Abstract
The estrogen receptor alpha (ERα) is a nuclear transcription factor that is expressed in more than 70% of all breast cancers. Key genes involved in proliferation and tumor progression are transcriptionally regulated by ERα making it an important therapeutic target. Indeed, the first class of targeted treatments in cancer are endocrine treatments that target ERα either by competitive inhibition, reduced ligand production or receptor degradation. Despite the efficacy of these drugs, resistance to endocrine treatment remains a key clinical challenge. Only about 50% of patients treated with endocrine treatment in early-stage disease will benefit from adjuvant endocrine treatment and nearly all patients treated in the metastatic setting will develop disease progression while on endocrine treatment. Multiple mechanisms of resistance to endocrine treatment have been identified in pre-clinical models and clinical samples. These include both intrinsic (de novo) mechanisms and adaptive, acquired mechanisms. Over the past few years, gain-of-function missense mutations of ESR1, the gene encoding ERα, have been unveiled and identified as the most common genomic mechanism of acquired resistance to endocrine treatments. These mutations are clustered in a “hot spot” region within the ligand binding domain and engender constitutive, ligand-independent activity. Clinical studies evaluating these ESR1 mutations in metastatic ERα positive breast cancer demonstrate decreased overall survival which also highlights their prognostic role. In this chapter, we will provide a detailed review of structural and biophysical characteristics, functional consequences and clinical implications of the ESR1 mutations. We will also discuss potential therapeutic strategies to overcome treatment resistance in the context of ESR1 mutations and implications for future treatment selection.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jensen EV (1962) On the mechanism of estrogen action. Perspect Biol Med 6:47–59. https://doi.org/10.1353/pbm.1963.0005
Jensen EV, Jacobson HI, Walf AA, Frye CA (2010) Estrogen action: a historic perspective on the implications of considering alternative approaches. Physiol Behav 99:151–162. https://doi.org/10.1016/j.physbeh.2009.08.013
Kuiper GG, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JA (1996) Cloning of a novel receptor expressed in rat prostate and ovary. Proc Natl Acad Sci U S A 93:5925–5930. https://doi.org/10.1073/pnas.93.12.5925
Mosselman S, Polman J, Dijkema R (1996) ER beta: identification and characterization of a novel human estrogen receptor. FEBS Lett 392:49–53. https://doi.org/10.1016/0014-5793(96)00782-x
Sever R, Glass CK (2013) Signaling by nuclear receptors. Cold Spring Harb Perspect Biol 5:a016709. https://doi.org/10.1101/cshperspect.a016709
Penvose A, Keenan JL, Bray D, Ramlall V, Siggers T (2019) Comprehensive study of nuclear receptor DNA binding provides a revised framework for understanding receptor specificity. Nat Commun 10:2514. https://doi.org/10.1038/s41467-019-10264-3
Bain DL, Heneghan AF, Connaghan-Jones KD, Miura MT (2007) Nuclear receptor structure: implications for function. Annu Rev Physiol 69:201–220. https://doi.org/10.1146/annurev.physiol.69.031905.160308
McKenna NJ, O’Malley BW (2002) Combinatorial control of gene expression by nuclear receptors and coregulators. Cell 108:465–474. https://doi.org/10.1016/s0092-8674(02)00641-4
Green S et al (1986) Human oestrogen receptor cDNA: sequence, expression and homology to v-erb-A. Nature 320:134–139. https://doi.org/10.1038/320134a0
Walter P et al (1985) Cloning of the human estrogen receptor cDNA. Proc Natl Acad Sci U S A 82:7889–7893. https://doi.org/10.1073/pnas.82.23.7889
Ogawa S et al (1998) The complete primary structure of human estrogen receptor β (hERβ) and its Heterodimerization with ER αin Vivoandin vitro. Biochem Biophys Res Commun 243:122–126. https://doi.org/10.1006/bbrc.1997.7893
Sand P, Luckhaus C, Schlurmann K, Götz M, Deckert J (2002) Untangling the human estrogen receptor gene structure. J Neural Transm 109:567–583. https://doi.org/10.1007/s007020200047
Kumar V et al (1987) Functional domains of the human estrogen receptor. Cell 51:941–951. https://doi.org/10.1016/0092-8674(87)90581-2
Bunce CM, Campbell MJ, Nilsson S, Gustafsson J-Å (2010) Estrogen receptors: their actions and functional roles in health and disease. In: Bunce CM, Campbell MJ (eds) Nuclear receptors. Springer, Dordrecht, pp 91–141. https://doi.org/10.1007/978-90-481-3303-1_5
Huang W et al (2018) Multidomain architecture of estrogen receptor reveals interfacial cross-talk between its DNA-binding and ligand-binding domains. Nat Commun 9:3520. https://doi.org/10.1038/s41467-018-06034-2
Métivier R et al (2003) Estrogen receptor-alpha directs ordered, cyclical, and combinatorial recruitment of cofactors on a natural target promoter. Cell 115:751–763. https://doi.org/10.1016/s0092-8674(03)00934-6
Katzenellenbogen BS (1996) Estrogen receptors: bioactivities and interactions with cell signaling pathways. Biol Reprod 54:287–293. https://doi.org/10.1095/biolreprod54.2.287
Bunone G, Briand PA, Miksicek RJ, Picard D (1996) Activation of the unliganded estrogen receptor by EGF involves the MAP kinase pathway and direct phosphorylation. EMBO J 15:2174–2183
Kato S et al (1995) Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase. Science (New York, NY) 270:1491–1494. https://doi.org/10.1126/science.270.5241.1491
Cenni B, Picard D (1999) Ligand-independent activation of steroid receptors: new roles for old players. Trends Endocrinol Metab 10:41–46. https://doi.org/10.1016/s1043-2760(98)00121-0
Lavery DN, McEwan IJ (2005) Structure and function of steroid receptor AF1 transactivation domains: induction of active conformations. Biochem J 391:449–464. https://doi.org/10.1042/BJ20050872
Ignar-Trowbridge DM, Pimentel M, Teng CT, Korach KS, McLachlan JA (1995) Cross talk between peptide growth factor and estrogen receptor signaling systems. Environ Health Perspect 103(Suppl 7):35–38. https://doi.org/10.1289/ehp.95103s735
Métivier RI, Petit FG, Valotaire Y, Pakdel F (2000) Function of N-terminal transactivation domain of the estrogen receptor requires a potential α-helical structure and is negatively regulated by the a domain. Mol Endocrinol 14:1849–1871. https://doi.org/10.1210/mend.14.11.0546
Schwabe JW, Chapman L, Finch JT, Rhodes D, Neuhaus D (1993) DNA recognition by the oestrogen receptor: from solution to the crystal. Structure (London, England: 1993) 1:187–204. https://doi.org/10.1016/0969-2126(93)90020-h
Carroll JS et al (2005) Chromosome-wide map** of estrogen receptor binding reveals long-range regulation requiring the forkhead protein FoxA1. Cell 122:33–43. https://doi.org/10.1016/j.cell.2005.05.008
Laganière J et al (2005) Location analysis of estrogen receptor α target promoters reveals that FOXA1 defines a domain of the estrogen response. Proc Natl Acad Sci U S A 102:11651–11656. https://doi.org/10.1073/pnas.0505575102
Lupien M et al (2008) FoxA1 translates epigenetic signatures into enhancer-driven lineage-specific transcription. Cell 132:958–970. https://doi.org/10.1016/j.cell.2008.01.018
Hurtado A, Holmes KA, Ross-Innes CS, Schmidt D, Carroll JS (2011) FOXA1 is a key determinant of estrogen receptor function and endocrine response. Nat Genet 43:27–33. https://doi.org/10.1038/ng.730
Wang C et al (2001) Direct acetylation of the estrogen receptor alpha hinge region by p300 regulates transactivation and hormone sensitivity. J Biol Chem 276:18375–18383. https://doi.org/10.1074/jbc.M100800200
Kim MY, Woo EM, Chong YTE, Homenko DR, Kraus WL (2006) Acetylation of estrogen receptor alpha by p300 at lysines 266 and 268 enhances the deoxyribonucleic acid binding and transactivation activities of the receptor. Mol Endocrinol 20:1479–1493. https://doi.org/10.1210/me.2005-0531
Sentis S, Le Romancer M, Bianchin C, Rostan MC, Corbo L (2005) Sumoylation of the estrogen receptor alpha hinge region regulates its transcriptional activity. Mol Endocrinol 19:2671–2684. https://doi.org/10.1210/me.2005-0042
Berry NB, Fan M, Nephew KP (2008) Estrogen receptor-alpha hinge-region lysines 302 and 303 regulate receptor degradation by the proteasome. Mol Endocrinol 22:1535–1551. https://doi.org/10.1210/me.2007-0449
Moras D, Gronemeyer H (1998) The nuclear receptor ligand-binding domain: structure and function. Curr Opin Cell Biol 10:384–391. https://doi.org/10.1016/s0955-0674(98)80015-x
Brzozowski AM et al (1997) Molecular basis of agonism and antagonism in the oestrogen receptor. Nature 389:753–758. https://doi.org/10.1038/39645
Shiau AK et al (1998) The structural basis of estrogen receptor/coactivator recognition and the antagonism of this interaction by tamoxifen. Cell 95:927–937. https://doi.org/10.1016/s0092-8674(00)81717-1
Pike AC et al (1999) Structure of the ligand-binding domain of oestrogen receptor beta in the presence of a partial agonist and a full antagonist. EMBO J 18:4608–4618. https://doi.org/10.1093/emboj/18.17.4608
Shang Y, Hu X, DiRenzo J, Lazar MA, Brown M (2000) Cofactor dynamics and sufficiency in estrogen receptor-regulated transcription. Cell 103:843–852. https://doi.org/10.1016/s0092-8674(00)00188-4
Pike AC et al (2001) Structural insights into the mode of action of a pure antiestrogen. Structure (London, England: 1993) 9:145–153. https://doi.org/10.1016/s0969-2126(01)00568-8
Valentine JE, Kalkhoven E, White R, Hoare S, Parker MG (2000) Mutations in the estrogen receptor ligand binding domain discriminate between hormone-dependent transactivation and transrepression. J Biol Chem 275:25322–25329. https://doi.org/10.1074/jbc.M002497200
Tamrazi A, Carlson KE, Daniels JR, Hurth KM, Katzenellenbogen JA (2002) Estrogen receptor dimerization: ligand binding regulates dimer affinity and DimerDissociation rate. Mol Endocrinol 16:2706–2719. https://doi.org/10.1210/me.2002-0250
Tamrazi A, Carlson KE, Rodriguez AL, Katzenellenbogen JA (2005) Coactivator proteins as determinants of estrogen receptor structure and function: spectroscopic evidence for a novel coactivator-stabilized receptor conformation. Mol Endocrinol 19:1516–1528. https://doi.org/10.1210/me.2004-0458
Montano MM, Müller V, Trobaugh A, Katzenellenbogen BS (1995) The carboxy-terminal F domain of the human estrogen receptor: role in the transcriptional activity of the receptor and the effectiveness of antiestrogens as estrogen antagonists. Mol Endocrinol 9:814–825. https://doi.org/10.1210/mend.9.7.7476965
Kim K, Thu N, Saville B, Safe S (2003) Domains of estrogen receptor alpha (ERalpha) required for ERalpha/Sp1-mediated activation of GC-rich promoters by estrogens and antiestrogens in breast cancer cells. Mol Endocrinol 17:804–817. https://doi.org/10.1210/me.2002-0406
Gruber CJ, Tschugguel W, Schneeberger C, Huber JC (2002) Production and actions of estrogens. N Engl J Med 346:340–352. https://doi.org/10.1056/NEJMra000471
Webb P et al (1999) The estrogen receptor enhances AP-1 activity by two distinct mechanisms with different requirements for receptor transactivation functions. Mol Endocrinol 13:1672–1685. https://doi.org/10.1210/mend.13.10.0357
Nagy L, Schwabe JW (2004) Mechanism of the nuclear receptor molecular switch. Trends Biochem Sci 29:317–324. https://doi.org/10.1016/j.tibs.2004.04.006
Wärnmark A et al (2002) Interaction of transcriptional intermediary factor 2 nuclear receptor box peptides with the coactivator binding site of estrogen receptor alpha. J Biol Chem 277:21862–21868. https://doi.org/10.1074/jbc.M200764200
Pike AC (2006) Lessons learnt from structural studies of the oestrogen receptor. Best Pract Res Clin Endocrinol Metab 20:1–14. https://doi.org/10.1016/j.beem.2005.09.002
Wu YL et al (2005) Structural basis for an unexpected mode of SERM-mediated ER antagonism. Mol Cell 18:413–424. https://doi.org/10.1016/j.molcel.2005.04.014
Hu X, Lazar MA (1999) The CoRNR motif controls the recruitment of corepressors by nuclear hormone receptors. Nature 402:93–96. https://doi.org/10.1038/47069
Chang CY et al (1999) Dissection of the LXXLL nuclear receptor-coactivator interaction motif using combinatorial peptide libraries: discovery of peptide antagonists of estrogen receptors alpha and beta. Mol Cell Biol 19:8226–8239. https://doi.org/10.1128/MCB.19.12.8226
Kong EH et al (2005) Delineation of a unique protein-protein interaction site on the surface of the estrogen receptor. Proc Natl Acad Sci U S A 102:3593–3598. https://doi.org/10.1073/pnas.0407189102
Webb P et al (1998) Estrogen receptor activation function 1 works by binding p160 coactivator proteins. Mol Endocrinol 12:1605–1618. https://doi.org/10.1210/mend.12.10.0185
Métivier RI, Penot G, Flouriot G, Pakdel F (2001) Synergism between ERα transactivation function 1 (AF-1) and AF-2 mediated by steroid receptor coactivator Protein-1: requirement for the AF-1 α-helical Core and for a direct interaction between the N- and C-terminal domains. Mol Endocrinol 15:1953–1970. https://doi.org/10.1210/mend.15.11.0727
Onate SA et al (1998) The steroid receptor coactivator-1 contains multiple receptor interacting and activation domains that cooperatively enhance the activation function 1 (AF1) and AF2 domains of steroid receptors. J Biol Chem 273:12101–12108. https://doi.org/10.1074/jbc.273.20.12101
Benecke A, Chambon P, Gronemeyer H (2000) Synergy between estrogen receptor alpha activation functions AF1 and AF2 mediated by transcription intermediary factor TIF2. EMBO Rep 1:151–157. https://doi.org/10.1093/embo-reports/kvd028
Cheskis BJ, Karathanasis S, Lyttle CR (1997) Estrogen receptor ligands modulate its interaction with DNA. J Biol Chem 272:11384–11391. https://doi.org/10.1074/jbc.272.17.11384
Krieg AJ, Krieg SA, Ahn BS, Shapiro DJ (2004) Interplay between estrogen response element sequence and ligands controls in vivo binding of estrogen receptor to regulated genes. J Biol Chem 279:5025–5034. https://doi.org/10.1074/jbc.M307076200
Treviño LS, Weigel NL (2013) Phosphorylation: a fundamental regulator of steroid receptor action. Trends Endocrinol Metab 24:515–524. https://doi.org/10.1016/j.tem.2013.05.008
Shao D, Lazar MA (1999) Modulating nuclear receptor function: may the phos be with you. J Clin Invest 103:1617–1618. https://doi.org/10.1172/JCI7421
Smith CL, Conneely OM, O’Malley BW (1993) Modulation of the ligand-independent activation of the human estrogen receptor by hormone and antihormone. Proc Natl Acad Sci U S A 90:6120–6124. https://doi.org/10.1073/pnas.90.13.6120
Ignar-Trowbridge DM et al (1993) Peptide growth factors elicit estrogen receptor-dependent transcriptional activation of an estrogen-responsive element. Mol Endocrinol 7:992–998. https://doi.org/10.1210/mend.7.8.8232319
Newton CJ et al (1994) The unliganded estrogen receptor (ER) transduces growth factor signals. J Steroid Biochem Mol Biol 48:481–486. https://doi.org/10.1016/0960-0760(94)90197-x
Pietras RJ et al (1995) HER-2 tyrosine kinase pathway targets estrogen receptor and promotes hormone-independent growth in human breast cancer cells. Oncogene 10:2435–2446
Gruber CJ, Gruber DM, Gruber IM, Wieser F, Huber JC (2004) Anatomy of the estrogen response element. Trends Endocrinol Metab 15:73–78. https://doi.org/10.1016/j.tem.2004.01.008
Zilliacus J, Wright AP, Carlstedt-Duke J, Gustafsson JA (1995) Structural determinants of DNA-binding specificity by steroid receptors. Mol Endocrinol 9:389–400. https://doi.org/10.1210/mend.9.4.7659083
Klinge CM (2001) Estrogen receptor interaction with estrogen response elements. Nucleic Acids Res 29:2905–2919. https://doi.org/10.1093/nar/29.14.2905
Anolik JH, Klinge CM, Hilf R, Bambara RA (1995) Cooperative binding of estrogen receptor to DNA depends on spacing of binding sites, flanking sequence, and ligand. Biochemistry 34:2511–2520. https://doi.org/10.1021/bi00008a015
Yi P et al (2002) The effects of estrogen-responsive element- and ligand-induced structural changes on the recruitment of cofactors and transcriptional responses by ER alpha and ER beta. Mol Endocrinol 16:674–693. https://doi.org/10.1210/mend.16.4.0810
Peale FV Jr, Ludwig LB, Zain S, Hilf R, Bambara RA (1988) Properties of a high-affinity DNA binding site for estrogen receptor. Proc Natl Acad Sci U S A 85:1038–1042. https://doi.org/10.1073/pnas.85.4.1038
Klein-Hitpass L, Ryffel GU, Heitlinger E, Cato AC (1988) A 13 bp palindrome is a functional estrogen responsive element and interacts specifically with estrogen receptor. Nucleic Acids Res 16:647–663. https://doi.org/10.1093/nar/16.2.647
Driscoll MD et al (1998) Sequence requirements for estrogen receptor binding to estrogen response elements. J Biol Chem 273:29321–29330. https://doi.org/10.1074/jbc.273.45.29321
Palaniappan M et al (2019) The genomic landscape of estrogen receptor α binding sites in mouse mammary gland. PLoS One 14:e0220311–e0220311. https://doi.org/10.1371/journal.pone.0220311
Grober OM et al (2011) Global analysis of estrogen receptor beta binding to breast cancer cell genome reveals an extensive interplay with estrogen receptor alpha for target gene regulation. BMC Genomics 12:36. https://doi.org/10.1186/1471-2164-12-36
Lee H-R, Kim T-H, Choi K-C (2012) Functions and physiological roles of two types of estrogen receptors, ERα and ERβ, identified by estrogen receptor knockout mouse. Lab Anim Res 28:71–76. https://doi.org/10.5625/lar.2012.28.2.71
Chi D et al (2019) Estrogen receptor signaling is reprogrammed during breast tumorigenesis. Proc Natl Acad Sci 116:11437–11443. https://doi.org/10.1073/pnas.1819155116
Hammond ME et al (2010) American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol 28:2784–2795. https://doi.org/10.1200/jco.2009.25.6529
Barnes DM, Harris WH, Smith P, Millis RR, Rubens RD (1996) Immunohistochemical determination of oestrogen receptor: comparison of different methods of assessment of staining and correlation with clinical outcome of breast cancer patients. Br J Cancer 74:1445–1451. https://doi.org/10.1038/bjc.1996.563
Harvey JM, Clark GM, Osborne CK, Allred DC (1999) Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol 17:1474–1474. https://doi.org/10.1200/jco.1999.17.5.1474
Elledge RM et al (2000) Estrogen receptor (ER) and progesterone receptor (PgR), by ligand-binding assay compared with ER, PgR and pS2, by immuno-histochemistry in predicting response to tamoxifen in metastatic breast cancer: A Southwest Oncology Group study. Int J Cancer 89:111–117. https://doi.org/10.1002/(SICI)1097-0215(20000320)89:2<111::AID-IJC2>3.0.CO;2-W
Baum M et al (1983) Improved survival among patients treated with adjuvant tamoxifen after mastectomy for early breast cancer. Lancet (London, England) 2:450. https://doi.org/10.1016/s0140-6736(83)90406-3
(1987) Adjuvant tamoxifen in the management of operable breast cancer: the Scottish trial. Report from the breast cancer trials committee, Scottish cancer trials office (MRC), Edinburgh. Lancet (London, England) 2:171–175
Fisher B et al (2001) Tamoxifen and chemotherapy for axillary node-negative, estrogen receptor-negative breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-23. J Clin Oncol 19:931–942. https://doi.org/10.1200/jco.2001.19.4.931
Mohsin SK et al (2004) Progesterone receptor by immunohistochemistry and clinical outcome in breast cancer: a validation study. Mod Pathol 17:1545–1554. https://doi.org/10.1038/modpathol.3800229
Lindström LS et al (2018) Intratumor heterogeneity of the estrogen receptor and the long-term risk of fatal breast cancer. J Natl Cancer Inst 110:726–733. https://doi.org/10.1093/jnci/djx270
Curigliano G et al (2017) De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen international expert consensus conference on the primary therapy of early breast cancer 2017. Ann Oncol 28:1700–1712. https://doi.org/10.1093/annonc/mdx308
Goldhirsch A et al (2003) Meeting highlights: updated international expert consensus on the primary therapy of early breast cancer. J Clin Oncol 21:3357–3365. https://doi.org/10.1200/jco.2003.04.576
Perou CM et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752. https://doi.org/10.1038/35021093
Sørlie T et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci 98:10869–10874. https://doi.org/10.1073/pnas.191367098
Yanagawa M et al (2012) Luminal A and luminal B (HER2 negative) subtypes of breast cancer consist of a mixture of tumors with different genotype. BMC Res Notes 5:376. https://doi.org/10.1186/1756-0500-5-376
Goldhirsch A et al (2011) Strategies for subtypes--dealing with the diversity of breast cancer: highlights of the St. Gallen international expert consensus on the primary therapy of early breast cancer 2011. Ann Oncol 22:1736–1747. https://doi.org/10.1093/annonc/mdr304
Sparano JA et al (2015) Prospective validation of a 21-gene expression assay in breast cancer. N Engl J Med 373:2005–2014. https://doi.org/10.1056/NEJMoa1510764
Knauer M et al (2010) The predictive value of the 70-gene signature for adjuvant chemotherapy in early breast cancer. Breast Cancer Res Treat 120:655–661. https://doi.org/10.1007/s10549-010-0814-2
Sparano JA et al (2019) Clinical and genomic risk to guide the use of adjuvant therapy for breast cancer. N Engl J Med 380:2395–2405. https://doi.org/10.1056/NEJMoa1904819
Paik S et al (2004) A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. New Eng J Med 351:2817–2826. https://doi.org/10.1056/NEJMoa041588
Sparano JA et al (2018) Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med 379:111–121. https://doi.org/10.1056/NEJMoa1804710
Jeselsohn R et al (2018) Allele-specific chromatin recruitment and therapeutic vulnerabilities of ESR1 activating mutations. Cancer Cell 33:173–186 e175. https://doi.org/10.1016/j.ccell.2018.01.004
Green KA, Carroll JS (2007) Oestrogen-receptor-mediated transcription and the influence of co-factors and chromatin state. Nat Rev Cancer 7:713–722. https://doi.org/10.1038/nrc2211
Siersbæk R, Kumar S, Carroll JS (2018) Signaling pathways and steroid receptors modulating estrogen receptor α function in breast cancer. Genes Dev 32:1141–1154. https://doi.org/10.1101/gad.316646.118
Anbalagan M, Rowan BG (2015) Estrogen receptor alpha phosphorylation and its functional impact in human breast cancer. Mol Cell Endocrinol 418(Pt 3):264–272. https://doi.org/10.1016/j.mce.2015.01.016
Stender JD et al (2017) Structural and molecular mechanisms of cytokine-mediated endocrine resistance in human breast cancer cells. Mol Cell 65:1122–1135.e1125. https://doi.org/10.1016/j.molcel.2017.02.008
Le Goff P, Montano MM, Schodin DJ, Katzenellenbogen BS (1994) Phosphorylation of the human estrogen receptor. Identification of hormone-regulated sites and examination of their influence on transcriptional activity. J Biol Chem 269:4458–4466
Chen D et al (2002) Phosphorylation of human estrogen receptor α at serine 118 by two distinct signal transduction pathways revealed by phosphorylation-specific antisera. Oncogene 21:4921–4931. https://doi.org/10.1038/sj.onc.1205420
Harrod A et al (2017) Genomic modelling of the ESR1 Y537S mutation for evaluating function and new therapeutic approaches for metastatic breast cancer. Oncogene 36:2286–2296. https://doi.org/10.1038/onc.2016.382
Lupien M et al (2010) Growth factor stimulation induces a distinct ER(alpha) cistrome underlying breast cancer endocrine resistance. Genes Dev 24:2219–2227. https://doi.org/10.1101/gad.1944810
Magnani L et al (2015) The pioneer factor PBX1 is a novel driver of metastatic progression in ERα-positive breast cancer. Oncotarget 6:21878–21891. https://doi.org/10.18632/oncotarget.4243
Joel PB et al (1998) pp90rsk1 regulates estrogen receptor-mediated transcription through phosphorylation of Ser-167. Mol Cell Biol 18:1978–1984. https://doi.org/10.1128/mcb.18.4.1978
Becker MA, Ibrahim YH, Cui X, Lee AV, Yee D (2011) The IGF pathway regulates ERα through a S6K1-dependent mechanism in breast cancer cells. Mol Endocrinol 25:516–528. https://doi.org/10.1210/me.2010-0373
Yamnik RL, Holz MK (2010) mTOR/S6K1 and MAPK/RSK signaling pathways coordinately regulate estrogen receptor alpha serine 167 phosphorylation. FEBS Lett 584:124–128. https://doi.org/10.1016/j.febslet.2009.11.041
Michalides R et al (2004) Tamoxifen resistance by a conformational arrest of the estrogen receptor alpha after PKA activation in breast cancer. Cancer Cell 5:597–605. https://doi.org/10.1016/j.ccr.2004.05.016
de Leeuw R et al (2013) PKA phosphorylation redirects ERα to promoters of a unique gene set to induce tamoxifen resistance. Oncogene 32:3543–3551. https://doi.org/10.1038/onc.2012.361
Finak G et al (2008) Stromal gene expression predicts clinical outcome in breast cancer. Nat Med 14:518–527. https://doi.org/10.1038/nm1764
Brechbuhl HM et al (2017) Fibroblast subtypes regulate responsiveness of luminal breast cancer to estrogen. Clin Cancer Res 23:1710–1721. https://doi.org/10.1158/1078-0432.ccr-15-2851
Huang J et al (2017) Downregulation of estrogen receptor and modulation of growth of breast cancer cell lines mediated by paracrine stromal cell signals. Breast Cancer Res Treat 161:229–243. https://doi.org/10.1007/s10549-016-4052-0
Morgan MM et al (2018) Mammary fibroblasts reduce apoptosis and speed estrogen-induced hyperplasia in an organotypic MCF7-derived duct model. Sci Rep 8:7139. https://doi.org/10.1038/s41598-018-25461-1
Madeddu C et al (2014) Role of inflammation and oxidative stress in post-menopausal oestrogen-dependent breast cancer. J Cell Mol Med 18:2519–2529. https://doi.org/10.1111/jcmm.12413
Castellaro AM, Rodriguez-Baili MC, Di Tada CE, Gil GA (2019) Tumor-associated macrophages induce endocrine therapy resistance in ER+ breast cancer cells. Cancers (Basel) 11:189. https://doi.org/10.3390/cancers11020189
Wu L et al (2003) Smad4 as a transcription corepressor for estrogen receptor alpha. J Biol Chem 278:15192–15200. https://doi.org/10.1074/jbc.M212332200
Ren Y et al (2009) Dual effects of TGF-beta on ERalpha-mediated estrogenic transcriptional activity in breast cancer. Mol Cancer 8:111. https://doi.org/10.1186/1476-4598-8-111
Deckers M et al (2006) The tumor suppressor Smad4 is required for transforming growth factor beta-induced epithelial to mesenchymal transition and bone metastasis of breast cancer cells. Cancer Res 66:2202–2209. https://doi.org/10.1158/0008-5472.can-05-3560
Magnani L, Ballantyne EB, Zhang X, Lupien M (2011) PBX1 genomic pioneer function drives ERα signaling underlying progression in breast cancer. PLoS Genet 7:e1002368. https://doi.org/10.1371/journal.pgen.1002368
Theodorou V, Stark R, Menon S, Carroll JS (2013) GATA3 acts upstream of FOXA1 in mediating ESR1 binding by sha** enhancer accessibility. Genome Res 23:12–22. https://doi.org/10.1101/gr.139469.112
Tan SK et al (2011) AP-2γ regulates oestrogen receptor-mediated long-range chromatin interaction and gene transcription. EMBO J 30:2569–2581. https://doi.org/10.1038/emboj.2011.151
Pradhan M, Baumgarten SC, Bembinster LA, Frasor J (2012) CBP mediates NF-κB-dependent histone acetylation and estrogen receptor recruitment to an estrogen response element in the BIRC3 promoter. Mol Cell Biol 32:569–575. https://doi.org/10.1128/MCB.05869-11
Franco HL, Nagari A, Kraus WL (2015) TNFα signaling exposes latent estrogen receptor binding sites to alter the breast cancer cell transcriptome. Mol Cell 58:21–34. https://doi.org/10.1016/j.molcel.2015.02.001
Ross-Innes CS et al (2012) Differential oestrogen receptor binding is associated with clinical outcome in breast cancer. Nature 481:389–393. https://doi.org/10.1038/nature10730
Surveillance E, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence – SEER Research Data, 9 Registries, Nov 2020 Sub (1975–2018) – Linked To County Attributes – Time Dependent (1990–2018) Income/Rurality, 1969–2019 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2021, based on the November 2020 submission
Nathan MR, Schmid P (2017) A review of Fulvestrant in breast cancer. Oncol Ther 5:17–29. https://doi.org/10.1007/s40487-017-0046-2
Brodie A (2002) Aromatase inhibitors in breast cancer. Trends Endocrinol Metab 13:61–65. https://doi.org/10.1016/s1043-2760(01)00529-x
Klein ME, Kovatcheva M, Davis LE, Tap WD, Koff A (2018) CDK4/6 inhibitors: the mechanism of action may not be as simple as once thought. Cancer Cell 34:9–20. https://doi.org/10.1016/j.ccell.2018.03.023
Baselga J et al (2011) Everolimus in postmenopausal hormone-receptor–positive advanced breast cancer. N Engl J Med 366:520–529. https://doi.org/10.1056/NEJMoa1109653
Quirke VM (2017) Tamoxifen from failed contraceptive pill to best-selling breast cancer medicine: a case-study in pharmaceutical innovation. Front Pharmacol 8:620–620. https://doi.org/10.3389/fphar.2017.00620
Jordan VC (2003) Tamoxifen: a most unlikely pioneering medicine. Nat Rev Drug Discov 2:205–213. https://doi.org/10.1038/nrd1031
Smith CL, Nawaz Z, O’Malley BW (1997) Coactivator and corepressor regulation of the agonist/antagonist activity of the mixed antiestrogen, 4-hydroxytamoxifen. Mol Endocrinol 11:657–666. https://doi.org/10.1210/mend.11.6.0009
Sunderland MC, Osborne CK (1991) Tamoxifen in premenopausal patients with metastatic breast cancer: a review. J Clin Oncol 9:1283–1297. https://doi.org/10.1200/jco.1991.9.7.1283
Robertson JF, Harrison M (2004) Fulvestrant: pharmacokinetics and pharmacology. Br J Cancer 90(Suppl 1):S7–S10. https://doi.org/10.1038/sj.bjc.6601630
Musgrove EA, Sutherland RL (2009) Biological determinants of endocrine resistance in breast cancer. Nat Rev Cancer 9:631–643. https://doi.org/10.1038/nrc2713
Hanker AB, Sudhan DR, Arteaga CL (2020) Overcoming endocrine resistance in breast cancer. Cancer Cell 37:496–513. https://doi.org/10.1016/j.ccell.2020.03.009
Finn RS et al (2016) Palbociclib and Letrozole in advanced breast cancer. N Engl J Med 375:1925–1936. https://doi.org/10.1056/NEJMoa1607303
Bertucci F et al (2019) Genomic characterization of metastatic breast cancers. Nature 569:560–564. https://doi.org/10.1038/s41586-019-1056-z
Griffith OL et al (2018) The prognostic effects of somatic mutations in ER-positive breast cancer. Nat Commun 9:3476. https://doi.org/10.1038/s41467-018-05914-x
Angus L et al (2019) The genomic landscape of metastatic breast cancer highlights changes in mutation and signature frequencies. Nat Genet 51:1450–1458. https://doi.org/10.1038/s41588-019-0507-7
Razavi P et al (2018) The genomic landscape of endocrine-resistant advanced breast cancers. Cancer Cell 34:427–438.e426. https://doi.org/10.1016/j.ccell.2018.08.008
Jeselsohn R et al (2014) Emergence of constitutively active estrogen receptor-alpha mutations in pretreated advanced estrogen receptor-positive breast cancer. Clin Cancer Res 20:1757–1767. https://doi.org/10.1158/1078-0432.ccr-13-2332
Patten DK et al (2018) Enhancer map** uncovers phenotypic heterogeneity and evolution in patients with luminal breast cancer. Nat Med 24:1469–1480. https://doi.org/10.1038/s41591-018-0091-x
Hinohara K et al (2018) KDM5 histone demethylase activity links cellular transcriptomic heterogeneity to therapeutic resistance. Cancer Cell 34:939–953.e939. https://doi.org/10.1016/j.ccell.2018.10.014
Gala K et al (2018) KMT2C mediates the estrogen dependence of breast cancer through regulation of ERα enhancer function. Oncogene 37:4692–4710. https://doi.org/10.1038/s41388-018-0273-5
** K et al (2015) HOXB7 is an ERα cofactor in the activation of HER2 and multiple ER target genes leading to endocrine resistance. Cancer Discov 5:944–959. https://doi.org/10.1158/2159-8290.cd-15-0090
Jeselsohn R et al (2017) Embryonic transcription factor SOX9 drives breast cancer endocrine resistance. Proc Natl Acad Sci 114:E4482–E4491. https://doi.org/10.1073/pnas.1620993114
Fu X et al (2016) FOXA1 overexpression mediates endocrine resistance by altering the ER transcriptome and IL-8 expression in ER-positive breast cancer. Proc Natl Acad Sci 113:E6600–E6609. https://doi.org/10.1073/pnas.1612835113
**ao T et al (2018) Estrogen-regulated feedback loop limits the efficacy of estrogen receptor–targeted breast cancer therapy. Proc Natl Acad Sci 115:7869–7878. https://doi.org/10.1073/pnas.1722617115
Hong SP et al (2019) Single-cell transcriptomics reveals multi-step adaptations to endocrine therapy. Nat Commun 10:3840. https://doi.org/10.1038/s41467-019-11721-9
Lei JT et al (2018) Functional annotation of ESR1 gene fusions in estrogen receptor-positive breast cancer. Cell Rep 24:1434–1444.e1437. https://doi.org/10.1016/j.celrep.2018.07.009
Generali D et al (2006) Hypoxia-inducible factor-1alpha expression predicts a poor response to primary chemoendocrine therapy and disease-free survival in primary human breast cancer. Clin Cancer Res 12:4562–4568. https://doi.org/10.1158/1078-0432.ccr-05-2690
Yang J et al (2015) Estrogen receptor-α directly regulates the hypoxia-inducible factor 1 pathway associated with antiestrogen response in breast cancer. Proc Natl Acad Sci U S A 112:15172–15177. https://doi.org/10.1073/pnas.1422015112
Todd VM et al (2021) Hypoxia inducible factor signaling in breast tumors controls spontaneous tumor dissemination in a site-specific manner. Commun Biol 4:1122. https://doi.org/10.1038/s42003-021-02648-3
Houthuijzen JM, Jonkers J (2018) Cancer-associated fibroblasts as key regulators of the breast cancer tumor microenvironment. Cancer Metastasis Rev 37:577–597. https://doi.org/10.1007/s10555-018-9768-3
Louault K et al (2019) Interactions between cancer-associated fibroblasts and tumor cells promote MCL-1 dependency in estrogen receptor-positive breast cancers. Oncogene 38:3261–3273. https://doi.org/10.1038/s41388-018-0635-z
Jallow F et al (2019) Dynamic interactions between the extracellular matrix and estrogen activity in progression of ER+ breast cancer. Oncogene 38:6913–6925. https://doi.org/10.1038/s41388-019-0941-0
Reyes-Ramos AM et al (2021) Collagen I fibrous substrates modulate the proliferation and Secretome of estrogen receptor-positive breast tumor cells in a hormone-restricted microenvironment. ACS Biomater Sci Eng 7:2430–2443. https://doi.org/10.1021/acsbiomaterials.0c01803
Joffroy CM et al (2010) Antiestrogens induce transforming growth factor beta-mediated immunosuppression in breast cancer. Cancer Res 70:1314–1322. https://doi.org/10.1158/0008-5472.can-09-3292
Anurag M et al (2019) Immune checkpoint profiles in luminal B breast cancer (Alliance). J Natl Cancer Inst 112:737–746. https://doi.org/10.1093/jnci/djz213
Karnik PS, Kulkarni S, Liu XP, Budd GT, Bukowski RM (1994) Estrogen receptor mutations in tamoxifen-resistant breast cancer. Cancer Res 54:349–353
Zhang QX, Borg A, Wolf DM, Oesterreich S, Fuqua SA (1997) An estrogen receptor mutant with strong hormone-independent activity from a metastatic breast cancer. Cancer Res 57:1244–1249
Toy W et al (2013) ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. Nat Genet 45:1439–1445. https://doi.org/10.1038/ng.2822
O’Leary B et al (2018) The genetic landscape and clonal evolution of breast cancer resistance to Palbociclib plus Fulvestrant in the PALOMA-3 trial. Cancer Discov 8:1390–1403. https://doi.org/10.1158/2159-8290.cd-18-0264
Koboldt DC et al (2012) Comprehensive molecular portraits of human breast tumours. Nature 490:61–70. https://doi.org/10.1038/nature11412
Jeselsohn R, Buchwalter G, De Angelis C, Brown M, Schiff R (2015) ESR1 mutations-a mechanism for acquired endocrine resistance in breast cancer. Nat Rev Clin Oncol 12:573–583. https://doi.org/10.1038/nrclinonc.2015.117
Dustin D, Gu G, Fuqua SAW (2019) ESR1 mutations in breast cancer. Cancer 125:3714–3728. https://doi.org/10.1002/cncr.32345
Holst F et al (2007) Estrogen receptor alpha (ESR1) gene amplification is frequent in breast cancer. Nat Genet 39:655–660. https://doi.org/10.1038/ng2006
Tomita S et al (2009) Estrogen receptor alpha gene ESR1 amplification may predict endocrine therapy responsiveness in breast cancer patients. Cancer Sci 100:1012–1017. https://doi.org/10.1111/j.1349-7006.2009.01145.x
Reis-Filho JS et al (2008) ESR1 gene amplification in breast cancer: a common phenomenon? Nat Genet 40:809–810; author reply 810-802. https://doi.org/10.1038/ng0708-809b
Ooi A et al (2012) Gene amplification of ESR1 in breast cancers--fact or fiction? A fluorescence in situ hybridization and multiplex ligation-dependent probe amplification study. J Pathol 227:8–16. https://doi.org/10.1002/path.3974
Veeraraghavan J et al (2014) Recurrent ESR1–CCDC170 rearrangements in an aggressive subset of oestrogen receptor-positive breast cancers. Nat Commun 5:4577. https://doi.org/10.1038/ncomms5577
Li S et al (2013) Endocrine-therapy-resistant ESR1 variants revealed by genomic characterization of breast-cancer-derived xenografts. Cell Rep 4:1116–1130. https://doi.org/10.1016/j.celrep.2013.08.022
Robinson DR et al (2013) Activating ESR1 mutations in hormone-resistant metastatic breast cancer. Nat Genet 45:1446–1451. https://doi.org/10.1038/ng.2823
Merenbakh-Lamin K et al (2013) D538G mutation in estrogen receptor-α: a novel mechanism for acquired endocrine resistance in breast cancer. Cancer Res 73:6856–6864. https://doi.org/10.1158/0008-5472.can-13-1197
Carlson KE, Choi I, Gee A, Katzenellenbogen BS, Katzenellenbogen JA (1997) Altered ligand binding properties and enhanced stability of a constitutively active estrogen receptor: evidence that an open pocket conformation is required for ligand interaction. Biochemistry 36:14897–14905. https://doi.org/10.1021/bi971746l
Lazennec G, Ediger TR, Petz LN, Nardulli AM, Katzenellenbogen BS (1997) Mechanistic aspects of estrogen receptor activation probed with constitutively active estrogen receptors: correlations with DNA and coregulator interactions and receptor conformational changes. Mol Endocrinol 11:1375–1386. https://doi.org/10.1210/mend.11.9.9983
Nettles KW et al (2008) NFκB selectivity of estrogen receptor ligands revealed by comparative crystallographic analyses. Nat Chem Biol 4:241–247. https://doi.org/10.1038/nchembio.76
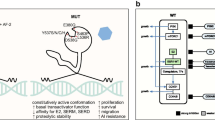
Fanning SW et al (2016) Estrogen receptor alpha somatic mutations Y537S and D538G confer breast cancer endocrine resistance by stabilizing the activating function-2 binding conformation. elife 5. https://doi.org/10.7554/eLife.12792
Bahreini A et al (2017) Mutation site and context dependent effects of ESR1 mutation in genome-edited breast cancer cell models. Breast Cancer Res 19:60. https://doi.org/10.1186/s13058-017-0851-4
Martin L-A et al (2017) Discovery of naturally occurring ESR1 mutations in breast cancer cell lines modelling endocrine resistance. Nat Commun 8:1865. https://doi.org/10.1038/s41467-017-01864-y
Arao Y, Hamilton KJ, Coons LA, Korach KS (2013) Estrogen receptor α L543A,L544A mutation changes antagonists to agonists, correlating with the ligand binding domain dimerization associated with DNA binding activity. J Biol Chem 288:21105–21116. https://doi.org/10.1074/jbc.M113.463455
Katzenellenbogen JA, Mayne CG, Katzenellenbogen BS, Greene GL, Chandarlapaty S (2018) Structural underpinnings of oestrogen receptor mutations in endocrine therapy resistance. Nat Rev Cancer 18:377–388. https://doi.org/10.1038/s41568-018-0001-z
Guan J et al (2019) Therapeutic ligands antagonize estrogen receptor function by impairing its mobility. Cell 178:949–963.e918. https://doi.org/10.1016/j.cell.2019.06.026
Spoerke JM et al (2016) Heterogeneity and clinical significance of ESR1 mutations in ER-positive metastatic breast cancer patients receiving fulvestrant. Nat Commun 7:11579. https://doi.org/10.1038/ncomms11579
Turner NC et al (2020) ESR1 mutations and overall survival on Fulvestrant versus Exemestane in advanced hormone receptor–positive breast cancer: a combined analysis of the phase III SoFEA and EFECT trials. Clin Cancer Res 26:5172–5177. https://doi.org/10.1158/1078-0432.ccr-20-0224
Schiavon G et al (2015) Analysis of ESR1 mutation in circulating tumor DNA demonstrates evolution during therapy for metastatic breast cancer. Sci Transl Med 7:313ra182. https://doi.org/10.1126/scitranslmed.aac7551
Fribbens C et al (2016) Plasma ESR1 mutations and the treatment of estrogen receptor-positive advanced breast cancer. J Clin Oncol 34:2961–2968. https://doi.org/10.1200/jco.2016.67.3061
O’Leary B et al (2018) Early circulating tumor DNA dynamics and clonal selection with palbociclib and fulvestrant for breast cancer. Nat Commun 9:896. https://doi.org/10.1038/s41467-018-03215-x
Krop IE et al (2016) Pictilisib for oestrogen receptor-positive, aromatase inhibitor-resistant, advanced or metastatic breast cancer (FERGI): a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol 17:811–821. https://doi.org/10.1016/s1470-2045(16)00106-6
Chandarlapaty S et al (2016) Prevalence of ESR1 mutations in cell-free DNA and outcomes in metastatic breast cancer: a secondary analysis of the BOLERO-2 clinical trial. JAMA Oncol 2:1310–1315. https://doi.org/10.1001/jamaoncol.2016.1279
Johnston SR et al (2013) Fulvestrant plus anastrozole or placebo versus exemestane alone after progression on non-steroidal aromatase inhibitors in postmenopausal patients with hormone-receptor-positive locally advanced or metastatic breast cancer (SoFEA): a composite, multicentre, phase 3 randomised trial. Lancet Oncol 14:989–998. https://doi.org/10.1016/s1470-2045(13)70322-x
Chia S et al (2008) Double-blind, randomized placebo controlled trial of fulvestrant compared with exemestane after prior nonsteroidal aromatase inhibitor therapy in postmenopausal women with hormone receptor-positive, advanced breast cancer: results from EFECT. J Clin Oncol 26:1664–1670. https://doi.org/10.1200/jco.2007.13.5822
Cristofanilli M et al (2016) Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol 17:425–439. https://doi.org/10.1016/s1470-2045(15)00613-0
Wang T, Wei JJ, Sabatini DM, Lander ES (2014) Genetic screens in human cells using the CRISPR-Cas9 system. Science (New York, NY) 343:80–84. https://doi.org/10.1126/science.1246981
Gojis O et al (2010) The role of SRC-3 in human breast cancer. Nat Rev Clin Oncol 7:83–89. https://doi.org/10.1038/nrclinonc.2009.219
Gates LA et al (2018) Proteomic profiling identifies key coactivators utilized by mutant ERα proteins as potential new therapeutic targets. Oncogene 37:4581–4598. https://doi.org/10.1038/s41388-018-0284-2
Scott GK et al (2016) ERpS294 is a biomarker of ligand or mutational ERα activation and a breast cancer target for CDK2 inhibition. Oncotarget 8:83432–83445. https://doi.org/10.18632/oncotarget.12735
Andruska ND et al (2015) Estrogen receptor α inhibitor activates the unfolded protein response, blocks protein synthesis, and induces tumor regression. Proc Natl Acad Sci U S A 112:4737–4742. https://doi.org/10.1073/pnas.1403685112
Mao C, Livezey M, Kim JE, Shapiro DJ (2016) Antiestrogen resistant cell lines expressing estrogen receptor α mutations upregulate the unfolded protein response and are killed by BHPI. Sci Rep 6:34753. https://doi.org/10.1038/srep34753
Gelsomino L et al (2018) Mutations in the estrogen receptor alpha hormone binding domain promote stem cell phenotype through notch activation in breast cancer cell lines. Cancer Lett 428:12–20. https://doi.org/10.1016/j.canlet.2018.04.023
Ladd B et al (2016) Effective combination therapies in preclinical endocrine resistant breast cancer models harboring ER mutations. Oncotarget 7:54120–54136
Yardley DA et al (2013) Randomized phase II, double-blind, placebo-controlled study of exemestane with or without entinostat in postmenopausal women with locally recurrent or metastatic estrogen receptor-positive breast cancer progressing on treatment with a nonsteroidal aromatase inhibitor. J Clin Oncol 31:2128–2135. https://doi.org/10.1200/jco.2012.43.7251
Connolly RM et al (2021) E2112: randomized phase III trial of endocrine therapy plus Entinostat or placebo in hormone receptor-positive advanced breast cancer. A trial of the ECOG-ACRIN cancer research group. J Clin Oncol 39:3171–3181. https://doi.org/10.1200/jco.21.00944
Waldman AD, Fritz JM, Lenardo MJ (2020) A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol 20:651–668. https://doi.org/10.1038/s41577-020-0306-5
Loi S et al (2013) Prognostic and predictive value of tumor-infiltrating lymphocytes in a phase III randomized adjuvant breast cancer trial in node-positive breast cancer comparing the addition of docetaxel to doxorubicin with doxorubicin-based chemotherapy: BIG 02-98. J Clin Oncol 31:860–867. https://doi.org/10.1200/jco.2011.41.0902
Rugo HS et al (2018) Safety and antitumor activity of Pembrolizumab in patients with estrogen receptor-positive/human epidermal growth factor receptor 2-negative advanced breast cancer. Clin Cancer Res 24:2804–2811. https://doi.org/10.1158/1078-0432.ccr-17-3452
Jeselsohn RM (2021) The evolving use of SERDs in estrogen receptor-positive, HER2-negative metastatic breast cancer. Clin Adv Hematol Oncol H&O 19:428–431
Menarini Group and Radius Health, I (2021) Menarini Group and Radius Health announce positive phase 3 topline results from the EMERALD trial evaluating elacestrant in breast cancer. News release, https://bit.ly/3E0Nmby
Bardia A et al (2019) EMERALD: phase III trial of elacestrant (RAD1901) vs endocrine therapy for previously treated ER+ advanced breast cancer. Future Oncol (London, England) 15:3209–3218. https://doi.org/10.2217/fon-2019-0370
Bardia A et al (2021) Phase I study of Elacestrant (RAD1901), a novel selective estrogen receptor degrader, in ER-positive, HER2-negative advanced breast cancer. J Clin Oncol 39:1360–1370. https://doi.org/10.1200/jco.20.02272
Jhaveri KL et al (2021) Safety and activity of single-agent giredestrant (GDC-9545) from a phase Ia/b study in patients (pts) with estrogen receptor-positive (ER+), HER2-negative locally advanced/metastatic breast cancer (LA/mBC). J Clin Oncol 39:1017–1017. https://doi.org/10.1200/JCO.2021.39.15_suppl.1017
Im S-A et al (2021) SERENA-4: a phase 3 comparison of AZD9833 (camizestrant) plus palbociclib, versus anastrozole plus palbociclib, for patients with ER-positive, HER2-negative advanced breast cancer who have not previously received systemic treatment for advanced disease. J Clin Oncol 39:TPS1101–TPS1101. https://doi.org/10.1200/JCO.2021.39.15_suppl.TPS1101
Jhaveri KL et al (2021) A first-in-human phase 1a/b trial of LY3484356, an oral selective estrogen receptor (ER) degrader (SERD) in ER+ advanced breast cancer (aBC) and endometrial endometrioid cancer (EEC): results from the EMBER study. J Clin Oncol 39:1050–1050. https://doi.org/10.1200/JCO.2021.39.15_suppl.1050
Samatar AA et al (2020) Abstract 4373: discovery of ZN-c5, a novel potent and oral selective estrogen receptor degrader. Cancer Res 80:4373–4373. https://doi.org/10.1158/1538-7445.am2020-4373
Maglakelidze M et al (2021) Rintodestrant (G1T48), an oral selective estrogen receptor degrader, in combination with palbociclib for ER+/HER2– advanced breast cancer: phase 1 results. J Clin Oncol 39:1063–1063. https://doi.org/10.1200/JCO.2021.39.15_suppl.1063
National Library of Medicine (U.S.) (2021) J.-. Phase III study to assess AZD9833+ CDK4/6 inhibitor in HR+/HER2-MBC with detectable ESR1m before progression (SERENA-6). https://ClinicalTrials.gov/show/NCT04964934
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Patel, J.M., Jeselsohn, R.M. (2022). Estrogen Receptor Alpha and ESR1 Mutations in Breast Cancer. In: Campbell, M.J., Bevan, C.L. (eds) Nuclear Receptors in Human Health and Disease. Advances in Experimental Medicine and Biology, vol 1390. Springer, Cham. https://doi.org/10.1007/978-3-031-11836-4_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-11836-4_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-11835-7
Online ISBN: 978-3-031-11836-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)