Abstract
Background
Cholangiocarcinoma (CCA) is a highly aggressive and fatal tumor. CCA occurs in the epithelial cells of bile ducts. Due to increasing incidences, CCA accounts for 3% of all gastrointestinal malignancies. In addition to comprehensive treatments for cancer, such as surgery, chemotherapy, and radiotherapy, during the past few years, cellular immunotherapy has played an increasingly important role. As a result of our research, we have discovered the γδ T cell-based immunotherapy for CCA.
Case presentation
A 30-year-old male (https://www.clinicaltrials.gov/ ID: NCT02425735) was diagnosed with recurrent mediastinal lymph node metastasis after liver transplantation because of Cholangiocarcinoma (stage IV). In the course of his therapy sessions, he only received allogenic γδ T cell immunotherapy from August, 2017 through February, 2018 (8 infusions in total). γδ T cells were expanded from peripheral blood mononuclear cells (PBMCs) of healthy donor, and ~ 4 × 108 cells were adoptive transferred to the patient.
Conclusion
In the above case report of the Cholangiocarcinoma (stage IV) patient who had received liver transplantation and afterward was diagnosed with recurrent mediastinal lymph node metastasis, we clinically proved that allogenic γδ T cell treatment had no adverse effects. We observed that allogenic γδ T cell treatments positively regulated peripheral immune functions of the patient, depleted tumor activity, improved quality of life, and prolonged his life span. After 8 γδ T cell treatments, the size of lymph nodes was remarkably reduced with activity depletion. This clinical work suggested that allogenic γδ T cell immunotherapy could be developed into a promising therapy drug for CCA.
Similar content being viewed by others
Introduction
Cholangiocarcinoma (CCA) is the most common malignancy of the biliary tree; it may cause fatal consequences in a short period of time [1,2,3]. Currently, the pathogenesis of this disease has not yet been clearly defined, although high-risk factors, such as Primary Sclerosing Cholangitis (PSC), fibrous polycystic liver, intrahepatic bile duct stones, parasitic infections, hepatitis B virus infection, chemical carcinogen exposure, diabetes, and smoking were reported to be probably related to CCA incidences [4, 5]. CCA is highly aggressive and metastatic; statistics have shown an approximate median survival of 24 months [6, 7].
For recurrent CCA, however, the median survival is only 9 months, and the five-year survival is less than 5% [8].
Because of poor efficacy results and prognoses of existing treatments for malignant cancer, the most up-to-date treatments are continually being researched, or under clinical trials. Among new develo** therapeutics, immune cell therapy is emerging as an important alterative for malignant cancer treatment, particularly after the success of CD19 CAR-T [9, 10]. However, for all existing adoptive immune cell therapy, autologous T cells were applied because of MHC restriction. Until present, there have been no reports concerning allogenic T cell applications regarding clinical safety or efficacy. As for γδ T cells, all previous reported works only focused on autologous cells (in vitro or in vivo expansion strategy) as well [11,12,13,14,15,16,17,18,19,20].
In this report, we applied allogenic γδ T cells (Vγ9Vδ2 subsets) as a new type of immune cell therapy to treat CCA. To our knowledge, our work provided the first paradigm on using allogenic γδ T cells to treat cancer. Previously, literatures have demonstrated that γδ T cells are the “first line of defense” as an antitumor effector cell [21, 22], for instance, γδ T cells provide an early source of IFN-γ in the tumor microenvironment [23]. Unlike αβ T cells, γδ T cells recognize antigens in a non-MHC restriction manner. Molecules like LFA, NKG2D, CD16, and others play key roles in γδ T cell recognition and killing of cancer cells. Altogether, γδ T cells could be a promising candidate for cancer immunotherapy [24,25,1: Figures S1 and S2
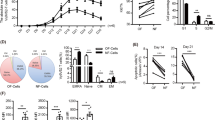
Immunophenotype evaluation
5 mL of peripheral blood was extracted from the patient each time, 1–3 days before receiving Vδ2 T cell treatment. Peripheral blood monocyte cells (PBMC) were isolated using the Ficoll recipe. Then immunofluorescence labeled cells were analyzed using flow cytometry (FACSanto™ II; BD Biosciences, San Jose, CA, USA). The analyzed immune cells mainly included T lymphocytes, NK cells, and γδ T cells.
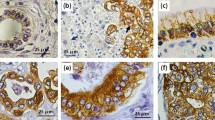
Tumor monitoring by MRI imaging and follow-up
During Vδ2 T cell treatment, tumor was routinely evaluated by using MRI imaging to monitor tumor size/area changes by the largest transverse diameter, particularly before and after treatment. The patient received plain and enhanced MRI 2 weeks before treatment, and then scanned periodically at the 3rd and 6th months after treatment.