Abstract
HSP90 action as a conserved chaperone is fundamental to the folding and maturation of a large fraction of structurally and functionally diverse proteins. Through its wide spectrum of clients and their connectivity within the proteome, HSP90 is a key factor in the processes that maintain cellular proteostasis, referred to as the proteostasis network (PN). Besides HSP90, hundreds of additional molecular chaperones and co-chaperones act together in complexes and intricate networks to maintain the cellular native folding environment, avoid misfolding, and redirect non-native folding intermediates back to their native state or towards clearance. The ensemble of all cellular chaperones and co-chaperones constitute the chaperome, which acts to regulate and maintain proteome functionality. Recent insights derived from systematic studies have revealed vast imbalances to cellular proteostasis reflected by chaperome deregulation from aging, to neurodegenerative diseases, and cancers. Deregulation states along with specific chaperome connectivity can represent biomarkers for certain diseases. HSP90 is not acting in isolation but as a component of a comprehensive and dynamic chaperome interactome network. Therefore, a detailed understanding of systems-level alterations of chaperome biology is fundamental towards a better understanding of HSP90 action and its therapeutic targeting in health and disease.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- HSP:
-
Heat shock protein
- HSP90:
-
Heat shock protein 90
- PN:
-
Proteostasis network
References
Ahn YY, Bagrow JP, Lehmann S (2010) Link communities reveal multiscale complexity in networks. Nature 466:761–764
Auluck PK, Chan HY, Trojanowski JQ, Lee VM, Bonini NM (2002) Chaperone suppression of alpha-synuclein toxicity in a Drosophila model for Parkinson’s disease. Science (New York, NY) 295:865–868
Balch WE, Morimoto RI, Dillin A, Kelly JW (2008) Adapting proteostasis for disease intervention. Science (New York, NY) 319:916–919
Balchin D, Hayer-Hartl M, Hartl FU (2016) In vivo aspects of protein folding and quality control. Science (New York, NY) 353:aac4354
Bilen J, Bonini NM (2007) Genome-wide screen for modifiers of ataxin-3 neurodegeneration in Drosophila. PLoS Genet 3:1950–1964
Brehme M, Voisine C (2016) Model systems of protein-misfolding diseases reveal chaperone modifiers of proteotoxicity. Dis Model Mech 9:823–838
Brehme M, Voisine C, Rolland T et al (2014) A chaperome subnetwork safeguards proteostasis in aging and neurodegenerative disease. Cell Rep 9:1135–1150
Brownridge P, Lawless C, Payapilly AB et al (2013) Quantitative analysis of chaperone network throughput in budding yeast. Proteomics 13:1276–1291
Butler EK, Voigt A, Lutz AK et al (2012) The mitochondrial chaperone protein TRAP1 mitigates alpha-Synuclein toxicity. PLoS Genet 8:e1002488
Calamini B, Silva MC, Madoux F et al (2011) Small-molecule proteostasis regulators for protein conformational diseases. Nat Chem Biol 8:185–196
Chan HY, Warrick JM, Andriola I, Merry D, Bonini NM (2002) Genetic modulation of polyglutamine toxicity by protein conjugation pathways in Drosophila. Hum Mol Genet 11:2895–2904
Duennwald ML, Echeverria A, Shorter J (2012) Small heat shock proteins potentiate amyloid dissolution by protein disaggregases from yeast and humans. PLoS Biol 10:e1001346
Fernandez-Funez P, Nino-Rosales ML, de Gouyon B et al (2000) Identification of genes that modify ataxin-1-induced neurodegeneration. Nature 408:101–106
Fonte V, Kapulkin WJ, Taft A, Fluet A, Friedman D, Link CD (2002) Interaction of intracellular beta amyloid peptide with chaperone proteins. Proc Natl Acad Sci U S A 99:9439–9444
Gong Y, Kakihara Y, Krogan N et al (2009) An atlas of chaperone-protein interactions in Saccharomyces cerevisiae: implications to protein folding pathways in the cell. Mol Syst Biol 5:275
Hadizadeh Esfahani A, Sverchkova A, Saez-Rodriguez J, Schuppert AA, Brehme M (2018) A systematic atlas of chaperome deregulation topologies across the human cancer landscape. PLoS Comput Biol 14:e1005890
van Ham TJ, Thijssen KL, Breitling R, Hofstra RM, Plasterk RH, Nollen EA (2008) C. elegans model identifies genetic modifiers of alpha-synuclein inclusion formation during aging. PLoS Genet 4:e1000027
Hartl FU, Hayer-Hartl M (2002) Molecular chaperones in the cytosol: from nascent chain to folded protein. Science (New York, NY) 295:1852–1858
Hartl FU, Bracher A, Hayer-Hartl M (2011) Molecular chaperones in protein folding and proteostasis. Nature 475:324–332
Horwich AL, Fenton WA, Chapman E, Farr GW (2007) Two families of chaperonin: physiology and mechanism. Annu Rev Cell Dev Biol 23:115–145
Jarosz DF, Lindquist S (2010) Hsp90 and environmental stress transform the adaptive value of natural genetic variation. Science (New York, NY) 330:1820–1824
Jimenez-Sanchez M, Lam W, Hannus M et al (2015) siRNA screen identifies QPCT as a druggable target for Huntington’s disease. Nat Chem Biol 11:347–354
Karras GI, Yi S, Sahni N et al (2017) HSP90 shapes the consequences of human genetic variation. Cell 168:856–866.e12
Kazemi-Esfarjani P, Benzer S (2000) Genetic suppression of polyglutamine toxicity in Drosophila. Science (New York, NY) 287:1837–1840
Khabirova E, Moloney A, Marciniak SJ et al (2014) The TRiC/CCT chaperone is implicated in Alzheimer’s disease based on patient GWAS and an RNAi screen in Abeta-expressing Caenorhabditis elegans. PLoS One 9:e102985
Kim YE, Hipp MS, Bracher A, Hayer-Hartl M, Hartl FU (2013) Molecular chaperone functions in protein folding and proteostasis. Annu Rev Biochem 82:323–355
Klaips CL, Jayaraj GG, Hartl FU (2018) Pathways of cellular proteostasis in aging and disease. J Cell Biol 217:51–63
Kleizen B, Braakman I (2004) Protein folding and quality control in the endoplasmic reticulum. Curr Opin Cell Biol 16:343–349
Kraemer BC, Burgess JK, Chen JH, Thomas JH, Schellenberg GD (2006) Molecular pathways that influence human tau-induced pathology in Caenorhabditis elegans. Hum Mol Genet 15:1483–1496
Krobitsch S, Lindquist S (2000) Aggregation of huntingtin in yeast varies with the length of the polyglutamine expansion and the expression of chaperone proteins. Proc Natl Acad Sci U S A 97:1589–1594
Labbadia J, Morimoto RI (2015) The biology of proteostasis in aging and disease. Annu Rev Biochem 84:435–464
Liang J, Clark-Dixon C, Wang S et al (2008) Novel suppressors of alpha-synuclein toxicity identified using yeast. Hum Mol Genet 17:3784–3795
Liberek K, Lewandowska A, Zietkiewicz S (2008) Chaperones in control of protein disaggregation. EMBO J 27:328–335
Luck K, Sheynkman GM, Zhang I, Vidal M (2017) Proteome-scale human interactomics. Trends Biochem Sci 42:342–354
Mayer MP, Bukau B (2005) Hsp70 chaperones: cellular functions and molecular mechanism. Cell Mol Life Sci 62:670–684
Niccoli T, Partridge L (2012) Ageing as a risk factor for disease. Curr Biol 22:R741–R752
Niccoli T, Partridge L, Isaacs AM (2017) Ageing as a risk factor for ALS/FTD. Hum Mol Genet 26:R105–RR13
Nollen EA, Garcia SM, van Haaften G et al (2004) Genome-wide RNA interference screen identifies previously undescribed regulators of polyglutamine aggregation. Proc Natl Acad Sci U S A 101:6403–6408
Oromendia AB, Amon A (2014) Aneuploidy: implications for protein homeostasis and disease. Dis Model Mech 7:15–20
Pearl LH, Prodromou C (2006) Structure and mechanism of the Hsp90 molecular chaperone machinery. Annu Rev Biochem 75:271–294
Phillipson OT (2014) Management of the aging risk factor for Parkinson’s disease. Neurobiol Aging 35:847–857
Powers ET, Morimoto RI, Dillin A, Kelly JW, Balch WE (2009) Biological and chemical approaches to diseases of proteostasis deficiency. Annu Rev Biochem 78:959–991
Queitsch C, Sangster TA, Lindquist S (2002) Hsp90 as a capacitor of phenotypic variation. Nature 417:618–624
Rodina A, Wang T, Yan P et al (2016) The epichaperome is an integrated chaperome network that facilitates tumour survival. Nature 538:397–401
Rolland T, Tasan M, Charloteaux B et al (2014) A proteome-scale map of the human interactome network. Cell 159:1212–1226
Rutherford SL, Lindquist S (1998) Hsp90 as a capacitor for morphological evolution. Nature 396:336–342
Sadlish H, Rampelt H, Shorter J et al (2008) Hsp110 chaperones regulate prion formation and propagation in S. cerevisiae by two discrete activities. PLoS One 3:e1763
Saibil HR (2008) Chaperone machines in action. Curr Opin Struct Biol 18:35–42
Sangster TA, Salathia N, Lee HN et al (2008) HSP90-buffered genetic variation is common in Arabidopsis thaliana. Proc Natl Acad Sci U S A 105:2969–2974
Schmidt M, Finley D (2014) Regulation of proteasome activity in health and disease. Biochim Biophys Acta 1843:13–25
Schopf FH, Biebl MM, Buchner J (2017) The HSP90 chaperone machinery. Nat Rev Mol Cell Biol 18:345–360
Silva MC, Fox S, Beam M, Thakkar H, Amaral MD, Morimoto RI (2011) A genetic screening strategy identifies novel regulators of the proteostasis network. PLoS Genet 7:e1002438
Sun Z, Diaz Z, Fang X et al (2011) Molecular determinants and genetic modifiers of aggregation and toxicity for the ALS disease protein FUS/TLS. PLoS Biol 9:e1000614
Taipale M, Jarosz DF, Lindquist S (2010) HSP90 at the hub of protein homeostasis: emerging mechanistic insights. Nat Rev Mol Cell Biol 11:515–528
Taipale M, Tucker G, Peng J et al (2014) A quantitative chaperone interaction network reveals the architecture of cellular protein homeostasis pathways. Cell 158:434–448
Tatsuta T, Model K, Langer T (2005) Formation of membrane-bound ring complexes by prohibitins in mitochondria. Mol Biol Cell 16:248–259
Valastyan JS, Lindquist S (2014) Mechanisms of protein-folding diseases at a glance. Dis Model Mech 7:9–14
Voisine C, Pedersen JS, Morimoto RI (2010) Chaperone networks: tip** the balance in protein folding diseases. Neurobiol Dis 40:12–20
Vos MJ, Zijlstra MP, Kanon B et al (2010) HSPB7 is the most potent polyQ aggregation suppressor within the HSPB family of molecular chaperones. Hum Mol Genet 19:4677–4693
Walter GM, Smith MC, Wisen S et al (2011) Ordered assembly of heat shock proteins, Hsp26, Hsp70, Hsp90, and Hsp104, on expanded polyglutamine fragments revealed by chemical probes. J Biol Chem 286:40486–40493
Wang Y, Meriin AB, Costello CE, Sherman MY (2007) Characterization of proteins associated with polyglutamine aggregates: a novel approach towards isolation of aggregates from protein conformation disorders. Prion 1:128–135
Wang J, Farr GW, Hall DH et al (2009) An ALS-linked mutant SOD1 produces a locomotor defect associated with aggregation and synaptic dysfunction when expressed in neurons of Caenorhabditis elegans. PLoS Genet 5:e1000350
Warrick JM, Chan HY, Gray-Board GL, Chai Y, Paulson HL, Bonini NM (1999) Suppression of polyglutamine-mediated neurodegeneration in Drosophila by the molecular chaperone HSP70. Nat Genet 23:425–428
Weinstein JN, Collisson EA, Mills GB et al (2013) The cancer genome atlas pan-cancer analysis project. Nat Genet 45:1113–1120
Whitesell L, Lindquist SL (2005) HSP90 and the chaperoning of cancer. Nat Rev Cancer 5:761–772
Young JC, Moarefi I, Hartl FU (2001) Hsp90: a specialized but essential protein-folding tool. J Cell Biol 154:267–273
Zhang S, Binari R, Zhou R, Perrimon N (2010) A genomewide RNA interference screen for modifiers of aggregates formation by mutant huntingtin in Drosophila. Genetics 184:1165–1179
Zhao R, Davey M, Hsu YC et al (2005) Navigating the chaperone network: an integrative map of physical and genetic interactions mediated by the hsp90 chaperone. Cell 120:715–727
Acknowledgements
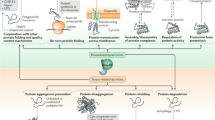
The authors would like to acknowledge Richard Morimoto, Marc Vidal, Andreas Schuppert, Julio Saez-Rodriguez, Angelina Sverchkova, and Ali Hadizadeh-Esfahani for their support and contributions to the authors’ research referenced in this chapter. Figure 27.1 reproduced from Brehme and Voisine, Disease Models & Mechanisms (2016) 9, 823–838, https://doi.org/10.1242/dmm.024703 with permission of Disease Models & Mechanisms.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Voisine, C., Brehme, M. (2019). HSP90 et al.: Chaperome and Proteostasis Deregulation in Human Disease. In: Asea, A., Kaur, P. (eds) Heat Shock Protein 90 in Human Diseases and Disorders. Heat Shock Proteins, vol 19. Springer, Cham. https://doi.org/10.1007/978-3-030-23158-3_27
Download citation
DOI: https://doi.org/10.1007/978-3-030-23158-3_27
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-23157-6
Online ISBN: 978-3-030-23158-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)