68:298–313
Boutte CC, Crosson S (2013) Bacterial lifestyle shapes stringent response activation. Trends Microbiol 21:174–180
Brauner A, Fridman O, Gefen O, Balaban NQ (2016) Distinguishing between resistance, tolerance and persistence to antibiotic treatment. Nat Rev Microbiol 14:320–330
Brauner A, Shoresh N, Fridman O, Balaban NQ (2017) An experimental framework for quantifying bacterial tolerance. Biophys J 112:2664–2671
Brileya KA, Camilleri LB, Fields MW (2014) 3D-fluorescence in situ hybridization of intact, anaerobic biofilm. Methods Mol Biol 1151:189–197
Bryk R et al (2008) Selective killing of nonreplicating mycobacteria. Cell Host Microbe 3:137–145
Butt A et al (2014) The HicA toxin from Burkholderia pseudomallei has a role in persister cell formation. Biochem J 459:333–344
Cabral DJ, Wurster JI, Belenky P (2018) Antibiotic persistence as a metabolic adaptation: stress, metabolism, the host, and new directions. Pharmaceuticals (Basel) 11(1):14
Cameron DR, Shan Y, Zalis EA, Isabella V, Lewis K (2018) A genetic determinant of Persister cell formation in bacterial pathogens. J Bacteriol 200(17):e00303–e00318
Chan K et al (2002) Complex pattern of Mycobacterium marinum gene expression during long-term granulomatous infection. Proc Natl Acad Sci U S A 99:3920–3925
Chen X, Zhang M, Zhou C, Kallenbach NR, Ren D (2011) Control of bacterial persister cells by Trp/Arg-containing antimicrobial peptides. Appl Environ Microbiol 77:4878–4885
Cheverton AM et al (2016) A Salmonella toxin promotes Persister formation through acetylation of tRNA. Mol Cell 63:86–96
Chowdhury N, Wood TL, Martinez-Vazquez M, Garcia-Contreras R, Wood TK (2016) DNA-crosslinker cisplatin eradicates bacterial persister cells. Biotechnol Bioeng 113:1984–1992
Christensen LD et al (2007) Impact of Pseudomonas aeruginosa quorum sensing on biofilm persistence in an in vivo intraperitoneal foreign-body infection model. Microbiology 153:2312–2320
Chuang YM et al (2015) Deficiency of the novel exopolyphosphatase Rv1026/PPX2 leads to metabolic downshift and altered cell wall permeability in Mycobacterium tuberculosis. mBio 6:e02428
Chubukov V, Sauer U (2014) Environmental dependence of stationary-phase metabolism in Bacillus subtilis and Escherichia coli. Appl Environ Microbiol 80:2901–2909
Claudi B et al (2014) Phenotypic variation of Salmonella in host tissues delays eradication by antimicrobial chemotherapy. Cell 158:722–733
Cohen NR, Lobritz MA, Collins JJ (2013) Microbial persistence and the road to drug resistance. Cell Host Microbe 13:632–642
Conlon BP et al (2013) Activated ClpP kills persisters and eradicates a chronic biofilm infection. Nature 503:365–370
Conlon BP et al (2016) Persister formation in Staphylococcus aureus is associated with ATP depletion. Nat Microbiol 1:16051
Corona F, Martinez JL (2013) Phenotypic resistance to antibiotics. Antibiotics 2:237–255
de Breij A et al (2018) The antimicrobial peptide SAAP-148 combats drug-resistant bacteria and biofilms. Sci Transl Med 10(423):eaan4044
De Groote VN et al (2009) Novel persistence genes in Pseudomonas aeruginosa identified by high-throughput screening. FEMS Microbiol Lett 297:73–79
de Jong IG, Haccou P, Kuipers OP (2011) Bet hedging or not? a guide to proper classification of microbial survival strategies. Bioessays 33:215–223
De Leenheer P, Cogan NG (2009) Failure of antibiotic treatment in microbial populations. J Math Biol 59:563–579
de Leseleuc L, Harris G, KuoLee R, Chen W (2012) In vitro and in vivo biological activities of iron chelators and gallium nitrate against Acinetobacter baumannii. Antimicrob Agents Chemother 56:5397–5400
Defraine V et al (2016) Efficacy of Artilysin Art-175 against resistant and persistent Acinetobacter baumannii. Antimicrob Agents Chemother 60:3480–3488
DeLeon K et al (2009) Gallium maltolate treatment eradicates Pseudomonas aeruginosa infection in thermally injured mice. Antimicrob Agents Chemother 53:1331–1337
Deng W et al (2020) L-lysine potentiates aminoglycosides against Acinetobacter baumannii via regulation of proton motive force and antibiotics uptake. Emerg Microbes Infect 9:639–650
Dhar N, McKinney JD (2010) Mycobacterium tuberculosis persistence mutants identified by screening in isoniazid-treated mice. Proc Natl Acad Sci U S A 107:12275–12280
Diard M et al (2017) Inflammation boosts bacteriophage transfer between Salmonella spp. Science 355:1211–1215
Dorr T, Lewis K, Vulic M (2009) SOS response induces persistence to fluoroquinolones in Escherichia coli. PLoS Genet 5:e1000760
Dorr T, Vulic M, Lewis K (2010) Ciprofloxacin causes persister formation by inducing the TisB toxin in Escherichia coli. PLoS Biol 8:e1000317
Duan X et al (2016) l-Serine potentiates fluoroquinolone activity against Escherichia coli by enhancing endogenous reactive oxygen species production. J Antimicrob Chemother 71:2192–2199
Eisenreich W, Dandekar T, Heesemann J, Goebel W (2010) Carbon metabolism of intracellular bacterial pathogens and possible links to virulence. Nat Rev Microbiol 8:401–412
Ethapa T et al (2013) Multiple factors modulate biofilm formation by the anaerobic pathogen Clostridium difficile. J Bacteriol 195:545–555
Fauvart M, De Groote VN, Michiels J (2011) Role of persister cells in chronic infections: clinical relevance and perspectives on anti-persister therapies. J Med Microbiol 60:699–709
Feng J, Zhang S, Shi W, Zhang Y (2016) Ceftriaxone pulse dosing fails to eradicate biofilm-like microcolony B. burgdorferi persisters which are sterilized by daptomycin/doxycycline/cefuroxime without pulse dosing. Front Microbiol 7:1744
Ford CB et al (2011) Use of whole genome sequencing to estimate the mutation rate of Mycobacterium tuberculosis during latent infection. Nat Genet 43:482–486
Foster TJ (2005) Immune evasion by staphylococci. Nat Rev Microbiol 3:948–958
Fridman O, Goldberg A, Ronin I, Shoresh N, Balaban NQ (2014) Optimization of lag time underlies antibiotic tolerance in evolved bacterial populations. Nature 513:418–421
Fung DK, Chan EW, Chin ML, Chan RC (2010) Delineation of a bacterial starvation stress response network which can mediate antibiotic tolerance development. Antimicrob Agents Chemother 54:1082–1093
Garrison AT et al (2015) Halogenated Phenazines that potently eradicate biofilms, MRSA Persister cells in non-biofilm cultures, and Mycobacterium tuberculosis. Angew Chem Int Ed Engl 54:14819–14823
Gefen O, Chekol B, Strahilevitz J, Balaban NQ (2017) TDtest: easy detection of bacterial tolerance and persistence in clinical isolates by a modified disk-diffusion assay. Sci Rep 7:41284
Germain E, Castro-Roa D, Zenkin N, Gerdes K (2013) Molecular mechanism of bacterial persistence by HipA. Mol Cell 52:248–254
Ghosh A et al (2018) Contact-dependent growth inhibition induces high levels of antibiotic-tolerant persister cells in clonal bacterial populations. EMBO J 37(9):e98026
Gi M et al (2014) A drug-repositioning screening identifies pentetic acid as a potential therapeutic agent for suppressing the elastase-mediated virulence of Pseudomonas aeruginosa. Antimicrob Agents Chemother 58:7205–7214
Giard JC et al (2001) The stress proteome of Enterococcus faecalis. Electrophoresis 22:2947–2954
Gobert AP et al (2001) Helicobacter pylori arginase inhibits nitric oxide production by eukaryotic cells: a strategy for bacterial survival. Proc Natl Acad Sci U S A 98:13844–13849
Goneau LW et al (2014) Selective target inactivation rather than global metabolic dormancy causes antibiotic tolerance in uropathogens. Antimicrob Agents Chemother 58:2089–2097
Grassi L, Maisetta G, Esin S, Batoni G (2017) Combination strategies to enhance the efficacy of antimicrobial peptides against bacterial biofilms. Front Microbiol 8:2409
Gutierrez A et al (2013) Beta-Lactam antibiotics promote bacterial mutagenesis via an RpoS-mediated reduction in replication fidelity. Nat Commun 4:1610
Hansen S, Lewis K, Vulic M (2008) Role of global regulators and nucleotide metabolism in antibiotic tolerance in Escherichia coli. Antimicrob Agents Chemother 52:2718–2726
Harrison JJ et al (2009) The chromosomal toxin gene yafQ is a determinant of multidrug tolerance for Escherichia coli growing in a biofilm. Antimicrob Agents Chemother 53:2253–2258
Haseltine WA, Block R (1973) Synthesis of guanosine tetra- and pentaphosphate requires the presence of a codon-specific, uncharged transfer ribonucleic acid in the acceptor site of ribosomes. Proc Natl Acad Sci U S A 70:1564–1568
He Z et al (2012) Use of the quorum sensing inhibitor furanone C-30 to interfere with biofilm formation by Streptococcus mutans and its luxS mutant strain. Int J Antimicrob Agents 40:30–35
Helaine S et al (2014) Internalization of Salmonella by macrophages induces formation of nonreplicating persisters. Science 343:204–208
Henry TC, Brynildsen MP (2016) Development of Persister-FACSeq: a method to massively parallelize quantification of persister physiology and its heterogeneity. Sci Rep 6:25100
Hijazi S et al (2018) Antimicrobial activity of gallium compounds on ESKAPE pathogens. Front Cell Infect Microbiol 8:316
Hong SH, Wang X, O'Connor HF, Benedik MJ, Wood TK (2012) Bacterial persistence increases as environmental fitness decreases. Microb Biotechnol 5:509–522
Honsa ES et al (2017) RelA mutant Enterococcus faecium with multiantibiotic tolerance arising in an immunocompromised host. mBio 8(1):e02124
Hubbard AT et al (2017) Mechanism of action of a membrane-active Quinoline-based antimicrobial on natural and model bacterial membranes. Biochemistry 56:1163–1174
Iino R, Matsumoto Y, Nishino K, Yamaguchi A, Noji H (2013) Design of a large-scale femtoliter droplet array for single-cell analysis of drug-tolerant and drug-resistant bacteria. Front Microbiol 4:300
Imperi F, Leoni L, Visca P (2014) Antivirulence activity of azithromycin in Pseudomonas aeruginosa. Front Microbiol 5:178
Imperi F et al (2013a) New life for an old drug: the anthelmintic drug niclosamide inhibits Pseudomonas aeruginosa quorum sensing. Antimicrob Agents Chemother 57:996–1005
Imperi F et al (2013b) Repurposing the antimycotic drug flucytosine for suppression of Pseudomonas aeruginosa pathogenicity. Proc Natl Acad Sci U S A 110:7458–7463
Jarchum I (2014) A one-two punch knocks out biofilms. Nat Biotechnol 32:142
Javid B et al (2014) Mycobacterial mistranslation is necessary and sufficient for rifampicin phenotypic resistance. Proc Natl Acad Sci U S A 111:1132–1137
Jiafeng L, Fu X, Chang Z (2015) Hypoionic shock treatment enables aminoglycosides antibiotics to eradicate bacterial persisters. Sci Rep 5:14247
Jiang M et al (2020) Antimicrobial activities of peptide Cbf-K16 against drug-resistant Helicobacter pylori infection in vitro and in vivo. Microb Pathog 138:103847
Joers A, Kaldalu N, Tenson T (2010) The frequency of persisters in Escherichia coli reflects the kinetics of awakening from dormancy. J Bacteriol 192:3379–3384
Joers A, Tenson T (2016) Growth resumption from stationary phase reveals memory in Escherichia coli cultures. Sci Rep 6:24055
Jubair M, Morris JG Jr, Ali A (2012) Survival of Vibrio cholerae in nutrient-poor environments is associated with a novel "persister" phenotype. PLoS One 7:e45187
Kalan L, Wright GD (2011) Antibiotic adjuvants: multicomponent anti-infective strategies. Expert Rev Mol Med 13:e5
Kaldalu N, Joers A, Ingelman H, Tenson T (2016) A general method for measuring Persister levels in Escherichia coli cultures. Methods Mol Biol 1333:29–42
Kaneko Y, Thoendel M, Olakanmi O, Britigan BE, Singh PK (2007) The transition metal gallium disrupts Pseudomonas aeruginosa iron metabolism and has antimicrobial and antibiofilm activity. J Clin Invest 117:877–888
Keren I, Minami S, Rubin E, Lewis K (2011) Characterization and transcriptome analysis of Mycobacterium tuberculosis persisters. mBio 2:e00100–e00111
Keren I, Shah D, Spoering A, Kaldalu N, Lewis K (2004) Specialized persister cells and the mechanism of multidrug tolerance in Escherichia coli. J Bacteriol 186:8172–8180
Keseler IM et al (2017) The EcoCyc database: reflecting new knowledge about Escherichia coli K-12. Nucleic Acids Res 45:D543–D550
Khan F, Pham DTN, Tabassum N, Oloketuyi SF, Kim YM (2020) Treatment strategies targeting persister cell formation in bacterial pathogens. Crit Rev Microbiol 46:665–688
Khodaverdian V et al (2013) Discovery of antivirulence agents against methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother 57:3645–3652
Kieser KJ, Rubin EJ (2014) How sisters grow apart: mycobacterial growth and division. Nat Rev Microbiol 12:550–562
Kim JH et al (2017) Synergistic antibacterial effects of chitosan-Caffeic acid conjugate against antibiotic-resistant acne-related Bacteria. Mar Drugs 15(6):167
Kim W et al (2018) A new class of synthetic retinoid antibiotics effective against bacterial persisters. Nature 556:103–107
Kim Y, Wood TK (2010) Toxins Hha and CspD and small RNA regulator Hfq are involved in persister cell formation through MqsR in Escherichia coli. Biochem Biophys Res Commun 391:209–213
Komp Lindgren P, Klockars O, Malmberg C, Cars O (2015) Pharmacodynamic studies of nitrofurantoin against common uropathogens. J Antimicrob Chemother 70:1076–1082
Koo H, Allan RN, Howlin RP, Stoodley P, Hall-Stoodley L (2017) Targeting microbial biofilms: current and prospective therapeutic strategies. Nat Rev Microbiol 15:740–755
Korch SB, Henderson TA, Hill TM (2003) Characterization of the hipA7 allele of Escherichia coli and evidence that high persistence is governed by (p)ppGpp synthesis. Mol Microbiol 50:1199–1213
Kostakioti M, Hadjifrangiskou M, Pinkner JS, Hultgren SJ (2009) QseC-mediated dephosphorylation of QseB is required for expression of genes associated with virulence in uropathogenic Escherichia coli. Mol Microbiol 73:1020–1031
Kotte O, Volkmer B, Radzikowski JL, Heinemann M (2014) Phenotypic bistability in Escherichia coli's central carbon metabolism. Mol Syst Biol 10:736
Kumar R et al (2019) Exploring the new horizons of drug repurposing: a vital tool for turning hard work into smart work. Eur J Med Chem 182:111602
Kwan BW, Chowdhury N, Wood TK (2015) Combatting bacterial infections by killing persister cells with mitomycin C. Environ Microbiol 17:4406–4414
Lehar SM et al (2015) Novel antibody-antibiotic conjugate eliminates intracellular S. aureus. Nature 527:323–328
Lei J et al (2019) The antimicrobial peptides and their potential clinical applications. Am J Transl Res 11:3919–3931
Leszczynska D, Matuszewska E, Kuczynska-Wisnik D, Furmanek-Blaszk B, Laskowska E (2013) The formation of persister cells in stationary-phase cultures of Escherichia coli is associated with the aggregation of endogenous proteins. PLoS One 8:e54737
Leung V, Levesque CM (2012) A stress-inducible quorum-sensing peptide mediates the formation of persister cells with noninherited multidrug tolerance. J Bacteriol 194:2265–2274
Levin BR, Concepcion-Acevedo J, Udekwu KI (2014) Persistence: a copacetic and parsimonious hypothesis for the existence of non-inherited resistance to antibiotics. Curr Opin Microbiol 21:18–21
Levin-Reisman I et al (2010) Automated imaging with ScanLag reveals previously undetectable bacterial growth phenotypes. Nat Methods 7:737–739
Levin-Reisman I et al (2017) Antibiotic tolerance facilitates the evolution of resistance. Science 355:826–830
Lewis K (2008) Multidrug tolerance of biofilms and persister cells. Curr Top Microbiol Immunol 322:107–131
Li C et al (2019) Two optimized antimicrobial peptides with therapeutic potential for clinical antibiotic-resistant Staphylococcus aureus. Eur J Med Chem 183:111686
Li F et al (2018) In vitro activity of beta-lactams in combination with beta-lactamase inhibitors against Mycobacterium tuberculosis clinical isolates. Biomed Res Int 2018:3579832
Li T, Yin N, Liu H, Pei J, Lai L (2016) Novel inhibitors of toxin HipA reduce multidrug tolerant Persisters. ACS Med Chem Lett 7:449–453
Li XZ, Nikaido H (2009) Efflux-mediated drug resistance in bacteria: an update. Drugs 69:1555–1623
Li Y, Zhang Y (2007) PhoU is a persistence switch involved in persister formation and tolerance to multiple antibiotics and stresses in Escherichia coli. Antimicrob Agents Chemother 51:2092–2099
Liebens V et al (2014) A putative de-N-acetylase of the PIG-L superfamily affects fluoroquinolone tolerance in Pseudomonas aeruginosa. Pathog Dis 71:39–54
Liu Q, Zheng Z, Kim W, Burgwyn Fuchs B, Mylonakis E (2018b) Influence of subinhibitory concentrations of NH125 on biofilm formation & virulence factors of Staphylococcus aureus. Future Med Chem 10:1319–1331
Liu S et al (2017) Variable Persister gene interactions with (p)ppGpp for Persister formation in Escherichia coli. Front Microbiol 8:1795
Liu Y, Kyle S, Straight PD (2018a) Antibiotic stimulation of a Bacillus subtilis migratory response. mSphere 3:00586
Lu TK, Collins JJ (2009) Engineered bacteriophage targeting gene networks as adjuvants for antibiotic therapy. Proc Natl Acad Sci U S A 106:4629–4634
Luidalepp H, Joers A, Kaldalu N, Tenson T (2011) Age of inoculum strongly influences persister frequency and can mask effects of mutations implicated in altered persistence. J Bacteriol 193:3598–3605
Ma D et al (2019) The toxin-antitoxin MazEF drives Staphylococcus aureus biofilm formation, antibiotic tolerance, and chronic infection. mBio 10(6):e01658
MacGilvary NJ, Tan S (2018) Fluorescent Mycobacterium tuberculosis reporters: illuminating host-pathogen interactions. Pathog Dis 76(3):fty017
Maiden MM et al (2018) Triclosan is an aminoglycoside adjuvant for eradication of Pseudomonas aeruginosa biofilms. Antimicrob Agents Chemother 62(6):e00146
Manina G, Dhar N, McKinney JD (2015) Stress and host immunity amplify Mycobacterium tuberculosis phenotypic heterogeneity and induce nongrowing metabolically active forms. Cell Host Microbe 17:32–46
Manuel J, Zhanel GG, de Kievit T (2010) Cadaverine suppresses persistence to carboxypenicillins in Pseudomonas aeruginosa PAO1. Antimicrob Agents Chemother 54:5173–5179
Marques CN, Morozov A, Planzos P, Zelaya HM (2014) The fatty acid signaling molecule cis-2-decenoic acid increases metabolic activity and reverts persister cells to an antimicrobial-susceptible state. Appl Environ Microbiol 80:6976–6991
Marr AK, Gooderham WJ, Hancock RE (2006) Antibacterial peptides for therapeutic use: obstacles and realistic outlook. Curr Opin Pharmacol 6:468–472
Meena M, Prasad V, Zehra A, Gupta VK, Upadhyay RS (2015) Mannitol metabolism during pathogenic fungal-host interactions under stressed conditions. Front Microbiol 6:1019
Meyer KJ, Taylor HB, Seidel J, Gates MF, Lewis K (2020) Pulse dosing of antibiotic enhances killing of a Staphylococcus aureus biofilm. Front Microbiol 11:596227
Michiels JE, Van den Bergh B, Verstraeten N, Michiels J (2016) Molecular mechanisms and clinical implications of bacterial persistence. Drug Resist Updat 29:76–89
Mohamed MF, Brezden A, Mohammad H, Chmielewski J, Seleem MN (2017) Targeting biofilms and persisters of ESKAPE pathogens with P14KanS, a kanamycin peptide conjugate. Biochim Biophys Acta Gen Subj 1861:848–859
Moker N, Dean CR, Tao J (2010) Pseudomonas aeruginosa increases formation of multidrug-tolerant persister cells in response to quorum-sensing signaling molecules. J Bacteriol 192:1946–1955
Mukamolova GV, Kaprelyants AS, Young DI, Young M, Kell DB (1998) A bacterial cytokine. Proc Natl Acad Sci U S A 95:8916–8921
Mukamolova GV et al (2006) Muralytic activity of Micrococcus luteus Rpf and its relationship to physiological activity in promoting bacterial growth and resuscitation. Mol Microbiol 59:84–98
Mukherjee D, Zou H, Liu S, Beuerman R, Dick T (2016) Membrane-targeting AM-0016 kills mycobacterial persisters and shows low propensity for resistance development. Future Microbiol 11:643–650
Mulcahy LR, Burns JL, Lory S, Lewis K (2010) Emergence of Pseudomonas aeruginosa strains producing high levels of persister cells in patients with cystic fibrosis. J Bacteriol 192:6191–6199
Mwangi J et al (2019) The antimicrobial peptide ZY4 combats multidrug-resistant Pseudomonas aeruginosa and Acinetobacter baumannii infection. Proc Natl Acad Sci U S A 116(52):26516–26522
Namugenyi SB, Aagesen AM, Elliott SR, Tischler AD (2017) Mycobacterium tuberculosis PhoY proteins promote persister formation by mediating Pst/SenX3-RegX3 phosphate sensing. mBio 8(4):e00494
Nathan C (2012) Fresh approaches to anti-infective therapies. Sci Transl Med 4:140–142
Nautiyal A, Patil KN, Muniyappa K (2014) Suramin is a potent and selective inhibitor of Mycobacterium tuberculosis RecA protein and the SOS response: RecA as a potential target for antibacterial drug discovery. J Antimicrob Chemother 69:1834–1843
Niepa TH, Gilbert JL, Ren D (2012) Controlling Pseudomonas aeruginosa persister cells by weak electrochemical currents and synergistic effects with tobramycin. Biomaterials 33:7356–7365
Nikitushkin VD et al (2015) A product of RpfB and RipA joint enzymatic action promotes the resuscitation of dormant mycobacteria. FEBS J 282:2500–2511
Nistico L et al (2009) Fluorescence "in situ" hybridization for the detection of biofilm in the middle ear and upper respiratory tract mucosa. Methods Mol Biol 493:191–213
Norton JP, Mulvey MA (2012) Toxin-antitoxin systems are important for niche-specific colonization and stress resistance of uropathogenic Escherichia coli. PLoS Pathog 8:e1002954
Nuermberger E et al (2006) Combination chemotherapy with the nitroimidazopyran PA-824 and first-line drugs in a murine model of tuberculosis. Antimicrob Agents Chemother 50:2621–2625
O'Driscoll T, Crank CW (2015) Vancomycin-resistant enterococcal infections: epidemiology, clinical manifestations, and optimal management. Infect Drug Resist 8:217–230
Orman MA, Brynildsen MP (2013a) Dormancy is not necessary or sufficient for bacterial persistence. Antimicrob Agents Chemother 57:3230–3239
Orman MA, Brynildsen MP (2013b) Establishment of a method to rapidly assay bacterial persister metabolism. Antimicrob Agents Chemother 57:4398–4409
Orman MA, Brynildsen MP (2015) Inhibition of stationary phase respiration impairs persister formation in E. coli. Nat Commun 6:7983
Pan J, Bahar AA, Syed H, Ren D (2012) Reverting antibiotic tolerance of Pseudomonas aeruginosa PAO1 persister cells by (Z)-4-bromo-5-(bromomethylene)-3-methylfuran-2(5H)-one. PLoS One 7:e45778
Pan J, Song F, Ren D (2013) Controlling persister cells of Pseudomonas aeruginosa PDO300 by (Z)-4-bromo-5-(bromomethylene)-3-methylfuran-2(5H)-one. Bioorg Med Chem Lett 23:4648–4651
Paranjape SS, Shashidhar R (2020) Glucose sensitizes the stationary and persistent population of Vibrio cholerae to ciprofloxacin. Arch Microbiol 202:343–349
Pedersen K et al (2003) The bacterial toxin RelE displays codon-specific cleavage of mRNAs in the ribosomal a site. Cell 112:131–140
Peng B et al (2015) Exogenous alanine and/or glucose plus kanamycin kills antibiotic-resistant bacteria. Cell Metab 21:249–262
Perlmutter JI et al (2014) Repurposing the antihistamine terfenadine for antimicrobial activity against Staphylococcus aureus. J Med Chem 57:8540–8562
Perry JA, Jones MB, Peterson SN, Cvitkovitch DG, Levesque CM (2009) Peptide alarmone signalling triggers an auto-active bacteriocin necessary for genetic competence. Mol Microbiol 72:905–917
Petchiappan A, Chatterji D (2017) Antibiotic resistance: current perspectives. ACS Omega 2:7400–7409
Pinault L, Han JS, Kang CM, Franco J, Ronning DR (2013) Zafirlukast inhibits complexation of Lsr2 with DNA and growth of Mycobacterium tuberculosis. Antimicrob Agents Chemother 57:2134–2140
Portela CA, Smart KF, Tumanov S, Cook GM, Villas-Boas SG (2014) Global metabolic response of Enterococcus faecalis to oxygen. J Bacteriol 196:2012–2022
Potrykus K, Cashel M (2008) (p)ppGpp: still magical? Annu Rev Microbiol 62:35–51
Prax M, Bertram R (2014) Metabolic aspects of bacterial persisters. Front Cell Infect Microbiol 4:148
Prysak MH et al (2009) Bacterial toxin YafQ is an endoribonuclease that associates with the ribosome and blocks translation elongation through sequence-specific and frame-dependent mRNA cleavage. Mol Microbiol 71:1071–1087
Pu Y et al (2016) Enhanced efflux activity facilitates drug tolerance in dormant bacterial cells. Mol Cell 62:284–294
Que YA et al (2013) A quorum sensing small volatile molecule promotes antibiotic tolerance in bacteria. PLoS One 8:e80140
Ragheb MN et al (2019) Inhibiting the evolution of antibiotic resistance. Mol Cell 73:157–165
Ramadoss NS et al (2013) Small molecule inhibitors of trans-translation have broad-spectrum antibiotic activity. Proc Natl Acad Sci U S A 110:10282–10287
Ramirez M et al (2016) Diverse drug-resistance mechanisms can emerge from drug-tolerant cancer persister cells. Nat Commun 7:10690
Rangel-Vega A, Bernstein LR, Mandujano-Tinoco EA, Garcia-Contreras SJ, Garcia-Contreras R (2015) Drug repurposing as an alternative for the treatment of recalcitrant bacterial infections. Front Microbiol 6:282
Rao NN, Liu S, Kornberg A (1998) Inorganic polyphosphate in Escherichia coli: the phosphate regulon and the stringent response. J Bacteriol 180:2186–2193
Ratcliff WC, Denison RF (2011) Bacterial persistence and bet hedging in Sinorhizobium meliloti. Commun Integr Biol 4:98–100
Ravagnani A, Finan CL, Young M (2005) A novel firmicute protein family related to the actinobacterial resuscitation-promoting factors by non-orthologous domain displacement. BMC Genom 6:39
Reffuveille F, de la Fuente-Nunez C, Mansour S, Hancock RE (2014) A broad-spectrum antibiofilm peptide enhances antibiotic action against bacterial biofilms. Antimicrob Agents Chemother 58:5363–5371
Rego EH, Audette RE, Rubin EJ (2017) Deletion of a mycobacterial divisome factor collapses single-cell phenotypic heterogeneity. Nature 546:153–157
Roostalu J, Joers A, Luidalepp H, Kaldalu N, Tenson T (2008) Cell division in Escherichia coli cultures monitored at single cell resolution. BMC Microbiol 8:68
Rosser A, Stover C, Pareek M, Mukamolova GV (2017) Resuscitation-promoting factors are important determinants of the pathophysiology in Mycobacterium tuberculosis infection. Crit Rev Microbiol 43:621–630
Rowe SE, Conlon BP, Keren I, Lewis K (2016) Persisters: methods for isolation and identifying contributing factors--a review. Methods Mol Biol 1333:17–28
Sakatos A et al (2018) Posttranslational modification of a histone-like protein regulates phenotypic resistance to isoniazid in mycobacteria. Sci Adv 4:eaao1478
Saliba AE et al (2016) Single-cell RNA-seq ties macrophage polarization to growth rate of intracellular Salmonella. Nat Microbiol 2:16206
Scherrer R, Moyed HS (1988) Conditional impairment of cell division and altered lethality in hipA mutants of Escherichia coli K-12. J Bacteriol 170:3321–3326
Schmidt NW et al (2014) Engineering persister-specific antibiotics with synergistic antimicrobial functions. ACS Nano 8:8786–8793
Schmidt NW et al (2015) Pentobra: a potent antibiotic with multiple layers of selective antimicrobial mechanisms against Propionibacterium acnes. J Invest Dermatol 135:1581–1589
Shah D et al (2006) Persisters: a distinct physiological state of E. coli. BMC Microbiol 6:53
Shan Y, Lazinski D, Rowe S, Camilli A, Lewis K (2015) Genetic basis of persister tolerance to aminoglycosides in Escherichia coli. mBio 6(2):e0007
Shapiro JA, Nguyen VL, Chamberlain NR (2011) Evidence for persisters in Staphylococcus epidermidis RP62a planktonic cultures and biofilms. J Med Microbiol 60:950–960
Sharma B, Brown AV, Matluck NE, Hu LT, Lewis K (2015) Borrelia burgdorferi, the causative agent of Lyme disease, forms drug-tolerant persister cells. Antimicrob Agents Chemother 59:4616–4624
Shi W et al (2011) Pyrazinamide inhibits trans-translation in Mycobacterium tuberculosis. Science 333:1630–1632
Singh R et al (2008) PA-824 kills nonreplicating Mycobacterium tuberculosis by intracellular NO release. Science 322:1392–1395
Spoering AL, Vulic M, Lewis K (2006) GlpD and PlsB participate in persister cell formation in Escherichia coli. J Bacteriol 188:5136–5144
Stapels DAC et al (2018) Salmonella persisters undermine host immune defenses during antibiotic treatment. Science 362:1156–1160
Tang HJ et al (2019) Colistin-sparing regimens against Klebsiella pneumoniae carbapenemase-producing K. pneumoniae isolates: Combination of tigecycline or doxycycline and gentamicin or amikacin. J Microbiol Immunol Infect 52:273–281
Tashiro Y et al (2012) RelE-mediated dormancy is enhanced at high cell density in Escherichia coli. J Bacteriol 194:1169–1176
Theodore A, Lewis K, Vulic M (2013) Tolerance of Escherichia coli to fluoroquinolone antibiotics depends on specific components of the SOS response pathway. Genetics 195:1265–1276
Tiwari P et al (2015) MazF ribonucleases promote Mycobacterium tuberculosis drug tolerance and virulence in Guinea pigs. Nat Commun 6:6059
Tkhilaishvili T, Lombardi L, Klatt AB, Trampuz A, Di Luca M (2018) Bacteriophage Sb-1 enhances antibiotic activity against biofilm, degrades exopolysaccharide matrix and targets persisters of Staphylococcus aureus. Int J Antimicrob Agents 52:842–853
Tkhilaishvili T, Wang L, Perka C, Trampuz A, Gonzalez Moreno M (2020) Using bacteriophages as a Trojan horse to the killing of dual-species biofilm formed by Pseudomonas aeruginosa and methicillin resistant Staphylococcus aureus. Front Microbiol 11:695
Torrey HL, Keren I, Via LE, Lee JS, Lewis K (2016) High Persister mutants in Mycobacterium tuberculosis. PLoS One 11:e0155127
Tripathi A, Dewan PC, Siddique SA, Varadarajan R (2014) MazF-induced growth inhibition and persister generation in Escherichia coli. J Biol Chem 289:4191–4205
Ueda A, Attila C, Whiteley M, Wood TK (2009) Uracil influences quorum sensing and biofilm formation in Pseudomonas aeruginosa and fluorouracil is an antagonist. Microb Biotechnol 2:62–74
Ueta M et al (2008) Role of HPF (hibernation promoting factor) in translational activity in Escherichia coli. J Biochem 143:425–433
Van Acker H, Sass A, Dhondt I, Nelis HJ, Coenye T (2014) Involvement of toxin-antitoxin modules in Burkholderia cenocepacia biofilm persistence. Pathog Dis 71:326–335
Van den Bergh B, Fauvart M, Michiels J (2017) Formation, physiology, ecology, evolution and clinical importance of bacterial persisters. FEMS Microbiol Rev 41:219–251
Van den Bergh B et al (2016) Frequency of antibiotic application drives rapid evolutionary adaptation of Escherichia coli persistence. Nat Microbiol 1:16020
van Opijnen T, Bodi KL, Camilli A (2009) Tn-seq: high-throughput parallel sequencing for fitness and genetic interaction studies in microorganisms. Nat Methods 6:767–772
Vazquez-Laslop N, Lee H, Neyfakh AA (2006) Increased persistence in Escherichia coli caused by controlled expression of toxins or other unrelated proteins. J Bacteriol 188:3494–3497
Vega NM, Allison KR, Khalil AS, Collins JJ (2012) Signaling-mediated bacterial persister formation. Nat Chem Biol 8:431–433
Vega NM, Allison KR, Samuels AN, Klempner MS, Collins JJ (2013) Salmonella typhimurium intercepts Escherichia coli signaling to enhance antibiotic tolerance. Proc Natl Acad Sci U S A 110:14420–14425
Verstraeten N et al (2015) Obg and membrane depolarization are part of a microbial bet-hedging strategy that leads to antibiotic tolerance. Mol Cell 59:9–21
Walz JM et al (2010) Anti-infective external coating of central venous catheters: a randomized, noninferiority trial comparing 5-fluorouracil with chlorhexidine/silver sulfadiazine in preventing catheter colonization. Crit Care Med 38:2095–2102
Wang F et al (2013) Identification of a small molecule with activity against drug-resistant and persistent tuberculosis. Proc Natl Acad Sci U S A 110:E2510–E2517
Wenthzel AM, Stancek M, Isaksson LA (1998) Growth phase dependent stop codon readthrough and shift of translation reading frame in Escherichia coli. FEBS Lett 421:237–242
Westermann AJ, Barquist L, Vogel J (2017) Resolving host-pathogen interactions by dual RNA-seq. PLoS Pathog 13:e1006033
Wilmaerts D, Windels EM, Verstraeten N, Michiels J (2019) General mechanisms leading to Persister formation and awakening. Trends Genet 35:401–411
Wilmaerts D et al (2018) The persistence-inducing toxin HokB forms dynamic pores that cause ATP leakage. mBio 9(4):e00744
Windels EM et al (2019) Bacterial persistence promotes the evolution of antibiotic resistance by increasing survival and mutation rates. ISME J 13:1239–1251
Wood DN, Chaussee MA, Chaussee MS, Buttaro BA (2005) Persistence of Streptococcus pyogenes in stationary-phase cultures. J Bacteriol 187:3319–3328
Wu N et al (2015) Ranking of persister genes in the same Escherichia coli genetic background demonstrates varying importance of individual persister genes in tolerance to different antibiotics. Front Microbiol 6:1003
Wu S, Yu P-L, Flint S (2017) Persister cell formation of Listeria monocytogenes in response to natural antimicrobial agent nisin. Food Control 77:243–250
Wu Y, Vulic M, Keren I, Lewis K (2012) Role of oxidative stress in persister tolerance. Antimicrob Agents Chemother 56:4922–4926
Wuyts J, Van Dijck P, Holtappels M (2018) Fungal persister cells: the basis for recalcitrant infections? PLoS Pathog 14:e1007301
**ao H et al (1991) Residual guanosine 3′,5′-bispyrophosphate synthetic activity of relA null mutants can be eliminated by spoT null mutations. J Biol Chem 266:5980–5990
Yamaguchi Y, Park JH, Inouye M (2009) MqsR, a crucial regulator for quorum sensing and biofilm formation, is a GCU-specific mRNA interferase in Escherichia coli. J Biol Chem 284:28746–28753
Yang S et al (2015) Antibiotic regimen based on population analysis of residing persister cells eradicates Staphylococcus epidermidis biofilms. Sci Rep 5:18578
Yuksel M, Power JJ, Ribbe J, Volkmann T, Maier B (2016) Fitness trade-offs in competence differentiation of Bacillus subtilis. Front Microbiol 7:888
Zalis EA et al (2019) Stochastic variation in expression of the tricarboxylic acid cycle produces persister cells. mBio 10(5):e01930
Zhang Y (2014) Persisters, persistent infections and the Yin-Yang model. Emerg Microbes Infect 3:e3
Zhang Y, Yew WW, Barer MR (2012) Targeting persisters for tuberculosis control. Antimicrob Agents Chemother 56:2223–2230
Zhang Y, Zhang J, Hara H, Kato I, Inouye M (2005) Insights into the mRNA cleavage mechanism by MazF, an mRNA interferase. J Biol Chem 280:3143–3150
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Mamta Singla, Chaudhary, V., Ghosh, A. (2022). Bacterial Multidrug Tolerance and Persisters: Understanding the Mechanisms, Clinical Implications, and Treatment Strategies. In: Kumar, V., Shriram, V., Paul, A., Thakur, M. (eds) Antimicrobial Resistance. Springer, Singapore. https://doi.org/10.1007/978-981-16-3120-7_2
Download citation
DOI: https://doi.org/10.1007/978-981-16-3120-7_2
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-3119-1
Online ISBN: 978-981-16-3120-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)