Abstract
Background
Although there has been much neurobiological research on major depressive disorder, research on the neurological function of depressive symptoms (DS) or subclinical depression is still scarce, especially in older women with DS.
Objectives
Resting-state functional magnetic resonance imaging (rs-fMRI) was used to compare functional connectivity (FC) between the cerebellum and cerebral in older women with DS and normal controls (NC), to explore unique changes in cerebellar FC in older women with DS.
Methods
In all, 16 older women with DS and 17 NC were recruited. All subjects completed rs-fMRI. The 26 sub-regions of the cerebellum divided by the AAL3 map were used as regions of interest (ROI) to analyze the difference in FC strength of cerebellar seeds from other cerebral regions between the two groups. Finally, partial correlation analysis between abnormal FC strength and Geriatric Depression Scale (GDS) score and Reminiscence Functions Scale (RFS) score in the DS group.
Results
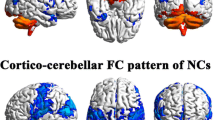
Compared with NC group, the DS group showed significantly reduced FC between Crus I, II and the left frontoparietal region, and reduced FC between Crus I and the left temporal gyrus. Reduced FC between right insula (INS), right rolandic operculum (ROL), right precentral gyrus (PreCG) and the Lobule IX, X. Moreover, the negative FC between Crus I, II, Lobule IX and visual regions was reduced in the DS group. The DS group correlation analysis showed a positive correlation between the left Crus I and the right cuneus (CUN) FC and GDS. In addition, the abnormal FC strength correlated with the scores in different dimensions of the RFS, such as the negative FC between the Crus I and the left middle temporal gyrus (MTG) was positively associated with intimacy maintenance, and so on.
Conclusion
Older women with DS have anomalous FC between the cerebellum and several regions of the cerebrum, which may be related to the neuropathophysiological mechanism of DS in the DS group.
Similar content being viewed by others
Introduction
Depressive symptoms are when a person shows signs of depression, such as sadness, anxiety and despair, but does not yet meet the clinical diagnostic criteria for depression, and are a precursor and transitional state to depression [1].The number of older people with DS is much higher than those who are clinically diagnosed. According to previous studies, the prevalence rate of DS in the older is as high as 37.52%. [63]. This study found that BD, IM and BR scores were higher in the DS group than in the NC group, and the difference in BR scores were statistically significant. BD, IM and BR all belong to the negative memory functions, the common feature is the immersion in repeated meditation on the past and the difficulty in accepting the unresolved events of the past [33]. Consistent with previous research, the results of this study suggest that the DS group is more likely to have negative reminiscences.
Previous research had already confirmed a significant impact of autobiographical memory on late-life depression [64, 65]. An fMRI study demonstrated that as the severity of late-life depressive symptoms increases, the preference for positive memories decreases, and this memory bias affects the depressive symptoms in the elderly [66]. Correlation analyses in this study indicated that the FC strength between the left MTG, right CUN, right IOG right PreCG, and the cerebellum is positively correlated with scores on IM, PD, PS, and TE within reminiscence functions. This involves multiple memory and emotion-related brain areas, suggesting that reminiscence function was related to the mechanism of cognitive emotion regulation in the cerebellum. However, current research mainly focuses on the impact of autobiographical memory on late-life depression, and there is relatively limited investigation into the neural mechanisms of reminiscence functions in depressive symptoms. Further exploration is still needed.
This study still has limitations. first, the sample size of this study is small, which may affect the results of the study, so the sample size should be expanded in subsequent studies. Second, in this study, only GDS scores were used to distinguish DS group and NC group in older women, which should be combined with the evaluation of professional psychologists in future studies.
Conclusion
In conclusion, our study found abnormal functional connectivity between the cerebellum and several cerebral regions in the older women with DS group, involving the frontoparietal network, visual network, right INS, right ROL, right PreCG, and so on. At present, there are few studies on the involvement of cerebellum in DS in older women, this study provides a reference for future research on the neural mechanism of DS in older women.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to privacy and ethical restrictions but are available from the corresponding author on reasonable request.
Abbreviations
- DS:
-
Depressive symptoms
- rs-fMRI:
-
Resting-state functional magnetic resonance imaging
- FC:
-
Functional connectivity
- NC:
-
Normal controls
- ROI:
-
Regions of interest
- GDS:
-
Geriatric Depression Scale
- RFS:
-
Reminiscence Functions Scale
- INS:
-
Insula
- ROL:
-
Rolandic operculum
- PreCG:
-
Precentral gyrus
- CUN:
-
Cuneus
- MTG:
-
Maiddle temporal gyrus
- PS:
-
Problem-solving
- ID:
-
Identity
- DP:
-
Death preparation
- BR:
-
Bitterness revival
- IM:
-
Intimacy maintenance
- TE:
-
Teaching
- BR:
-
Boredom reduction
- BOLD:
-
Blood oxygen level-dependent
- EPI:
-
Echo Planar Imaging
- FOV:
-
Field of view
- MNI:
-
Montreal Neurological Institute
- GRF:
-
Gaussian random field
- IPL:
-
Inferior parietal gyrus, excluding supramarginal and angular gyri
- SFG:
-
Superior frontal gyrus
- LING:
-
Lingual gyrus
- IOG:
-
Inferior occipital gyrus
- PCUN:
-
Precuneus
- ANG:
-
Angular gyrus
- IPG:
-
Inferior parietal gyrus
- SMG:
-
Supramarginal gyrus
- MFG:
-
Middle frontal gyrus
- ITG:
-
Inferior temporal gyrus
- MOG:
-
Middle occipital gyrus
- THA:
-
Thalamus
- DLPFC:
-
Dorsolateral prefrontal cortex
References
Hu Y, Wu X, Chen X, Maguire P, Wang D. Can increased cognitive load help people with subthreshold depression to forget negative information? J Affect Disord. 2021;283:384–94.
**e Y, Ma M, Wu W, Zhang Y, Zhang Y, Tan X. Factors associated with depressive symptoms among the elderly in China: structural equation model. Int Psychogeriatr. 2021;33(2):157–67.
Qiu QW, Qian S, Li JY, Jia RX, Wang YQ, Xu Y. Risk factors for depressive symptoms among older chinese adults: a meta-analysis. J Affect Disord. 2020;277:341–6.
Chan WS, Law CK, Liu KY, Wong PW, Law YW, Yip PS. Suicidality in chinese adolescents in Hong Kong: the role of family and cultural influences. Soc Psychiatry Psychiatr Epidemiol. 2009;44(4):278–84.
Rock PL, Roiser JP, Riedel WJ, Blackwell AD. Cognitive impairment in depression: a systematic review and meta-analysis. Psychol Med. 2014;44(10):2029–40.
Bianco F, Bucciarelli V, Gallina S. The association between cardiovascular diseases and depressive symptoms in adults: the classic chicken or egg causality. Pol Arch Intern Med. 2021;131(6):497–8.
Diniz BS, Reynolds CF 3rd, Butters MA, et al. The effect of gender, age, and symptom severity in late-life depression on the risk of all-cause mortality: the Bambuí Cohort Study of Aging. Depress Anxiety. 2014;31(9):787–95.
Xu LY, Xu FC, Liu C, et al. Relationship between cerebellar structure and emotional memory in depression. Brain Behav. 2017;7(7):e00738.
Gong J, Wang J, Qiu S, et al. Common and distinct patterns of intrinsic brain activity alterations in major depression and bipolar disorder: voxel-based meta-analysis. Transl Psychiatry. 2020;10(1):353.
Zhang L, Sui RB. Proton magnetic resonance spectroscopy study on the metabolism changes of Cerebellum in patients with Post-Stroke Depression. Cell Physiol Biochem. 2017;41(4):1393–402.
Stoodley CJ, Schmahmann JD. Evidence for topographic organization in the cerebellum of motor control versus cognitive and affective processing. Cortex. 2010;46(7):831–44.
Baumann O, Mattingley JB. Functional topography of primary emotion processing in the human cerebellum. NeuroImage. 2012;61(4):805–11.
Adamaszek M, D’Agata F, Ferrucci R, et al. Consensus Paper: Cerebellum and Emotion Cerebellum. 2017;16(2):552–76.
Guell X, Gabrieli JDE, Schmahmann JD. Triple representation of language, working memory, social and emotion processing in the cerebellum: convergent evidence from task and seed-based resting-state fMRI analyses in a single large cohort. NeuroImage. 2018;172:437–49.
Stoodley CJ, Schmahmann JD. Functional topography in the human cerebellum: a meta-analysis of neuroimaging studie. NeuroImage. 2009;44(2):489–501.
Cattaneo Z, Ferrari C, Ciricugno A, et al. New Horizons on non-invasive brain stimulation of the Social and Affective Cerebellum. Cerebellum. 2022;21(3):482–96.
Schienle A, Scharmüller W. Cerebellar activity and connectivity during the experience of disgust and happiness. Neuroscience. 2013;246:375–81.
Ruggiero F, Dini M, Cortese F, et al. Anodal Transcranial Direct Current Stimulation over the Cerebellum enhances sadness recognition in Parkinson’s Disease Patients: a pilot study. Cerebellum. 2022;21(2):234–43.
Ferrari C, Ciricugno A, Urgesi C, Cattaneo Z. Cerebellar contribution to emotional body language perception: a TMS study. Soc Cogn Affect Neurosci. 2019;17(1):81–90.
Ferrari C, Fiori F, Suchan B, Plow EB, Cattaneo Z. TMS over the posterior cerebellum modulates motor cortical excitability in response to facial emotional expressions. Eur J Neurosci. 2021;53(4):1029–39.
E KH, Chen SH, Ho MH, Desmond JE. A meta-analysis of cerebellar contributions to higher cognition from PET and fMRI studies. Hum Brain Mapp. 2014;35(2):593–615.
Lupo M, Troisi E, Chiricozzi FR, Clausi S, Molinari M, Leggio M. Inability to process negative emotions in cerebellar damage: a functional Transcranial Doppler Sonographic Study. Cerebellum. 2015;14(6):663–9.
Gamond L, Ferrari C, La Rocca S, Cattaneo Z. Dorsomedial prefrontal cortex and cerebellar contribution to in-group attitudes: a transcranial magnetic stimulation study. Eur J Neurosci. 2017;45(7):932–9.
Stoodley CJ. The cerebellum and cognition: evidence from functional imaging studies. Cerebellum. 2012;11(2):352–65.
Luo X, Chen G, Jia Y, et al. Disrupted cerebellar Connectivity with the Central Executive Network and the Default-Mode Network in Unmedicated Bipolar II disorder. Front Psychiatry. 2018;9:705.
Guo W, Liu F, Liu J, et al. Increased cerebellar-default-mode-network connectivity in drug-naive major depressive disorder at rest. Med (Baltim). 2015;94(9):e560.
Alalade E, Denny K, Potter G, Steffens D, Wang L. Altered cerebellar-cerebral functional connectivity in geriatric depression. PLoS ONE. 2011;6(5):e20035.
Guo W, Liu F, Xue Z, et al. Abnormal resting-state cerebellar-cerebral functional connectivity in treatment-resistant depression and treatment sensitive depression. Prog Neuropsychopharmacol Biol Psychiatry. 2013;44:51–7.
Ma Q, Zeng LL, Shen H, Liu L, Hu D. Altered cerebellar-cerebral resting-state functional connectivity reliably identifies major depressive disorder. Brain Res. 2013;1495:86–94.
Bluck S, Levine LJ. Reminiscence as autobiographical memory: a catalyst for reminiscence theory development. Ageing Soc. 1998;18(2):185–208.
Webster JD. Construction and validation of the reminiscence functions Scale. J Gerontol. 1993;48(5):256–62.
Cappeliez P, O’Rourke N. Empirical validation of a model of reminiscence and health in later life. J Gerontol B Psychol Sci Soc Sci. 2006;61(4):237–44.
Ros L, Meléndez JC, Webster JD, et al. Reminiscence functions scale: factorial structure and its relation with mental health in a sample of Spanish older adults. Int Psychogeriatr. 2016;28(9):1521–32.
Hofer J, Busch H, Poláčková Šolcová I, Tavel P. When reminiscence is harmful: the relationship between self-negative reminiscence functions, need satisfaction, and DS among older people from Cameroon, the Czech Republic, and Germany. J Happiness Stud. 2017;18:389–407.
McDonald DD, Shellman JM, Graham L, Harrison L. The relationship between reminiscence functions, optimism, depressive symptoms, physical activity, and Pain in older adults. Res Gerontol Nurs. 2016;9(5):223–31.
Korte J, Bohlmeijer ET, Westerhof GJ, Pot AM. Reminiscence and adaptation to critical life events in older adults with mild to moderate DS[J]. Aging Ment Health. 2011;15(5):638–46.
King DB, Cappeliez P, Canham SL, O’Rourke N. Functions of reminiscence in later life: Predicting change in the physical and mental health of older adults over time. Aging Ment Health. 2019;23(2):246–54.
Tam W, Poon SN, Mahendran R, Kua EH, Wu XV. The effectiveness of reminiscence-based intervention on improving psychological well-being in cognitively intact older adults: a systematic review and meta-analysis. Int J Nurs Stud. 2021;114:103847.
Liu Z, Yang F, Lou Y, Zhou W, Tong F. The effectiveness of reminiscence therapy on alleviating depressive symptoms in older adults: a systematic review. Front Psychol. 2021;12:709853.
Yesavage JA, Brink TL, Rose TL, et al. Development and validation of a geriatric depression screening scale: a preliminary report. J Psychiatr Res. 1982;17(1):37–49.
Power JD, Mitra A, Laumann TO, Snyder AZ, Schlaggar BL, Petersen SE. Methods to detect, characterize, and remove motion artifact in resting state fMRI. NeuroImage. 2014;84:320–41.
Rolls ET, Huang CC, Lin CP, Feng J, Joliot M. Automated anatomical labelling atlas 3. NeuroImage. 2020;206:116189.
Zhu D, Yang Y, Zhang Y, et al. Cerebellar-cerebral dynamic functional connectivity alterations in major depressive disorder[J]. J Affect Disord. 2020;275:319–28.
Shinn AK, Roh YS, Ravichandran CT, Baker JT, Öngür D, Cohen BM. Aberrant cerebellar connectivity in bipolar disorder with psychosis. Biol Psychiatry Cogn Neurosci Neuroimaging. 2017;2(5):438–48.
Frodl T, Scheuerecker J, Albrecht J, et al. Neuronal correlates of emotional processing in patients with major depression. World J Biol Psychiatry. 2009;10(3):202–8.
Kennedy DP, Courchesne E. Functional abnormalities of the default network during self- and other-reflection in autism. Soc Cogn Affect Neurosci. 2008;3(2):177–90.
Zeng LL, Shen H, Liu L, et al. Identifying major depression using whole-brain functional connectivity: a multivariate pattern analysis. Brain. 2012;135:1498–507.
Carter RM, Huettel SA. A nexus model of the temporal-parietal junction. Trends Cogn Sci. 2013;17(7):328–36.
Mel’nikov ME, Petrovskii ED, Bezmaternykh DD, et al. fMRI response of parietal brain areas to sad facial stimuli in mild depression. Bull Exp Biol Med. 2018;165(6):741–5.
Chen Y, Wang C, Zhu X, Tan Y, Zhong Y. Aberrant connectivity within the default mode network in first-episode, treatment-naïve major depressive disorder. J Affect Disord. 2015;183:49–56.
Zhang YL, Zhong SM, Lai SK. The brain imaging mechanisms of visual cognitive impairment in depression. Chin J Behav Med Brain Sci. 2019;28(10):951–5.
Song XL, Mu XN, Ding MX, et al. The study of Brain fMRI in Task State of First-Episode patients with mild and moderate depression before and after treatment. J Clin Radiol. 2019;38(7):1174–9.
Teng C, Zhou J, Ma H, et al. Abnormal resting state activity of left middle occipital gyrus and its functional connectivity in female patients with major depressive disorder. BMC Psychiatry. 2018;18(1):370.
Fox MD, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, Raichle ME. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci U S A. 2005;102(27):9673–8.
Yuan Y, Zhang Z, Bai F, et al. Abnormal neural activity in the patients with remitted geriatric depression: a resting-state functional magnetic resonance imaging study. J Affect Disord. 2008;111(2–3):145–52.
Manoliu A, Meng C, Brandl F, et al. Insular dysfunction within the salience network is associated with severity of symptoms and aberrant inter-network connectivity in major depressive disorder[J]. Front Hum Neurosci. 2014;7:930.
Van Overwalle F, Baetens K, Mariën P, Vandekerckhove M. Social cognition and the cerebellum: a meta-analysis of over 350 fMRI studies. NeuroImage. 2014;86:554–72.
Strata P, Scelfo B, Sacchetti B. Involvement of cerebellum in emotional behavior. Physiol Res. 2011;60(Suppl 1):39–48.
Tian J, Zhao L, Liu R, Lu Y2, Huang G, Gong R. Liang F, Gao Y, Zhang W. Functional connectivity of the cerebellar Crus I in type 2 diabetes mellitus: a resting-state functional MRI study. Chinese J Magn Reson Imaging. 2022;13(05):64–9.
Salmi J, Pallesen KJ, Neuvonen T, Brattico E, Korvenoja A, Salonen O, Carlson S. Cognitive and motor loops of the human cerebro-cerebellar system. J Cogn Neurosci. 2010;22(11):2663–76.
Naismith SL, Lagopoulos J, Ward PB, Davey CG, Little C, Hickie IB. Fronto-striatal correlates of impaired implicit sequence learning in major depression: an fMRI study. J Affect Disord. 2010;125(1–3):256–61.
Wu D, Chen T, Yang H, Gong Q, Hu X. Verbal responses, DS, reminiscence functions and cognitive emotion regulation in older women receiving individual reminiscence therapy[J]. J Clin Nurs. 2018;27(13–14):2609–19.
Fernández-Pérez D, Ros L, Escribano F, Serrano JP. Reminiscence, personality, co** and mood state in institutionalised older adults: a cross-sectional study. Psychogeriatrics. 2020;20(3):310–20.
Bahk YC, Choi KH. The relationship between autobiographical memory, cognition, and emotion in older adults: a review. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2018;25(6):874–92.
Köhler CA, Carvalho AF, Alves GS, McIntyre RS, Hyphantis TN, Cammarota M. Autobiographical memory disturbances in Depression: a Novel Therapeutic Target? Neural Plast. 2015;2015:759139.
James TA, Duarte A. Depressive symptoms are associated with reduced positivity preferences in episodic memory in aging. Neurobiol Aging. 2023;121:38–51.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China, China. (Grant No.82001444)
Author information
Authors and Affiliations
Contributions
Lanling Feng and Dongmei Wu involved in all aspects of the study from design, analysis, interpretation of data and preparation of the manuscript. Shaolun Ma and Li Dong involved in the analysis and interpretation of the data.Yuchuan Yue, Tao Li took responsibility for project conceptualization and draft supervision. Guoju Mao,Yixun Tang, Zixiang Ye provided assistances in the acquisition, analysis, or interpretation of data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Sichuan University Ethics Review Board reviewed and approved the study. the study is done in accordance with the Declaration of Helsinki. Each participant provided written informed consent to accomplish this study after a complete description of the protocol. For individuals with a GDS score of ≥ 26 in the screening, it is recommended to seek assessment and treatment from mental health professional institutions.This study was registered in the Chinese Clinical Trial Registry (ChiCTROCC-14004510).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Feng, L., Wu, D., Ma, S. et al. Resting-state functional connectivity of the cerebellum-cerebrum in older women with depressive symptoms. BMC Psychiatry 23, 732 (2023). https://doi.org/10.1186/s12888-023-05232-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12888-023-05232-7