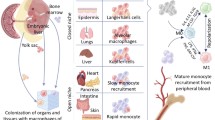
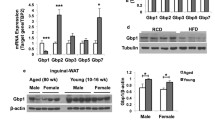
Abstract
Many serious illnesses, including diabetes, obesity, osteoporosis, and neurodegenerative diseases, are caused by chronic inflammation that develops in fat tissue, bones, or the brain. This inflammation occurs due to a shift in macrophage (microglia) polarization toward a proinflammatory M1 phenotype. It has now been proven that macrophage polarization is determined by the intracellular level of autophagy in the macrophage. By modulating autophagy, it is possible to cause a switch in macrophage activity towards M1 or M2. Summarizing the material accumulated in the literature, we believe that activation of autophagy reprograms the macrophage towards M2, replacing its protein content and receptor apparatus, and activate a another type of metabolism. The term “reprogramming” is most appropriate for this process, since it is followed by a change in the functional activity of the macrophage, namely a switch from cytotoxic proinflammatory activity to anti-inflammatory (regenerative) activity. Modulation of autophagy may be an approach to the treatment of cancer, neurodegenerative disorders, osteoporosis, diabetes and other serious diseases.


Similar content being viewed by others
REFERENCES
Agrawal, I. and Jha, S., Mitochondrial dysfunction and Alzheimer’s disease: role of microglia, Front. Aging Neurosci., 2020, vol. 12, p. 252.
Ahmed, B., Sultana, R., and Greene, M.W., Adipose tissue and insulin resistance in obese, Biomed. Pharmacother., 2021, vol. 137, p. 111315. https://doi.org/10.1016/j.biopha.2021.111315
Aoki, S., Shimizu, K., and Ito, K., Autophagy-dependent mitochondrial function regulates osteoclast differentiation and maturation, Biochem. Biophys. Res. Commun., 2020, vol. 527, p. 874.
Azam, S., Haque, M.E., Kim, I.S., and Choi, D.K., Microglial turnover in ageing-related neurodegeneration: therapeutic avenue to intervene in disease progression, Cells, 2021, vol. 10, p. 150.
Blagosklonny, M.V., Progeria, rapamycin and normal aging: recent breakthrough, Aging, 2011, vol. 3, p. 685.
Blagosklonny, M.V., Does rapamycin slow down time?, Oncotarget, 2018, vol. 9, p. 30210. https://doi.org/10.18632/oncotarget.25788
Cao, L. and He, C., Polarization of macrophages and microglia in inflammatory demyelination, Neurosci. Bull., 2013, vol. 29, p. 189.
Carroll, B. and Dunlop, E.A., The lysosome: a crucial hub for AMPK and mTORC1 signalling, Biochem. J., 2017, vol. 474, p. 1453.
Chen, W., Chen, Y., Liu, Y., and Wang, X.J., Autophagy in muscle regeneration: potential therapies for myopathies, Cachexia Sarcopenia Muscle, 2022, vol. 13, p. 1673.
Cheng, J., Liao, Y., Dong, Y., Hu, H., Yang, N., Kong, X., Li, S., Li, X., Guo, J., Qin, L., Yu, J., Ma, C., Li, J., Li, M., Tang, B., and Yuan, Z., Microglial autophagy defect causes parkinson disease-like symptoms by accelerating inflammasome activation in mice, Autophagy, 2020, vol. 16, pp. 2193–2205. https://doi.org/10.1080/15548627.2020.1719723
Chylikova, J., Dvorackova, J., Tauber, Z., and Kamarad, V., M1/M2 macrophage polarization in human obese adipose tissue, Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech. Repub., 2018, vol. 162, p. 79.
Cui, X., Morales, R.T., Qian, W., Wang, H., Gagner, J.P., Dolgalev, I., Placantonakis, D., Zagzag, D., Cimmino, L., Snuderl, M., Lam, R.H.W., and Chen, W., Hacking macrophage-associated immunosuppression for regulating glioblastoma angiogenesis, Biomaterials, 2018, vol. 161, p. 164.
Davies, L.C., Jenkins, S.J., Allen, J.E., and Taylor, P.R., Tissue-resident macrophages, Nat. Immunol., 2013, vol. 14, p. 986.
Fenn, A.M., Henry, C.J., Huang, Y., Dugan, A., and Godbout, J.P., Lipopolysaccharide-induced interleukin (IL)-4 receptor-α expression and corresponding sensitivity to the M2 promoting effects of IL-4 are impaired in microglia of aged mice, Brain Behav. Immunol., 2012, vol. 26, p. 7667.
Florencio-Silva, R., Sasso, G.R., Simões, M.J., Simões, R.S., Baracat, M.C., Sasso-Cerri, E., and Cerri, P.S., Osteoporosis and autophagy: what is the relationship?, Rev. Assoc. Med. Bras., 2017, vol. 63, p. 173.
Fujisaka, S., Usui, I., Nawaz, A., Takikawa, A., Kado, T., Igarashi, Y., and Tobe, K., M2 macrophages in metabolism, Diabetol. Int., 2016, vol. 7, p. 342.
Ghosh, A.K., Mau, T., O’Brien, M., Garg, S., and Yung, R., Impaired autophagy activity is linked to elevated ER-stress and inflammation in aging adipose tissue, Aging (Albany NY), 2016, vol. 8, p. 2525.
Glick, D., Barth, S., and Macleod, K., Autophagy: cellular and molecular mechanisms, J. Pathol., 2010, vol. 221, p. 3.
Green, D.R. and Llambi, F., Cell death signaling, Cold Spring Harb. Perspect. Biol., 2015 vol. 7, p. a006080. https://doi.org/10.1101/cshperspect.a006080
Guo, Y., Lin, C., Xu, P., Wu, S., Fu, X., **a, W., and Yao, M., AGEs induced autophagy impairs cutaneous wound healing via stimulating macrophage polarization to M1 in diabetes, Sci. Rep., 2016, vol. 6, p. 36416. https://doi.org/10.1038/srep36416
Guo, Y., Feng, Y., Cui, X., Wang, Q., and Pan, X., Autophagy inhibition induces the repolarisation of tumour-associated macrophages and enhances chemosensitivity of laryngeal cancer cells to cisplatin in mice, Cancer Immunol. Immunother., 2019, vol. 68, p. 1909.
Han, X., Sun, S., Sun, Y., Song, Q., Zhu, J., Song, N., Chen, M., Sun, T., **a, M., Ding, J., Lu, M., Yao, H., and Hu, G., Small molecule-driven NLRP3 inflammation inhibition via interplay between ubiquitination and autophagy: implications for Parkinson disease, Autophagy, 2019, vol. 15, p. 1860.
Hesketh, M., Sahin, K.B., West, Z.E, and Murray, R.Z., Macrophage phenotypes regulate scar formation and chronic wound healing, Int. J. Mol. Sci., 2017, vol. 18, p. 1545. https://doi.org/10.3390/ijms18071545
Jha, M.K. and Lee, W.H., Functional polarization of neuroglia: Implications in neuroinflammation and neurological disorders, Suk. K. Biochem. Pharmacol., 2016, vol. 103, p. 1.
**, M.M., Wang, F., Qi, D., Liu, W.W., Gu, C., Mao, C.J., Yang, Y.P., Zhao, Z., Hu, L.F., and Liu, C.F., A critical role of autophagy in regulating microglia polarization in neurodegeneration, Front. Aging Neurosci., 2018, vol. 10, p. 378.
Kabeya, Y., Mizushima, N., Ueno, T., Yamamoto, A., Kirisako, T., Noda, T., Kominami, E., Ohsumi, Y., and Yoshimori, T., LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing, EMBO J., 2000, vol. 19, p. 5720.
Kametaka, S., Okano, T., Ohsumi, M., and Ohsumi, Y., Apg14p and Apg6/Vps30p form a protein complex essential for autophagy in the yeast, Saccharomyces cerevisiae, J. Biol. Chem., 1998, vol. 273, p. 22284.
Kang, Y.H., Cho, M.H., Kim, J.Y., Kwon, M.S., Peak, J.J., Kang, S.W., Yoon, S.Y., and Song, Y., Impaired macrophage autophagy induces systemic insulin resistance in obesity, Oncotarget, 2016, vol. 7, p. 35577.
Kapetanovic, R., Bokil, N.J., and Sweet, M.J., Innate immune perturbations, accumulating DAMPs and inflammasome dysregulation: a ticking time bomb in ageing, Ageing Res. Rev., Part A, 2015, vol. 24, p. 40.
Kapoor, N., Niu, J., Saad, Y., Kumar, S., Sirakova, T., Becerra, E., Li, X., and Kolattukudy, P.E., Transcription factors STAT6 and KLF4 implement macrophage polarization via the dual catalytic powers of MCPIP, J. Immunol., 2015, vol. 194, p. 6011.
Kawamata, T., Kamada, Y., Kabeya, Y., Sekito, T., and Ohsumi, Y., Organization of the pre-autophagosomal structure responsible for autophagosome formation, Mol. Biol. Cell, 2008, vol. 19, p. 2039.
Kawano, A., Ariyoshi, W., Yoshioka, Y., Hikiji, H., Nishihara, T., and Okinaga, T., Docosahexaenoic acid enhances M2 macrophage polarization via the p38 signaling pathway and autophagy, J. Cell Biochem., 2019, vol. 120, pp. 12604–12617.
Klionsky, D.J., Abdel-Aziz, A.K., Abdelfatah, S., Abdellatif, M., Abdoli, A., Abel, S., Abeliovich, H., Abild-gaard, M.H., Abudu, Y.P., Acevedo-Arozena, A., Adamopoulos, I.E., Adeli, K., Adolph, T.E., Adornetto, A., Aflaki, E., et al., Guidelines for the Use and Interpretation of Assays for Monitoring Autophagy, 4th ed., Autophagy, vol. 17, p. 1.
Kuo, W.T., Chang, J.M., Chen, C.C., Tsao, N., and Chang, C.P., Autophagy drives plasticity and functional polarization of tumor-associated macrophages, IUBMB Life, 2022, vol. 74, p. 157.
Lee, J.W., Park, S., Takahashi, Y., and Wang, H.G., The association of AMPK with ULK1 regulates autophagy, PLoS One, 2010, vol. 5, p. e15394.
Lee, D.E., Bareja, A., Bartlett, D.B., and White, J.P., Autophagy as a therapeutic target to enhance aged muscle regeneration, Cells, 2019, vol. 8, p. 183.
Liu, K., Zhao, E., Ilyas, G., Lalazar, G., Lin, Y., Haseeb, M., Tanaka, K.E., and Czaja, M.J., Impaired macrophage autophagy increases the immune response in obese mice by promoting proinflammatory macrophage polarization, Autophagy, 2015, vol. 11, p. 271.
Liu, R., Cui, J., Sun, Y., Xu, W., Wang, Z., Wu, M., Dong, H., Yang, C., Hong, S., Yin, S., and Wang, H., Autophagy deficiency promotes M1 macrophage polarization to exacerbate acute liver injury via ATG5 repression during aging, Cell Death Discovery, 2021, vol. 7, p. 397.
Lu, B., Huang, L., Cao, J., Li, L., Wu, W., Chen, X., and Ding, C., Adipose tissue macrophages in aging-associated adipose tissue function, J. Physiol. Sci., 2021, vol. 71, p. 38.
Mauthe, M., Orhon, I., Rocchi, C., Zhou, X., Luhr, M., Hijlkema, K.J., Coppes, R.P., Engedal, N., Mari, M., and Reggiori, F., Chloroquine inhibits autophagic flux by decreasing autophagosome-lysosome fusion, Autophagy, 2018, vol. 14, p. 1435.
Mazher, M., Moqidem, Y.A., Zidan, M., Sayed, A.A., and Abdellatif, A., Autophagic reprogramming of bone marrow-derived macrophages, Immunol. Res., 2023, vol. 71, p. 229.
Metchnikoff, E., Lecons sur la Pathologie Comparee de L’Inflammation, Masson: Paris, 1892.
Miron, V.E., Boyd, A., Zhao, J.W., Yuen, T.J., Ruckh, J.M., Shadrach, J.L., van Wijngaarden, P., Wagers, A.J., Williams, A., Franklin, R.J.M., and Ffrench-Constant, C., M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination, Nat. Neurosci., 2013, vol. 16, p. 1211.
Moehle, M.S. and West, A.B., M1 and M2 immune activation in Parkinson’s disease: foe and ally?, Neuroscience, 2015, vol. 302, p. 59.
Montaseri, A., Giampietri, C., Rossi, M., Riccioli, A., Del Fattore, A., and Filippini, A., The role of autophagy in osteoclast differentiation and bone resorption function, Biomolecules, 2020, vol. 10, p. 1398.
Nawaz, A. and Tobe, K., M2-like macrophages serve as a niche for adipocyte progenitors in adipose tissue, J. Diabetes Invest., 2019, vol. 10, p. 1394.
Nikodemova, M., Small, A.L., Kimyon, R.S., and Watters, J.J., Age-dependent differences in microglial responses to systemic inflammation are evident as early as middle age, Physiol. Genomics, 2016, vol. 48, p. 336.
Nayak, D., Roth, T.L., and McGavern, D.B., Microglia development and function, Annu. Rev. Immunol., 2014, vol. 32, p. 367.
Orihuela, R., McPherson, C.A., and Harry, G.J., Microglial M1/M2 polarization and metabolic states, Br. J. Pharmacol., 2016, vol. 73, p. 649.
Peled, M. and Fisher, E.A., Dynamic aspects of macrophage polarization during atherosclerosis progression and regression, Front. Immunol., 2014, vol. 5, p. 579.
Perandini, L.A., Chimin, P., Lutkemeyer, D.D.S, and Câmara, N.O.S., Chronic inflammation in skeletal muscle impairs satellite cells function during regeneration: can physical exercise restore the satellite cell niche?, FEBS J., 2018, vol. 285, p. 1973.
Pomilio, C., Gorojod, R.M., Riudavets, M., Vinuesa, A., Presa, J., Gregosa, A., Bentivegna, M., Alaimo, A., Alcon, S.P., Sevlever, G., Kotler, M.L., Beauquis, J., and Saravia, F., Microglial autophagy is impaired by prolonged exposure to β-amyloid peptides: evidence from experimental models and Alzheimer’s disease patients, Geroscience, 2020, vol. 42, p. 613.
Rojas, J., Salazar, J., Martínez, M.S., Palmar, J., Bautista, J., Chávez-Castillo, M., Gómez, A., and Bermúdez, V., Macrophage heterogeneity and plasticity: impact of macrophage biomarkers on atherosclerosis, Hindawi Publ. Corporation Scientifica, 2015, p. 851252. https://doi.org/10.1155/2015/851252
Schlundt, C., Fischer, H., Bucher, C.H., Rendenbach, C., Duda, G.N., and Schmidt-Bleek, K., The multifaceted roles of macrophages in bone regeneration: a story of polarization, activation and time, Acta Biomater., 2021, vol. 133, p. 46.
Serý, O., Povová, J., Míšek, I., Pešák, L., and Janout, V., Molecular mechanisms of neuropathological changes in Alzheimer’s disease: a review, Folia Neuropathol., 2013, vol. 51, p. 1.
Singh, L.P., Yumnamcha, T., and Swornalata Devi, T., Mitophagic flux deregulation, lysosomal destabilization and NLRP3 inflammasome activation in diabetic retinopathy: potentials of gene therapy targeting TXNIP and the redox system, Ophthalmol. Res. Rep., 2018, vol. 3, p. ORRT-126.
Stone, A.E.L., Green, R., Wilkins, C., Hemann, E.A., and Gale, M., RIG-I-like receptors direct inflammatory macrophage polarization against West Nile virus infection, J. Nat. Commun., 2019, vol. 10, p. 3649.
Su, T.T., Cellular plasticity, caspases and autophagy; that which does not kill us, well, makes us different, Open Biol., 2018, vol. 8, p. 180157. https://doi.org/10.1098/rsob.180157
Suzuki, K., Akioka, M., Kondo-Kakuta, C., Yamamoto, H., and Ohsumi, Y., Fine map** of autophagy-related proteins during autophagosome formation in Saccharomyces cerevisiae, J. Cell Sci, 2013, vol. 126, p. 2534.
Urwanisch, L., Luciano, M., and Horejs-Hoeck, J., The NLRP3 inflammasome and its role in the pathogenicity of leukemia, Int. J. Mol. Sci., 2021, vol. 22, p. 1271. https://doi.org/10.3390/ijms22031271
Van Eijk, M. and Aerts, J.M.F.G., The unique phenotype of lipid-laden macrophages, Int. J. Mol. Sci., 2021, vol. 22, p. 4039. https://doi.org/10.3390/ijms22084039
Xue, Y., Nie, D., Wang, L.J., Qiu, H.C., Ma, L., Dong, M.X., Tu, W.J., and Zhao, J., Microglial polarization: novel therapeutic strategy against ischemic stroke, Aging Dis., 2021, vol. 12, p. 466.
Yamate, J., Izawa, T., and Kuwamura, M.J., Macrophage pathology in hepatotoxicity, Toxicol. Pathol., 2023, vol. 36, p. 51. https://doi.org/10.1293/tox.2022-0112
Yang, L., **ao, L., Gao, W., Huang, X., Wei, F., Zhang, Q., and **ao, Y., Macrophages at low-inflammatory status improved osteogenesis via autophagy regulation, Tissue Eng., Part A, 2021, p. 021.
Yao, K. and Zhao, Y.F., Aging modulates microglia phenotypes in neuroinflammation of MPTP-PD mice, Exp. Gerontol., 2018, vol. 111, p. 86.
Yuan, Y., Li, L., Zhu, L., Liu, F., Tang, X., Liao, G., Liu, J., Cheng, J., Chen, Y., and Lu, Y., Mesenchymal stem cells elicit macrophages into M2 phenotype via improving transcription factor EB-mediated autophagy to alleviate diabetic nephropathy, Stem Cells, 2020, vol. 38, p. 639.
Zhang, Q. and Sioud, M., Tumor-associated macrophage subsets: sha** polarization and targeting, Int. J. Mol. Sci., 2023, vol. 24, p. 7493. https://doi.org/10.3390/ijms24087493
Zubova, S.G., Suvorova, I.I., and Karpenko, M.N., Macrophage and microglia polarization: focus on autophagy-dependent reprogramming, Front. Biosci. (Schol Ed.), 2022, vol. 14, p. 3. https://doi.org/10.31083/j.fbs1401003
Funding
This work was supported by the Russian Science Foundation (project no. 22-25-20229, https://rscf.ru/project/22-25-20229/) and the St. Petersburg Science Foundation in accordance with agreement dated April 13, 2022, no. 05/2022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This work does not contain any studies involving human and animal subjects.
CONFLICT OF INTEREST
The authors of this work declare that they have no conflicts of interest.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zubova, S.G., Morshneva, A.V. The Role of Autophagy and Macrophage Polarization in the Process of Chronic Inflammation and Regeneration. Cell Tiss. Biol. 18, 244–256 (2024). https://doi.org/10.1134/S1990519X24700184
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X24700184