Abstract
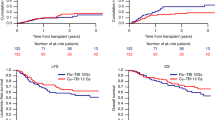
As there is concern regarding the high carcinogenic potential of melphalan (Mel), 725 patients with haematological malignancies who received allogeneic (n = 714) or syngeneic (n = 11) transplants over the last 26 years were followed-up to evaluate if melphalan was more likely to result in secondary malignant neoplasms (SMNs) than cyclophosphamide (Cy). Three hundred and ninety-five were treated with Cy/TBI and 330 with Mel/TBI. Twelve patients developed non-haematological SMN. Median time to develop a SMN was 7 years (range 2–17 years). Age-adjusted rate was significantly higher than in the general population (observed 12 expected 1.2, risk 10; P < 0.0001). the cumulative overall risk of develo** a smn at 2, 5, 10 and 15 years post transplant was 0.4% (95% ci 0.1–2.6%), 1.7% (95% ci 0.6–4.4%), 6.4% (95% ci 2.8–10.8%) and 6.6% (95% ci 3.4–12.4%), respectively. even though age-adjusted rates were higher than the general population melphalan/tbi was not associated with higher age-adjusted risk than cy/tbi (increased risk 7.9 vs 11.4; P = NS). The cumulative overall risk of SMNs was not different with CY/TBI or Mel/TBI (8/393 vs4/363; 10 year risk 4.4%, 95% CI 1.8–10.6 vs 8.4%, 95% CI 2.9–22.9; P = NS). The risk was significantly higher with use of additional cranial or cranio-spinal irradiation (17.5% vs 2.7% at 10 years; P = 0.0241). Transplants for acute lymphatic leukaemia resulted in a higher incidence of SMNs than did transplants for other diseases (ALL: 17.4%, 95% CI 6.3–42.6%; other diseases: 3.4% (95% 1.3–8.5%, P = 0.0469). The risk of SMN for patients with chronic GVHD was 8.4% (95% CI 3.7–18.7%) as compared to 3.5% (95% CI 1–11.1%) for patients without chronic GVHD (P = NS). No factor was associated with independently increased risk in multivariate analysis. Use of melphalan and TBI for transplant conditioning does not appear to be associated with higher risk of second malignant neoplasms than cyclophosphamide and TBI. Bone Marrow Transplantation (2000) 25, 365–370.





Similar content being viewed by others
References
Lowsky R, Lipton J, Fyles G et al. Secondary malignancies after bone marrow transplantation in adults J Clin Oncol 1994 12: 2187–2192
Kolb HJ, Guenther W, Duell T et al. Cancer after bone marrow transplantation. IBMTR and EBMT/EULEP Study Group on Late Effects Bone Marrow Transplant 1992 10: (Suppl. 1) 135–138
Bhatia S, Ramsay NK, Steinbuch M et al. Malignant neoplasms following bone marrow transplantation Blood 1996 87: 3633–3639
Witherspoon RP, Deeg HJ, Storb R et al. Secondary malignancies after marrow transplantation for leukemia or aplastic anemia Transplantation 1994 57: 1413–1418
Traweek ST, Slovak ML, Nademanee AP et al. Myelodysplasia and acute myeloid leukemia occurring after autologous bone marrow transplantation for lymphoma Leuk Lymphoma 1996 20: 365–372
Curtis RE, Rowlings PA, Deeg HJ et al. Solid cancers after bone marrow transplantation New Engl J Med 1997 336: 897–904
Powles RL, Smith CL, Tiley C et al. The anti-leukaemic action of melphalan and total body irradiation in bone marrow transplantation Haematol Rev 1990 4: 23–26
Greene MH, Harris EL, Gershenson DM et al. Melphalan may be more potent leukemogen than cyclophosphamide Ann Intern Med 1996 105: 360–367
Cuzick J, Erskine S, Edelman D, Galton DA . Comparison of incidence of the myelodysplastic syndrome and acute myeloid leukaemia following melphalan and cyclophosphamide treatment for myelomatosis Br J Cancer 1987 55: 523–529
Chou RH, Wong GB, Kramer JH et al. Leukaemia following chemotherapy for breast cancer Cancer Res 1990 50: 2741–2746
Powles R, Milan S, Horton C et al. A prospective haemato-oncology patient database: structure and description Bone Marrow Transplant 1996 17: (Suppl. 1) S140
Helenglass G, Powles RL, McElwain TJ et al. Melphalan and total body irradiation (TBI) versus cyclophosphamide and TBI as conditioning for allogeneic matched sibling bone marrow transplants for acute myeloblastic leukaemia in first remission Bone Marrow Transplant 1988 3: 21–31
Mehta J, Powles R, Treleaven J et al. Long term follow-up of patients undergoing allogeneic bone marrow transplantation for acute myeloid leukaemia in first complete remission after cyclophosphamide-total body irradiation and cyclosporine Bone Marrow Transplant 1996 18: 741–746
Hjiyannakis P, Mehta J, Milan S et al. Melphalan, single-fraction total-body irradiation and allogeneic bone marrow transplantation for acute leukaemia: review of transplant related mortality Leuk Lymphoma 1997 25: 565–572
Powles R, Singhal S, Treleaven J et al. Identification of patients who may benefit from prophylactic immunotherapy after bone marrow transplantation for acute myeloid leukaemia on the basis of lymphocyte recovery early after transplantation Blood 1998 91: 3481–3486
Parkin DM, Whelan SL, Ferlay J et al. (eds). Cancer Incidence in Five Continents, vol. 7. International Agency for Research on Cancer: Lyon, France (IARC scientific publication No. 143)
Kaplan EL, Meier PJ . Non parametric estimation from incomplete observations Am Stat Assoc 1958 53: 457–481
Cox DR . Regression models and life tables J Roy Stat Soc 1972 34B: 187–220
Swift PS, Cowan MJ, Wara WM et al. Toxicities of total body irradiation for pediatric bone marrow transplantation Int J Radiat Oncol Biol Phys 1996 34: 843–851
Kolb HJ, Poetscher C . Late effects after allogeneic bone marrow transplantation Curr Opin Hematol 1997 4: 401–407
Yoshimoto Y . Cancer risk among children of atomic bomb survivors. A review of RERF epidemiologic studies. Radiation Effects Research Foundation JAMA 1990 264: 596–600
Shimizu Y, Schull WJ, Kato H . Cancer risk among atomic bomb survivors. The RERF Life Span Study. Radiation Effects Research Foundation JAMA 1990 264: 601–604
Pierce DA, Shimizu Y, Preston DL et al. Studies of the mortality of atomic bomb survivors. Report 12, Part I. Cancer: 1950–1990 Radiat Res 1996 146: 1–27
Levine EG, Bloomfield CD . Leukemias and myelodysplastic syndromes secondary to drug, radiation, and environmental exposure Semin Oncol 1992 19: 47–84
Saso R, Kulkarni S, Powles R et al. Secondary MDS/AML in patients treated for myeloma Blood 1998 92: (Suppl. 1) 455a
Acknowledgements
This work was supported by Bud Flannagan Leukaemia Fund and David Adams Leukaemia Fund.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kulkarni, S., Powles, R., Treleaven, J. et al. Melphalan/TBI is not more carcinogenic than cyclophosphamide/TBI for transplant conditioning: follow-up of 725 patients from a single centre over a period of 26 years. Bone Marrow Transplant 25, 365–370 (2000). https://doi.org/10.1038/sj.bmt.1702148
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702148
- Springer Nature Limited
Keywords
This article is cited by
-
Single center experience with total body irradiation and melphalan (TBI-MEL) myeloablative conditioning regimen for allogeneic stem cell transplantation (SCT) in patients with refractory hematologic malignancies
Annals of Hematology (2014)
-
Long-term follow-up of secondary malignancies in adults after allogeneic bone marrow transplantation
Bone Marrow Transplantation (2005)
-
Thalidomide after allogeneic haematopoietic stem cell transplantation: activity in chronic but not in acute graft-versus-host disease
Bone Marrow Transplantation (2003)
-
Comparison of new patients with Bence-Jones, IgG and IgA myeloma receiving sequential therapy: the need to regard these immunologic subtypes as separate disease entities with specific prognostic criteria
Bone Marrow Transplantation (2001)