Abstract
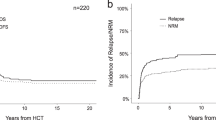
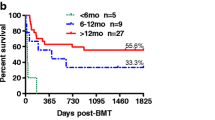
Long-term outcomes after allogeneic hematopoietic cell transplantation (HCT) for therapy-related myeloid neoplasms (tMNs) are dismal. There are few multicenter studies defining prognostic factors in pediatric patients with tMNs. We have accumulated the largest cohort of pediatric patients who have undergone HCT for a tMN to perform a multivariate analysis defining factors predictive of long-term survival. Sixty-eight percent of the 401 patients underwent HCT using a myeloablative conditioning (MAC) regimen, but there were no statistically significant differences in the overall survival (OS), event-free survival (EFS), or cumulative incidence of relapse and non-relapse mortality based on the conditioning intensity. Among the recipients of MAC regimens, 38.4% of deaths were from treatment-related causes, especially acute graft versus host disease (GVHD) and end-organ failure, as compared to only 20.9% of deaths in the reduced-intensity conditioning (RIC) cohort. Exposure to total body irradiation (TBI) during conditioning and experiencing grade III/IV acute GVHD was associated with worse OS. In addition, a diagnosis of therapy-related myelodysplastic syndrome and having a structurally complex karyotype at tMN diagnosis were associated with worse EFS. Reduced-toxicity (but not reduced-intensity) regimens might help to decrease relapse while limiting mortality associated with TBI-based HCT conditioning in pediatric patients with tMNs.





Similar content being viewed by others
References
Davies SM. Subsequent malignant neoplasms in survivors of childhood cancer: childhood cancer survivor study (CCSS) studies. Pediatr Blood Cancer. 2007;48:727–730. https://doi.org/10.1002/pbc.21113
Aguilera DG, Vaklavas C, Tsimberidou AM, Wen S, Medeiros LJ, Corey SJ. Pediatric therapy-related myelodysplastic syndrome/acute myeloid leukemia: the MD Anderson Cancer Center experience. J Pediatr Hematol Oncol. 2009;31:803–11. https://doi.org/10.1097/MPH.0b013e3181ba43dc. e-pub ahead of print 2009/10/06
Bhatia S, Krailo MD, Chen Z, Burden L, Askin FB, Dickman PS, et al. Therapy-related myelodysplasia and acute myeloid leukemia after Ewing sarcoma and primitive neuroectodermal tumor of bone: a report from the Children’s Oncology Group. Blood. 2007;109:46–51. https://doi.org/10.1182/blood-2006-01-023101
Schmiegelow K, Levinsen MF, Attarbaschi A, Baruchel A, Devidas M, Escherich G, et al. Second malignant neoplasms after treatment of childhood acute lymphoblastic leukemia. J Clin Oncol. 2013;31:2469–76. https://doi.org/10.1200/JCO.2012.47.0500
Kayser S, Dohner K, Krauter J, Kohne CH, Horst HA, Held G, et al. The impact of therapy-related acute myeloid leukemia (AML) on outcome in 2853 adult patients with newly diagnosed AML. Blood. 2011;117:2137–45. https://doi.org/10.1182/blood-2010-08-301713. e-pub ahead of print 2010/12/04
Bhatia S. Therapy-related myelodysplasia and acute myeloid leukemia. Semin Oncol. 2013;40:666–75. https://doi.org/10.1053/j.seminoncol.2013.09.013. e-pub ahead of print 2013/12/18
Schoch C, Kern W, Schnittger S, Hiddemann W, Haferlach T. Karyotype is an independent prognostic parameter in therapy-related acute myeloid leukemia (t-AML): an analysis of 93 patients with t-AML in comparison to 1091 patients with de novo AML. Leukemia. 2004;18:120–5. https://doi.org/10.1038/sj.leu.2403187. e-pub ahead of print 2003/10/31
Finke J, Schmoor C, Bertz H, Marks R, Wasch R, Zeiser R, et al. Long-term follow-up of therapy-related myelodysplasia and AML patients treated with allogeneic hematopoietic cell transplantation. Bone Marrow Transpl. 2016;51:771–7. https://doi.org/10.1038/bmt.2015.338. e-pub ahead of print 2016/01/12
Nilsson C, Hulegardh E, Garelius H, Mollgard L, Brune M, Wahlin A, et al. Secondary acute myeloid leukemia and the role of allogeneic stem cell transplantation in a population-based setting. Biol Blood Marrow Transpl. 2019;25:1770–8. https://doi.org/10.1016/j.bbmt.2019.05.038. e-pub ahead of print 2019/06/10
Kroger N, Brand R, van Biezen A, Zander A, Dierlamm J, Niederwieser D, et al. Risk factors for therapy-related myelodysplastic syndrome and acute myeloid leukemia treated with allogeneic stem cell transplantation. Haematologica. 2009;94:542–9. https://doi.org/10.3324/haematol.2008.000927
Litzow MR, Tarima S, Perez WS, Bolwell BJ, Cairo MS, Camitta BM, et al. Allogeneic transplantation for therapy-related myelodysplastic syndrome and acute myeloid leukemia. Blood. 2010;115:1850–7. https://doi.org/10.1182/blood-2009-10-249128
Kida M, Usuki K, Uchida N, Fukuda T, Katayama Y, Kondo T, et al. Outcome and risk factors for therapy-related myeloid neoplasms treated with allogeneic stem cell transplantation in Japan. Biol Blood Marrow Transpl. 2020;26:1543–51. https://doi.org/10.1016/j.bbmt.2020.04.004. e-pub ahead of print 2020/04/23
Brown CA, Youlden DR, Aitken JF, Moore AS. Therapy-related acute myeloid leukemia following treatment for cancer in childhood: a population-based registry study. Pediatr Blood Cancer. 2018;65:e27410 https://doi.org/10.1002/pbc.27410. e-pub ahead of print 2018/09/06
Gassas A, Sivaprakasam P, Cummins M, Breslin P, Patrick K, Slatter M, et al. High transplant-related mortality associated with haematopoietic stem cell transplantation for paediatric therapy-related acute myeloid leukaemia (t-AML). A study on behalf of the United Kingdom Paediatric Blood and Bone Marrow Transplant Group. Bone Marrow Transpl. 2018;53:1165–9. https://doi.org/10.1038/s41409-018-0157-x. e-pub ahead of print 2018/03/17
Imamura T, Taga T, Takagi M, Kawasaki H, Koh K, Taki T, et al. Nationwide survey of therapy-related leukemia in childhood in Japan. Int J Hematol. 2018;108:91–97. https://doi.org/10.1007/s12185-018-2439-x. e-pub ahead of print 2018/03/27
Kobos R, Steinherz PG, Kernan NA, Prockop SE, Scaradavou A, Small TN, et al. Allogeneic hematopoietic stem cell transplantation for pediatric patients with treatment-related myelodysplastic syndrome or acute myelogenous leukemia. Biol Blood Marrow Transpl. 2012;18:473–80. https://doi.org/10.1016/j.bbmt.2011.11.009. e-pub ahead of print 2011/11/15
Woodard P, Barfield R, Hale G, Horwitz E, Leung W, Ribeiro R, et al. Outcome of hematopoietic stem cell transplantation for pediatric patients with therapy-related acute myeloid leukemia or myelodysplastic syndrome. Pediatr Blood Cancer. 2006;47:931–5. https://doi.org/10.1002/pbc.20596
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H et al. WHO classification of tumours of haematopoietic and lymphoid tissue. Lyon, France: International Agency for Research on Cancer; 2017.
Godley LA, Larson RA. Therapy-related myeloid leukemia. Semin Oncol. 2008;35:418–29. https://doi.org/10.1053/j.seminoncol.2008.04.012. e-pub ahead of print 2008/08/12
Lee CJ, Labopin M, Beelen D, Finke J, Blaise D, Ganser A, et al. Comparative outcomes of myeloablative and reduced-intensity conditioning allogeneic hematopoietic cell transplantation for therapy-related acute myeloid leukemia with prior solid tumor: a report from the acute leukemia working party of the European society for blood and bone marrow transplantation. Am J Hematol. 2019;94:431–8. https://doi.org/10.1002/ajh.25395. e-pub ahead of print 2019/01/01
Sengsayadeth S, Gatwood KS, Boumendil A, Labopin M, Finke J, Ganser A, et al. Conditioning intensity in secondary AML with prior myelodysplastic syndrome/myeloproliferative disorders: an EBMT ALWP study. Blood Adv. 2018;2:2127–35. https://doi.org/10.1182/bloodadvances.2018019976. e-pub ahead of print 2018/08/26
Gatwood KS, Labopin M, Savani BN, Finke J, Socie G, Beelen D, et al. Transplant outcomes for patients with therapy-related acute myeloid leukemia with prior lymphoid malignancy: an ALWP of EBMT study. Bone Marrow Transpl. 2020;55:224–32. https://doi.org/10.1038/s41409-019-0673-3. e-pub ahead of print 2019/09/19
Kroger N. Induction, bridging, or straight ahead: the ongoing dilemma of allografting in advanced myelodysplastic syndrome. Biol Blood Marrow Transpl. 2019;25:e247–e249. https://doi.org/10.1016/j.bbmt.2019.06.016. e-pub ahead of print 2019/06/24
McDonald GB, Sandmaier BM, Mielcarek M, Sorror M, Pergam SA, Cheng GS, et al. Survival, nonrelapse mortality, and relapse-related mortality after allogeneic hematopoietic cell transplantation: comparing 2003–2007 versus 2013–2017 cohorts. Ann Intern Med. 2020;172:229–39. https://doi.org/10.7326/M19-2936. e-pub ahead of print 2020/01/21
Scott BL, Pasquini MC, Logan BR, Wu J, Devine SM, Porter DL, et al. Myeloablative versus reduced-intensity hematopoietic cell transplantation for acute myeloid leukemia and myelodysplastic syndromes. J Clin Oncol. 2017;35:1154–61. https://doi.org/10.1200/JCO.2016.70.7091. e-pub ahead of print 2017/04/06
Singhal D, Hahn CN, Feurstein S, Wee LYA, Moma L, Kutyna MM et al. Targeted gene panels identify a high frequency of pathogenic germline variants in patients diagnosed with a hematological malignancy and at least one other independent cancer. Leukemia. 2021. e-pub ahead of print 2021/04/15; https://doi.org/10.1038/s41375-021-01246-w
Gibson CJ, Lindsley RC, Tchekmedyian V, Mar BG, Shi J, Jaiswal S, et al. Clonal hematopoiesis associated with adverse outcomes after autologous stem-cell transplantation for lymphoma. J Clin Oncol. 2017;35:1598–605. https://doi.org/10.1200/JCO.2016.71.6712. e-pub ahead of print 2017/01/10
Wong TN, Ramsingh G, Young AL, Miller CA, Touma W, Welch JS, et al. Role of TP53 mutations in the origin and evolution of therapy-related acute myeloid leukaemia. Nature. 2015;518:552–5. https://doi.org/10.1038/nature13968. e-pub ahead of print 2014/12/10
Berger G, Kroeze LI, Koorenhof-Scheele TN, de Graaf AO, Yoshida K, Ueno H, et al. Early detection and evolution of preleukemic clones in therapy-related myeloid neoplasms following autologous SCT. Blood. 2018;131:1846–57. https://doi.org/10.1182/blood-2017-09-805879. e-pub ahead of print 2018/01/10
Schwartz JR, Ma J, Kamens J, Westover T, Walsh MP, Brady SW, et al. The acquisition of molecular drivers in pediatric therapy-related myeloid neoplasms. Nat Commun. 2021;12:985 https://doi.org/10.1038/s41467-021-21255-8. e-pub ahead of print 2021/02/14
Waanders E, Gu Z, Dobson SM, Antic Z, Crawford JC, Ma X, et al. Mutational landscape and patterns of clonal evolution in relapsed pediatric acute lymphoblastic leukemia. Blood Cancer Discov. 2020;1:96–111. https://doi.org/10.1158/0008-5472.BCD-19-0041. e-pub ahead of print 2020/08/15
Myllymaki M, Redd R, Reilly CR, Saber W, Spellman SR, Gibson CJ, et al. Short telomere length predicts nonrelapse mortality after stem cell transplantation for myelodysplastic syndrome. Blood. 2020;136:3070–81. https://doi.org/10.1182/blood.2020005397. e-pub ahead of print 2020/12/29
Baron F, Labopin M, Savani BN, Beohou E, Niederwieser D, Eder M, et al. Graft-versus-host disease and graft-versus-leukaemia effects in secondary acute myeloid leukaemia: a retrospective, multicentre registry analysis from the Acute Leukaemia Working Party of the EBMT. Br J Haematol. 2020;188:428–37. https://doi.org/10.1111/bjh.16185. e-pub ahead of print 2019/10/16
Kongtim P, Parmar S, Milton DR, Perez JMR, Rondon G, Chen J, et al. Impact of a novel prognostic model, hematopoietic cell transplant-composite risk (HCT-CR), on allogeneic transplant outcomes in patients with acute myeloid leukemia and myelodysplastic syndrome. Bone Marrow Transpl. 2019;54:839–48. https://doi.org/10.1038/s41409-018-0344-9. e-pub ahead of print 2018/09/28
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the disease risk index for allogeneic stem cell transplantation. Blood. 2014;123:3664–71. https://doi.org/10.1182/blood-2014-01-552984. e-pub ahead of print 2014/04/20
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9. https://doi.org/10.1182/blood-2005-05-2004. e-pub ahead of print 2005/07/05
Broglie L, Ruiz J, ** Z, Kahn JM, Bhatia M, George D et al. Limitations of applying the hematopoietic cell transplantation comorbidity index in pediatric patients receiving allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transpl. 2020. e-pub ahead of print 2020/10/12; https://doi.org/10.1016/j.bbmt.2020.10.003
Acknowledgements
We would like to thank Keith A. Laycock, PhD, ELS for the scientific editing of the manuscript. We thank Dr. Stephen Gottschalk for helpful comments on the manuscript. We would like to thank our colleagues, advanced practice providers, nurses, data managers and other healthcare professional who participated in patient care and data collection. We also would like to thank the parents, who entrusted the care of our children to us. This work was supported by the American Society of Hematology (Scholar Award to AS) and the American Lebanese Syrian Associated Charities (ALSAC).
Author information
Authors and Affiliations
Contributions
AS, SH, CC, and BMT designed the study. AS, YL, IA, HBA, PA, AB, NSB, MBB, JB, CB, JEB, AD, J-HD, HE, MF, AG, NJG, WSG, ESG, KH, RH, MPH, JSH, DJ, KAK, EK, SK, AKK, NAK, YPK, CRL, GL-H, PM, KCM, SN, AO-V, TO, MR, SR, KR, HGR, PAR, MES, PJS, JLS, KS, HJS, MdT, ANU, and BV acquired the data and verified it. AS, SH, RJB, CC, and BMT analyzed the data. AS and BMT wrote the manuscript, and YL, IA, HBA, PA, AB, NSB, MBB, JB, CB, JEB, AD, J-HD, HE, MF, AG, NJG, WSG, ESG, KH, RH, MPH, JSH, DJ, KAK, EK, SK, AKK, NAK, YPK, CRL, GL-H, PM, KCM, SN, AO-V, TO, MR, SR, KR, HGR, PAR, MES, PJS, JLS, KS, HJS, MdT, ANU, and BV critically reviewed the manuscript. All authors agree with and take full responsibility for the content of this manuscript.
Corresponding author
Ethics declarations
Competing interests
AS’s institution receives support for the conduct of industry sponsored trials from Vertex Pharmaceuticals, CRISPR Therapeutics and Novartis. AS has received consulting fee from Spotlight Therapeutics and Medexus Inc, and honoraria from Vindico Medical Education. HBA reports that she is an employee of BeiGene Ltd and began her employment after the contribution of any clinical data. RJB reports that he currently is an employee of Smith & Nephew, a position that he transitioned to while this project was ongoing. J-HD reports receiving honoraria from blue bird bio, Orchard, Jazz Pharmaceuticals, Novartis, Sanofi Genzyme and Gilead. MPH serves on the advisory board for Mesoblast. NAK has equity interest in Amgen, Johnson and Johnson, Merck and Pfizer and has received financial support for research from Jazz Pharmaceuticals. HJS reports receiving honoraria from Jazz Pharmaceuticals and has a patent US-2020-0163997-A1 with royalties paid. BMT has received financial support for research and travel from Miltenyi Biotec. Remaining authors do not have any conflicts of interest to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Sharma, A., Huang, S., Li, Y. et al. Outcomes of pediatric patients with therapy-related myeloid neoplasms. Bone Marrow Transplant 56, 2997–3007 (2021). https://doi.org/10.1038/s41409-021-01448-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-021-01448-x
- Springer Nature Limited