Abstract
Despite recent relevant therapeutic progresses, chronic lymphocytic leukemia (CLL) remains an incurable disease. Selinexor, an oral inhibitor of the nuclear export protein XPO1, is active as single agent in different hematologic malignancies, including CLL. The purpose of this study was to evaluate the anti-tumor effects of selinexor, used in combination with chemotherapy drugs (i.e. fludarabine and bendamustine) or with the PI3Kδ inhibitor idelalisib in CLL. Our results showed a significant decrease in CLL cell viability after treatment with selinexor-containing drug combinations compared to each single compound, with demonstration of synergistic cytotoxic effects. Interestingly, this drug synergism was exerted also in the presence of the protective effect of stromal cells. From the molecular standpoint, the synergistic cytotoxic activity of selinexor plus idelalisib was associated with increased regulatory effects of this drug combination on the tumor suppressors FOXO3A and IkBα compared to each single compound. Finally, selinexor was also effective in potentiating the in vivo anti-tumor effects of the PI3Kδ inhibitor in mice treated with the drug combination compared to single agents. Our data provide preclinical evidence of the synergism and potential efficacy of a combination treatment targeting XPO1 and PI3Kδ in CLL.
Similar content being viewed by others
Introduction
Chronic lymphocytic leukemia (CLL) is a very heterogeneous disease, from both the biological and clinical standpoint. Whole-genome sequencing analyses identified recurrent mutations with oncogenic roles1, but a univocal founding lesion has not been identified. Besides single-gene mutations, several signaling and transcriptional pathways implicated in cell survival—such as phosphoinositide 3-kinase/protein kinase B (PI3K/Akt), B-cell receptor (BCR)/BTK and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB)—are aberrantly activated in CLL, also due to the interactions with the protective tumor microenvironment2,3,4,5.
The treatment landscape for CLL has been dramatically changing over the last few years, with the availability of small molecules targeting BTK, PI3K and B-cell lymphoma 2 (Bcl-2)6. However, despite the therapeutic efficacy of new targeted drugs, treatment of specific subgroups of patients, especially those with high-risk features, remains an unmet clinical need, due to the shorter duration of response and the possible development of resistance7,8,9. The simultaneous targeting of different pro-survival pathways may therefore represent an appealing therapeutic strategy, equally effective for multiple CLL subsets.
Exportin 1 (XPO1) is among the best-characterized nuclear exporters in humans, responsible for the translocation of hundreds of different proteins from the nucleus to the cytoplasm through the nuclear pore complex10. Elevated protein levels of XPO1 have been reported in different tumors, especially hematologic malignancies11, leading to an altered regulation and localization of cargo proteins and often correlating with poor prognosis12,13. In CLL cells, XPO1 is overexpressed and recurrently mutated1,11,14. Moreover, many tumor suppressors and growth regulatory proteins with a known pathogenic role in CLL (e.g. cellular tumor antigen p53, cyclin-dependent kinase inhibitor 1 p21, the effector of Akt signal transduction forkhead box O3 [FOXO3A] and IkappaBα [IkBα], the endogenous inhibitor of NF-kB) are recognized by XPO1 cargo proteins15.
Selective inhibitors of nuclear export (SINEs) block XPO1 and their use in vitro and in vivo in a mouse model has confirmed that XPO1 is a valid target in CLL14. Selinexor (KPT-330) is a clinically available, orally administered, SINE which affects CLL cells’ viability also in presence of microenvironment-derived pro-survival signals16, and elicits a synergistic cytotoxic effect with ibrutinib in vitro17. As a single agent, selinexor proved to be safe and showed preliminary anti-cancer activity in phase I studies enrolling patients with relapsed or refractory non-Hodgkin lymphoma, including CLL, acute myeloid leukemia and sarcomas13,18,19. A phase I study also showed the safety and some signs of clinical activity of selinexor in combination with ibrutinib in relapsed or refractory patients with CLL or non-Hodgkin lymphomas20. Currently, selinexor is approved by the FDA for clinical use in patients affected by relapsed/refractory multiple myeloma and has received accelerated approval in diffuse large B-cell lymphoma21,22,23.
The aim of this study was to identify new selinexor-containing drug combinations that display synergistic mechanisms of cytotoxicity and show anti-tumor efficacy in CLL models, for a possible subsequent transition to a clinical-level experimental phase.
Results
Selinexor significantly increases the cytotoxic effect of bendamustine, fludarabine and idelalisib against primary CLL cells
Primary CLL cells from an exploratory cohort of patients were treated with increasing concentrations of bendamustine, fludarabine or idelalisib, used alone or in combination with selinexor. Viability was evaluated at different time points. Based on the obtained results (Supplementary Fig. 1), the most promising doses and timing for each drug combination (i.e. selinexor + bendamustine, selinexor + fludarabine and selinexor + idelalisib) were selected and tested on an expanded cohort of 11 patients (Fig. 1). Overall, the most effective results in terms of cytotoxicity and drug synergism were obtained after 72-h treatment with selinexor + bendamustine and selinexor + fludarabine, and after 24- and 72-h treatment with selinexor + idelalisib. For all time points and concentrations, we also demonstrated the absence of selinexor cytotoxicity towards non-neoplastic CD3+ and CD14+ cells (data not shown). Based on the difference between the viability after combination and single-agent treatment, and on the combination index (CI) results, we further selected specific drug combinations and time points, and extended the analyses to an expanded cohort of primary CLL samples (Fig. 2). We confirmed a significant decrease in CLL cell viability when samples were treated with drug combinations compared to single compounds. We observed a synergistic cytotoxic effect of selinexor 0.1 µM + bendamustine 30 µM and 50 µM, after 72-h culture (Fig. 2a). At the same time point, combination analysis for selinexor + fludarabine showed a strong synergism for selinexor 0.1 µM + fludarabine 1 µM, a synergism for selinexor 0.1 µM + fludarabine 10 µM, and a synergism for selinexor 1 µM + fludarabine 1 and 10 µM (Fig. 2b).
Selinexor (Sel) significantly increases the cytotoxic effect of bendamustine (Ben), fludarabine (F-ara-A) and idelalisib (Ide) against primary CLL cells at multiple concentrations and time points. Heatmaps show the mean viability (% of AnnV-/PI- CLL cells) for 11 samples of patients with CLL that were co-treated in vitro with selinexor + bendamustine, selinexor + fludarabine and selinexor + idelalisib at specific drug concentrations and time points. Cells highlighted with borders indicate the combinations with a CI < 1 (synergistic). Asterisks indicate the combinations which determined a significant reduction in cell viability compared to both single agents, at the corresponding concentration. ****p < 0.0001, ***p < 0.001, **p < 0.01 and *p < 0.05.
The synergism of selinexor (Sel) + bendamustine (Ben), selinexor + fludarabine (F-ara-A) and selinexor + idelalisib (Ide) is confirmed on an expanded sample cohort. CLL cell viability significantly decreased when samples from an expanded patient cohort were treated with the drug combinations compared to single compounds. (a), (b) and (d) represent 72-h viability, whereas panel (c) represents 24-h viability. Box plots represent median value and 25–75% percentiles, whiskers represent minimum and maximum values for each group; each point represents a single sample. CI for all combinations is depicted in tables. Asterisks indicate the combinations which resulted in a significant reduction in cell viability compared to both single agents, at the corresponding concentration. ****p < 0.0001, ***p < 0.001, **p < 0.01 and *p < 0.05.
The combinations selinexor 0.1 µM + idelalisib 0.1 µM, 1 µM and 10 µM resulted synergistic or strongly synergistic both at 24-h and 72-h time points (Fig. 2c,d). Interestingly, in the cohorts of CLL samples that were characterized by in vitro resistance to single-agent bendamustine, fludarabine or idelalisib (i.e. samples showing a normalized cell viability after single-agent-treatment > 0.5, Supplementary Fig. 2), the addition of selinexor was effective in restoring drug-induced cytotoxicity (Fig. 3).
Selinexor (Sel) is effective in restoring drug-induced cytotoxicity. CLL samples were categorized as drug-sensitive or drug-resistant if their normalized cell viability after the specific drug treatment was ≤ or > 0.5, respectively. In the CLL samples characterized by in vitro resistance to bendamustine (Ben) (a,b), fludarabine (F-ara-A) (c,d) or idelalisib (Ide) (e,f), the addition of selinexor significantly increased drug-induced cytotoxicity at the majority of tested concentrations. Box plots represent median value and 25–75% percentiles for 72-h viability, whiskers represent minimum and maximum values for each group; each point represents a single sample. Asterisks indicate the combinations determining a significant reduction in cell viability compared to both single agents, at the corresponding concentration. ****p < 0.0001, ***p < 0.001, **p < 0.01 and *p < 0.05.
Selinexor-based combinations differently affect CLL cell viability in specific molecular subgroups
It has been previously reported that CLL cells characterized by Immunoglobulin Heavy chain Variable region genes (IGHV) mutated (M) status and by the presence of del(17p) are less sensitive to SINE KPT-185 and to selinexor14,16. In line with previous data, our results show that single-agent selinexor is less effective in inducing cytotoxicity towards CLL cells carrying del(17p) and/or TP53 mutations (TP53 disrupted [TP53dis] subset). By contrast, we did not observe a difference in selinexor-induced cytotoxicity between IGHV M and unmutated (UM) CLL cells (Supplementary Fig. 3). When selinexor was used in combination with bendamustine, fludarabine or idelalisib, there was no significant difference in 72-h cell viability based on IGHV mutational status (Supplementary Fig. 4). By contrast, the cytotoxic effect of selinexor in combination with chemotherapy drugs was dependent on the TP53 status. Indeed, TP53dis samples exposed to selinexor + bendamustine, selinexor + fludarabine or selinexor + idelalisib were less susceptible to drug-induced cytotoxicity compared to TP53 wild type (TP53wt) (Supplementary Fig. 5).
Selinexor effectively counteracts the stromal cell-induced resistance of CLL cells to bendamustine, fludarabine and idelalisib
Stromal cells protect CLL cells from spontaneous apoptosis and drug-induced cytotoxicity24,25,26. Previously reported data show that single-agent selinexor exerts a cytotoxic effect toward primary CLL cells also in the presence of the pro-survival effect exerted by stromal cells16,17. Therefore, we next wanted to evaluate the most effective drug combinations for their ability to reverse the protection exerted by stromal cells toward drug-induced cytotoxicity. First, a direct cytotoxic effect of each drug combination toward the stromal cell component was excluded (data not shown). The protective effect of stromal cells was confirmed by the high values of 72-h viability of CLL cells co-cultured with stromal cells, both in the absence and in the presence of selinexor, bendamustine, fludarabine and idelalisib used as single agents. By contrast, most drug combinations significantly reduced the viability of leukemic cells co-cultured with stromal cells, thus confirming their ability to at least partially hamper the protective effect exerted by stromal cells toward drug-induced cytotoxicity (Fig. 4). Interestingly, most of the tested drug combinations also showed synergism in co-culture system, with CI < 1.
Selinexor (Sel) exerts synergistic cytotoxic activity with bendamustine (Ben), fludarabine (F-ara-A) or idelalisib (Ide) towards CLL cells co-cultured with stromal cells. CLL cells were co-cultured with M2-10B4 stromal cells (SC) and exposed to selinexor, bendamustine, fludarabine and idelalisib (Ide), at specified concentrations for 72 h. Box plots represent median value and 25–75% percentiles for viability, whiskers represent minimum and maximum values of each group; each point represents a single sample. CI for all combinations is depicted in tables. Asterisks indicate the combinations which determined a significant reduction in cell viability compared to both single agents, at the corresponding concentration. ****p < 0.0001, ***p < 0.001, **p < 0.01 and *p < 0.05.
The cytotoxicity of the selinexor and idelalisib combination is mediated by IkBα and FOXO3A nuclear retention
To explore the molecular basis of the synergistic cytotoxic effect of selinexor and idelalisib, we investigated the effects of XPO1 inhibition in combination with PI3Kδ inhibition on the nuclear retention of IkBα and FOXO3A, which are intracellular mediators functionally regulated by the PI3K/Akt pathway and are known cargo proteins of XPO1. Of note, for this set of experiments, we only selected the concentrations of idelalisib that exhibited an inhibitory effect on Akt phosphorylation (i.e. 1 and 10 µM, Supplementary Fig. 6). A schematic representation of the role of XPO1, XPO1 inhibition, PI3K pathway activity and PI3K inhibition on the NF-kB inhibitor IkBα and on the tumor suppressor FOXO3A cellular localization is depicted in Supplementary Fig. 714,27,28. After 6-h culture, the combination of selinexor and idelalisib at different concentrations induced a nuclear accumulation of IkBα (Fig. 5a), which was paralleled by a significant decrease in NF-kB activity (Fig. 5b) in primary CLL cells. The nuclear retention of IkBα was confirmed also at the 12-h time point when we observed an even more pronounced inhibition of the transcriptional activity of NF-kB (Fig. 5c,d). Similarly, also FOXO3A nuclear accumulation was promoted after 6-h exposure of CLL cells to the combination selinexor + idelalisib (Fig. 5e). Interestingly, all these molecular effects were not attributable to a reduction in cell viability, which was only negligibly decreased after short-term (i.e. 6-h and 12-h) exposure to the drug combination (Fig. 5f,g).
The effects of the selinexor (Sel) + idelalisib (Ide) combination are mediated by IkBα and FOXO3A. Primary CLL cells were exposed to selinexor and idelalisib at specified concentrations. IkBα nuclear amount (a,c), NF-kB activity (b,d) and cell viability (f,g) were evaluated after 6 and 12 h. FOXO3A nuclear amount (e) was evaluated after 6 h. In (a), (c) and (e) a representative blot (with relative Unique Patient Number, UPN13 and UNP42), together with the corresponding cumulative band intensity data of 7 independent experiments, respectively, is shown. Box and whiskers plots show median values, 25–75% percentiles, and minimum and maximum values for each group. Repositioned gel lanes are indicated by vertical lines. Asterisks indicate the combinations which determined a significant effect compared to both single agents, at the corresponding concentration. ****p < 0.0001, ***p < 0.001, **p < 0.01 and *p < 0.05.
In vivo evaluation of the activity of selinexor in combination with bendamustine or acalisib
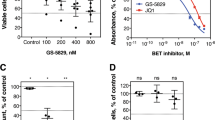
To further corroborate in vitro data and the anti-tumor effect of selinexor-based combinations, we exploited a murine model derived from the engraftment of Eµ-TCL1 leukemic cells into syngeneic mice. In these experiments we tested selinexor in combination with bendamustine and with acalisib—a second generation PI3Kδ inhibitor whose pharmacokinetic profile renders it more suitable than idelalisib for in vivo experiments. After 2-week treatment, the combination of selinexor and bendamustine was effective in reducing the tumor burden in the splenic compartment (Supplementary Fig. 8). In this setting, bendamustine and selinexor, used alone or in combination, induced a decrease in the spleen volume of treated mice when compared to vehicles. However, single-agent bendamustine already exerted a strong anti-tumor effect thus masking the possible beneficial effect of adding selinexor to the treatment regimen. Conversely, selinexor clearly potentiated the anti-tumor activity of acalisib (Fig. 6a–d). In fact, mice treated with selinexor + acalisib showed a significantly lower infiltration by leukemic cells and a significant reduction of the volume of the spleen compared to mice treated with each agent alone.
Selinexor (Sel) in combination with acalisib (Aca) is effective in reducing the tumor burden in vivo. Mice transplanted with the Eµ-TCL1-derived leukemia were randomly treated with selinexor and acalisib, used as single agents or in combination, or with a vehicle. A schematic outline of in vivo experiments is depicted in (a). (b) Shows the percentage of leukemic cells (CD5+/B220+ cells) in the spleen of mice transplanted with the Eµ-TCL1-derived leukemia after 2 weeks of treatment. (c) shows the macroscopic view of the spleens from two representative animals for each treatment group, and (d) shows the spleen volume for all treated animals. In (b) and (d) box and whiskers plots represent median values, 25–75% percentiles, and minimum and maximum values for each group; each point represents a single sample. **p < 0.01 and *p < 0.05.
Discussion
In this study we evaluated the cytotoxic effects of the XPO1 inhibitor selinexor, used in combination with chemotherapy drugs or the PI3Kδ inhibitor idelalisib in CLL. We showed that selinexor significantly increases the cytotoxic effect of fludarabine, bendamustine and idelalisib against primary CLL cells in vitro, exerting synergism at multiple concentrations, also when leukemic cells are cultured in the presence of the protective effect of stromal cells. Selinexor in combination with idelalisib affected NF-kB and PI3K/Akt pathways, and the synergic cytotoxic effect of the combination was mediated by IkBα and FOXO3A nuclear retention. Finally, in vivo, selinexor was able to increase the anti-tumor effect of the PI3Kδ inhibition.
A curative approach for CLL has not yet been identified, and the need to overcome the mechanisms of drug resistance and to achieve better efficacy results constitutes the rationale for investigating novel drug combinations. It has been previously shown that, in cell lines derived from different tumors, selinexor significantly affects the mechanisms of DNA damage repair and synergistically increases the cytotoxicity of chemotherapy agents exerting DNA damaging effects29. Additionally, it has been shown that selinexor inhibits the activation of pathways downstream to BCR16, thus synergizing with the BTK inhibitor ibrutinib in CLL17. This dual blocking of BTK by selinexor plus ibrutinib has already shown to result in promising clinical activity20.
In this paper, we tested selinexor in combination with two different chemotherapy agents used for the treatment of CLL—fludarabine and bendamustine—and also with the targeted PI3Kδ inhibitor idelalisib. PI3K inhibitors—among which the δ subunit inhibitor idelalisib has been the first and most widely tested, also at a clinical level—are another class of drugs that induce the apoptosis of CLL cells through the dual inhibition of the BCR signaling and of the chemokine receptor-dependent activation of Akt, that are key mechanisms in promoting microenvironment-induced survival of leukemic cells2,3. Our data show that selinexor synergizes with both standard chemotherapy agents and idelalisib in affecting primary CLL cells viability in vitro. Of note, selinexor could overcome the intrinsic in vitro resistance of leukemic cells to fludarabine, bendamustine and idelalisib, as shown by the significant reduction of cell viability after exposure of CLL cells to drug combinations containing selinexor compared to each single-agent treatment. Even more interestingly, our combination experiments showed that the addition of selinexor allows to lower the in vitro cytotoxic concentrations of each partner drug (i.e. fludarabine, bendamustine or idelalisib) much below the Cmax achieved with doses currently administered to patients30. From the clinical standpoint, this observation may open the way to the testing of combination regimens containing reduced doses of each drug, thus limiting the potential side effects.
In CLL, drug sensitivity is affected not only by intrinsic features of the leukemic cells, such as the presence of TP53 abnormalities and UM IGHV status, but also by the interactions of neoplastic cells with the surrounding microenvironment26,31,32,33. Interestingly, we observed that selinexor—when used in combination with fludarabine, bendamustine or idelalisib—was capable to partially overcome the protective effect exerted by stromal cells toward the cytotoxicity of each single drug, confirming the synergism of all selinexor-containing combinations also in the co-culture system.
With the idea of pursuing the design of a chemo-free regimen, we next investigated the molecular mechanisms underlying the synergistic activity of the combination selinexor + idelalisib. The PI3K/Akt signaling is an important regulator of both FOXO3A and IkBα tumor suppressors. Akt phosphorylates FOXO3A and leads to its cytoplasmic sequestration and degradation, thus reducing its nuclear accumulation and transcriptional activity28,Animal experiments C57BL/6 Eμ-TCL1 mice were maintained in specific pathogen-free animal facilities and treated in accordance with European Union guidelines, Institutional Animal Care and Use Committee, and ARRIVE guidelines. The in vivo experiments were approved by the Italian Ministry of Health (Authorization n. 47/2018-PR). Splenic leukemic cells from Eμ-TCL1 mice (107 cells) were injected i.p. into syngeneic C57BL/6 mice46,47, and experiments were performed with 4 mice groups. Engrafted mice were randomly assigned to treatment groups: vehicle, selinexor, bendamustine, acalisib, selinexor + bendamustine, selinexor + acalisib. Leukemic mice were treated after 3 or 4 days from the injection. Selinexor (kindly provided by Karyopharm Therapeutics, Newton, MA, USA) was administered at a dose of 7 mg/kg, formulated in Plasdone PVP K29/32 and Poloxamer Pluroniv F-68 (kindly provided by Karyopharm Therapeutics, Newton, MA, USA), three times a week by oral gavage. Bendamustine was administered at a dose of 24 mg/kg, formulated in 1% DMSO, 30% PEG300, 1% Tween 80 and saline solution, once a week i.v. The PI3Kδ inhibitor acalisib (GS-9820, # HY-12644, MedChemExpress, Monmouth Junction, NJ, USA) was used for animal experiments due to its isoform specificity and better pharmacokinetic properties than idelalisib in the mouse48, and was administered at a dose of 25 mg/kg, formulated in in 10% DMSO, 40% PEG300, 5% Tween 80 and saline solution, by daily oral gavage. Mice were euthanized at after 2 weeks of treatment by CO2 inhalation followed by cervical dislocation. The percentage of TCL1 cells was evaluated in PB, bone marrow and spleen compartments using anti-mouse CD45R/B220 PE-Cy7 and anti-mouse CD5 BB515 (#552772 and #565504, BD Biosciences, Allschwil, Switzerland) by flow cytometry (FACS Celesta, BD Biosciences). Spleen volume was also measured. GraphPad Prism (San Diego, CA, USA) was used to perform statistical analyses (using paired t-test or Wilcoxon matched-pairs signed rank test, or unpaired t-test or Mann–Whitney test, based on the data distribution). A p value < 0.05 was considered significant. For synergism analyses, the CI was determined using CompuSyn software (ComboSyn, Inc.). CI values < 0.3, < 1, = 1 and > 1 indicate strong synergism, synergism, additivity and antagonism, respectively.Statistical analysis
Data availability
The data that support the findings of this study are available from the corresponding author, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the corresponding author upon reasonable request and with permission of Karyopharm Therapeutics Inc.
References
Puente, X. S. et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature 475, 101–105. https://doi.org/10.1038/nature10113 (2011).
Herman, S. E. et al. Phosphatidylinositol 3-kinase-delta inhibitor CAL-101 shows promising preclinical activity in chronic lymphocytic leukemia by antagonizing intrinsic and extrinsic cellular survival signals. Blood 116, 2078–2088. https://doi.org/10.1182/blood-2010-02-271171 (2010).
Hoellenriegel, J. et al. The phosphoinositide 3′-kinase delta inhibitor, CAL-101, inhibits B-cell receptor signaling and chemokine networks in chronic lymphocytic leukemia. Blood 118, 3603–3612. https://doi.org/10.1182/blood-2011-05-352492 (2011).
Herishanu, Y. et al. The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia. Blood 117, 563–574. https://doi.org/10.1182/blood-2010-05-284984 (2011).
Herman, S. E. et al. Bruton tyrosine kinase represents a promising therapeutic target for treatment of chronic lymphocytic leukemia and is effectively targeted by PCI-32765. Blood 117, 6287–6296. https://doi.org/10.1182/blood-2011-01-328484 (2011).
Vitale, C. et al. Magic pills: New oral drugs to treat chronic lymphocytic leukemia. Expert Opin. Pharmacother. 18, 411–425. https://doi.org/10.1080/14656566.2017.1293655 (2017).
Woyach, J. A. et al. BTK(C481S)-mediated resistance to ibrutinib in chronic lymphocytic leukemia. J. Clin. Oncol. 35, 1437–1443. https://doi.org/10.1200/jco.2016.70.2282 (2017).
O’Brien, S. et al. Single-agent ibrutinib in treatment-naive and relapsed/refractory chronic lymphocytic leukemia: A 5-year experience. Blood https://doi.org/10.1182/blood-2017-10-810044 (2018).
Blombery, P. et al. Acquisition of the recurrent Gly101Val mutation in BCL2 confers resistance to venetoclax in patients with progressive chronic lymphocytic leukemia. Cancer Discov. 9, 342–353. https://doi.org/10.1158/2159-8290.cd-18-1119 (2019).
Fung, H. Y. & Chook, Y. M. Atomic basis of CRM1-cargo recognition, release and inhibition. Semin. Cancer Biol. 27, 52–61. https://doi.org/10.1016/j.semcancer.2014.03.002 (2014).
Walker, J. S. et al. Recurrent XPO1 mutations alter pathogenesis of chronic lymphocytic leukemia. J. Hematol. Oncol. 14, 17. https://doi.org/10.1186/s13045-021-01032-2 (2021).
Turner, J. G., Dawson, J. & Sullivan, D. M. Nuclear export of proteins and drug resistance in cancer. Biochem. Pharmacol. 83, 1021–1032. https://doi.org/10.1016/j.bcp.2011.12.016 (2012).
Kuruvilla, J. et al. Selective inhibition of nuclear export with selinexor in patients with non-Hodgkin lymphoma. Blood 129, 3175–3183. https://doi.org/10.1182/blood-2016-11-750174 (2017).
Lapalombella, R. et al. Selective inhibitors of nuclear export show that CRM1/XPO1 is a target in chronic lymphocytic leukemia. Blood 120, 4621–4634. https://doi.org/10.1182/blood-2012-05-429506 (2012).
Xu, D., Grishin, N. V. & Chook, Y. M. NESdb: A database of NES-containing CRM1 cargoes. Mol. Biol. Cell 23, 3673–3676. https://doi.org/10.1091/mbc.E12-01-0045 (2012).
Zhong, Y. et al. Selinexor suppresses downstream effectors of B-cell activation, proliferation and migration in chronic lymphocytic leukemia cells. Leukemia 28, 1158–1163. https://doi.org/10.1038/leu.2014.9 (2014).
Hing, Z. A. et al. Selinexor is effective in acquired resistance to ibrutinib and synergizes with ibrutinib in chronic lymphocytic leukemia. Blood 125, 3128–3132. https://doi.org/10.1182/blood-2015-01-621391 (2015).
Gounder, M. M. et al. Phase IB study of selinexor, a first-in-class inhibitor of nuclear export, in patients with advanced refractory bone or soft tissue sarcoma. J. Clin. Oncol. 34, 3166–3174. https://doi.org/10.1200/jco.2016.67.6346 (2016).
Garzon, R. et al. A phase 1 clinical trial of single-agent selinexor in acute myeloid leukemia. Blood 129, 3165–3174. https://doi.org/10.1182/blood-2016-11-750158 (2017).
Stephens, D. M. et al. Selinexor combined with ibrutinib demonstrates tolerability and safety in advanced B-cell malignancies: A phase I study. Clin. Cancer Res. 28, 3242–3247. https://doi.org/10.1158/1078-0432.Ccr-21-3867 (2022).
Chari, A. et al. Oral selinexor-dexamethasone for triple-class refractory multiple myeloma. N. Engl. J. Med. 381, 727–738. https://doi.org/10.1056/NEJMoa1903455 (2019).
Grosicki, S. et al. Once-per-week selinexor, bortezomib, and dexamethasone versus twice-per-week bortezomib and dexamethasone in patients with multiple myeloma (BOSTON): A randomised, open-label, phase 3 trial. Lancet 396, 1563–1573. https://doi.org/10.1016/s0140-6736(20)32292-3 (2020).
Kalakonda, N. et al. Selinexor in patients with relapsed or refractory diffuse large B-cell lymphoma (SADAL): A single-arm, multinational, multicentre, open-label, phase 2 trial. Lancet Haematol. 7, e511–e522. https://doi.org/10.1016/s2352-3026(20)30120-4 (2020).
Niedermeier, M. et al. Isoform-selective phosphoinositide 3′-kinase inhibitors inhibit CXCR4 signaling and overcome stromal cell-mediated drug resistance in chronic lymphocytic leukemia: A novel therapeutic approach. Blood 113, 5549–5557. https://doi.org/10.1182/blood-2008-06-165068 (2009).
Rigoni, M. et al. Simvastatin and downstream inhibitors circumvent constitutive and stromal cell-induced resistance to doxorubicin in IGHV unmutated CLL cells. Oncotarget 6, 29833–29846. https://doi.org/10.18632/oncotarget.4006 (2015).
Vitale, C. et al. Targeting HIF-1α regulatory pathways as a strategy to hamper tumor-microenvironment interactions in CLL. Cancers https://doi.org/10.3390/cancers13122883 (2021).
Zhang, X., Tang, N., Hadden, T. J. & Rishi, A. K. Akt, FoxO and regulation of apoptosis. Biochim. Biophys. Acta 1978–1986, 2011. https://doi.org/10.1016/j.bbamcr.2011.03.010 (1813).
Kapoor, I. et al. Resistance to BTK inhibition by ibrutinib can be overcome by preventing FOXO3a nuclear export and PI3K/AKT activation in B-cell lymphoid malignancies. Cell Death Dis. 10, 924. https://doi.org/10.1038/s41419-019-2158-0 (2019).
Kashyap, T. et al. Selinexor reduces the expression of DNA damage repair proteins and sensitizes cancer cells to DNA damaging agents. Oncotarget 9, 30773–30786. https://doi.org/10.18632/oncotarget.25637 (2018).
Liston, D. R. & Davis, M. Clinically relevant concentrations of anticancer drugs: A guide for nonclinical studies. Clin. Cancer Res. 23, 3489–3498. https://doi.org/10.1158/1078-0432.ccr-16-3083 (2017).
Burger, J. A., Ghia, P., Rosenwald, A. & Caligaris-Cappio, F. The microenvironment in mature B-cell malignancies: A target for new treatment strategies. Blood 114, 3367–3375. https://doi.org/10.1182/blood-2009-06-225326 (2009).
Griggio, V. et al. HIF-1alpha is over-expressed in leukemic cells from TP53-disrupted patients and is a promising therapeutic target in chronic lymphocytic leukemia. Haematologica 105, 1042–1054. https://doi.org/10.3324/haematol.2019.217430 (2020).
Coscia, M. et al. IGHV unmutated CLL B cells are more prone to spontaneous apoptosis and subject to environmental prosurvival signals than mutated CLL B cells. Leukemia 25, 828–837. https://doi.org/10.1038/leu.2011.12 (2011).
**e, M. et al. Akt2 mediates glucocorticoid resistance in lymphoid malignancies through FoxO3a/Bim axis and serves as a direct target for resistance reversal. Cell Death Dis. 9, 1013. https://doi.org/10.1038/s41419-018-1043-6 (2019).
Obrador-Hevia, A., Serra-Sitjar, M., Rodríguez, J., Villalonga, P. & Fernández de Mattos, S. The tumour suppressor FOXO3 is a key regulator of mantle cell lymphoma proliferation and survival. Br. J. Haematol. 156, 334–345. https://doi.org/10.1111/j.1365-2141.2011.08951.x (2012).
Dan, H. C. et al. Akt-dependent regulation of NF-{kappa}B is controlled by mTOR and Raptor in association with IKK. Genes Dev. 22, 1490–1500. https://doi.org/10.1101/gad.1662308 (2008).
Xu, Z. et al. Prognostic value and therapeutic targeting of XPO1 in chronic lymphocytic leukemia. Clin. Exp. Med. https://doi.org/10.1007/s10238-023-01003-6 (2023).
U.S. Food and Drug Administration. Phosphatidylinositol 3-Kinase (PI3K) Inhibitors in Hematologic Malignancies - Oncologic Drugs Advisory Committee Meeting. https://www.fda.gov/media/157762/download (2022).
Furman, R. R. et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N. Engl. J. Med. 370, 997–1007. https://doi.org/10.1056/NEJMoa1315226 (2014).
Gopal, A. K. et al. PI3Kδ inhibition by idelalisib in patients with relapsed indolent lymphoma. N. Engl. J. Med. 370, 1008–1018. https://doi.org/10.1056/NEJMoa1314583 (2014).
Dreyling, M. et al. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann. Oncol. 28, 2169–2178. https://doi.org/10.1093/annonc/mdx289 (2017).
Flinn, I. W. et al. The phase 3 DUO trial: Duvelisib vs ofatumumab in relapsed and refractory CLL/SLL. Blood 132, 2446–2455. https://doi.org/10.1182/blood-2018-05-850461 (2018).
Flinn, I. W. et al. DYNAMO: A phase II study of duvelisib (IPI-145) in patients with refractory indolent non-hodgkin lymphoma. J. Clin. Oncol. 37, 912–922. https://doi.org/10.1200/jco.18.00915 (2019).
Hallek, M. et al. iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood 131, 2745–2760. https://doi.org/10.1182/blood-2017-09-806398 (2018).
Ghia, P. et al. ERIC recommendations on IGHV gene mutational status analysis in chronic lymphocytic leukemia. Leukemia 21, 1–3. https://doi.org/10.1038/sj.leu.2404457 (2007).
Johnson, A. J. et al. Characterization of the TCL-1 transgenic mouse as a preclinical drug development tool for human chronic lymphocytic leukemia. Blood 108, 1334–1338. https://doi.org/10.1182/blood-2005-12-011213 (2006).
Yan, X. J. et al. B cell receptors in TCL1 transgenic mice resemble those of aggressive, treatment-resistant human chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 103, 11713–11718. https://doi.org/10.1073/pnas.0604564103 (2006).
Carter, M. J. et al. PI3Kδ inhibition elicits anti-leukemic effects through Bim-dependent apoptosis. Leukemia 31, 1423–1433. https://doi.org/10.1038/leu.2016.333 (2017).
Acknowledgements
This study was supported by Karyopharm Therapeutics (M.C., UNITO-CLL01 study), University of Torino (local funds ex-60%) (M.C.) and Italian Association for Cancer Research (AIRC) (C.R. was recipient of grants AIRC IG15232 and IG21408, S.D. was recipient of grant AIRC IG23095). C.V. was recipient of a fellowship from Fondazione Pezcoller–Ferruccio ed Elena Bernardi” in collaboration with Società Italiana di Cancerologia (SIC). V.G. was recipient of a fellowship from AIRC (“Anna Nappa” fellowship, Ref 16343) and a fellowship from Fondazione “Angela Bossolasco” Torino, Italy (“Giorgio Bissolotti e Teresina Bosio” fellowship). M.T. was recipient of a fellowship from AIRC (Ref 19653).
Author information
Authors and Affiliations
Contributions
C.V., V.G. and M.C. designed the experiments, analyzed the results, interpreted data and wrote the manuscript. V.G., M.T., C.R., R.J., F.A. and T.V. performed the experiments and contributed to data interpretation. E.B. and F.P. supported experiment conduction. D.G.E and S.D. supported experiment conduction and contributed to data interpretation. Y.L. contributed to data interpretation. All authors reviewed the manuscript and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
C.V. has received consultancy fees from Abbvie, Janssen and Astra Zeneca. Y.L. was an employee of Karyopharm Therapeutics Inc. M.C. received honoraria from Janssen, Gilead, Abbvie, Shire, Astrazeneca and research support from Janssen, Abbvie and Karyopharm Therapeutics. The remaining authors declare no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vitale, C., Griggio, V., Todaro, M. et al. Anti-tumor activity of selinexor in combination with antineoplastic agents in chronic lymphocytic leukemia. Sci Rep 13, 16950 (2023). https://doi.org/10.1038/s41598-023-44039-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-44039-0
- Springer Nature Limited
This article is cited by
-
Nuclear to cytoplasmic transport is a druggable dependency in MYC-driven hepatocellular carcinoma
Nature Communications (2024)