Abstract
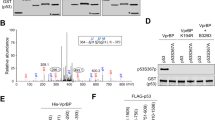
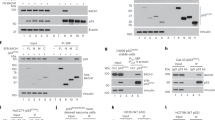
Analyses of five wild-type p53 containing cell lines revealed lineage specific differences in phosphorylation of Thr18 after treatment with ionizing (IR) or ultraviolet (UV) radiation. Importantly, Thr18 phosphorylation correlated with induction of the p53 downstream targets p21Waf1/Cip1 (p21) and Mdm-2, suggesting a transactivation enhancing role. Thr18 phosphorylation has been shown to abolish side-chain hydrogen bonding between Thr18 and Asp21, an interaction necessary for stabilizing alpha–helical conformation within the transactivation domain. Mutagenesis-derived hydrogen bond disruption attenuated the interaction of p53 with the transactivation repressor Mdm-2 but had no direct effect on the interaction of p53 with the basal transcription factor TAFII31. However, prior incubation of p53 mutants with Mdm-2 modulated TAFII31 interaction with p53, suggesting Mdm-2 blocks the accessibility of p53 to TAFII31. Consistently, p53-null cells transfected with hydrogen bond disrupting p53 mutants demonstrated enhanced endogenous p21 expression, whereas p53/Mdm-2-double null cells exhibited no discernible differences in p21 expression. We conclude disruption of intramolecular hydrogen bonding between Thr18 and Asp21 enhances p53 transactivation by modulating Mdm-2 binding, facilitating TAFII31 recruitment.







Similar content being viewed by others
References
Avantaggiati ML, Ogryzko V, Gardner K, Giordano A, Levine AS, Kelly K . 1997 Cell 89: 1175–1184
Banin S, Moyal L, Khostravi R, Shieh SY, Taya Y, Anderson CW, Chessa L, Smorodinsky NI, Prives C, Shiloh Y, Ziv Y . 1998 Science 281: 1674–1677
Bottger V, Bottger A, Garcia-Echeverria C, Ramos YF, van der Eb AJ, Jochemsen AG, Lane DP . 1999 Oncogene 18: 189–199
Bulavin DV, Saito S, Hollander MC, Sakaguchi K, Anderson CW, Appella E, Fornace AJ . 1999 EMBO J. 18: 6845–6854
Canagarajah BJ, Khokhlatchev A, Cobb MH, Goldsmith EJ . 1997 Cell 90: 859–869
Canman DE, Lim DS, Cimprich KA, Taya Y, Tamai K, Sakaguchi K, Appella E, Kastan MB, Siliciano DD . 1998 Science 281: 1677–1679
Chehab NH, Malikzay A, Appel M, Halazonetis TD . 2000 Genes Dev. 14: 278–288
Chehab NH, Malikzay A, Stavridi ES, Halazonetis TD . 1999 Proc. Natl. Acad. Sci. USA 96: 13777–13782
Chen X, Farmer G, Zhu H, Prywes R, Prives C . 1993 Genes Dev. 7: 1837–1849
Choi Y, Asada S, Uesugi M . 2000 J. Biol. Chem. 275: 15912–15916
Collins A . 1987 Int. J. Radia. Biol. Relat. Stud. Phys. Chem. Med. 51: 971–983
Craig AL, Burch L, Vojtesek B, Mikutowska J, Thompson A, Hupp TR . 1999 British J. Biochem. 342: 133–141
Drouin R, Therrien JP . 1997 Photochem. Photobiol. 66: 719–726
Dumaz N, Meek DW . 1999 EMBO J. 24: 7002–7010
Dumaz N, Milne DM, Meek DW . 1999 FEBS Lett. 463: 312–316
de Oca Luna RM, Wagner DS, Lozano G . 1995 Nature 378: 203–206
El-Deiry WS . 1998 Semin. Cancer Biol. 8: 345–357
Farmer G, Colgan J, Nakatani Y, Manley JL, Prives C . 1996 Mol. Cell. Biol. 16: 4295–4303
Feng S, Jensen JP, Ludwig RL, Vousden KH, Weissman AM . 2000 J. Biol. Chem. 275: 8945–8951
Giaccia AJ, Kastan MB . 1998 Genes Dev. 12: 2973–2983
Grossman SR, Perez M, Kung AL, Joseph M, Mansur C, ** Zhang for their helpful assistance and discussion. This work was supported in part by NIH grant CA67987. JR Jabbur was a recipient of the American Legion Auxiliary Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jabbur, J., Tabor, A., Cheng, X. et al. Mdm-2 binding and TAFII31 recruitment is regulated by hydrogen bond disruption between the p53 residues Thr18 and Asp21. Oncogene 21, 7100–7113 (2002). https://doi.org/10.1038/sj.onc.1205856
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205856
- Springer Nature Limited
Keywords
This article is cited by
-
Tumour suppression by p53: a role for the DNA damage response?
Nature Reviews Cancer (2009)
-
Small molecule RITA binds to p53, blocks p53–HDM-2 interaction and activates p53 function in tumors
Nature Medicine (2004)
-
Inhibiting the p53–MDM2 interaction: an important target for cancer therapy
Nature Reviews Cancer (2003)