Abstract
The A1762T/G1764A double mutant in the basal core promoter (BCP) region of the hepatitis B virus (HBV) is associated with severe hepatic lesions while the G1899A mutation with the double mutant is associated with a significant reduction in the risk of severe fibrosis. This study aims to measure a number of markers in the serum of patients with chronic HBV infection and to assess relationships between these markers and BCP/precore mutants with consideration of the stage of fibrosis. The serum levels of resistin, TGF-β1, MMP-1, TIMP-1, collagen IA1 and PDGF-BB, which are markers that are known to be involved in the process of hepatic fibrosis, were assayed. The serum levels of PDGF-BB and TIMP-1, and the mutation profile were independently associated with advanced fibrosis. A higher level of TIMP-1 was associated with advanced fibrosis regardless of the mutation status, and a higher level of PDGF-BB was associated with nonsevere fibrosis in patients infected with viruses harboring the A1762T/G1764A or A1762T/G1764A/G1899A mutations. Our results suggest an impact of the A1762T/G1764A mutant on the biological pathway related to TGF-β1 and PDGF-BB. In vitro studies are needed to understand the impact of these mutants on the serum secretion of markers involved in fibrosis severity.
Similar content being viewed by others
Introduction
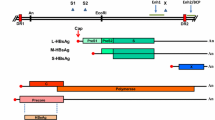
At present, chronic hepatitis B (CHB) remains a serious global health concern, with approximately 296 million people living with CHB infection worldwide1. Given that the virus is not directly cytopathic in itself, it is the conflict intensity between the hepatitis B virus (HBV) and the host immune response2,3 that determines the control of HBV infection, the development of liver damage (fibrosis, cirrhosis or hepatocellular carcinoma [HCC]) and the progressive selection of HBV variants. The typical course of HBV infection tends to involve a hepatitis B e antigen (HBeAg)-positive phase with high HBV DNA replication. This is followed by an HBeAg seroconversion phase with clinical remission and a decline in HBV viral load4. Nevertheless, during the HBeAg seroconversion phase, immune pressure can select a number of genetic variants in the basal core promoter (BCP) and precore (PC) regions that downregulate or abolish HBeAg production with active DNA replication5,6. Chronic HBeAg-negative hepatitis B has become the predominant type of CHB infection in France7 and worldwide. However, the impact of these mutations on the natural course of infection and on the severity of liver damage has not yet been clearly established for some of these mutants. The G1896A mutation in codon 28 of the PC region is the most frequently described mutation in this region and creates a premature stop codon that stops HBeAg synthesis8. Some studies reported a relationship between the G1896A mutation and severe liver hepatitis, whereas others reported no associations between the mutation and clinical outcomes9,10. In the BCP region, the A1762T/G1764A double mutant has been found to downregulate precore mRNA production, resulting in reduced HBeAg secretion11. An association between the A1762T/G1764A double mutant and severe hepatic lesions or increased risk for HCC has been frequently described12,13,14,15,51.
Detection of basal core promoter and precore mutations
The BCP (nucleotide position 1742–1849) and PC (nucleotide position 1814–1900) regions of the HBV genome were amplified by nested PCR as previously described17,52.
Nucleotide mutations were defined by their differences from the consensus sequence, and dual signals (mixed type) were considered a mutant type. The dominant viral strain was determined by Sanger sequencing and defined as > 50% of the virus.
Fibrosis marker analysis
The secreted fibrosis markers were measured in the serum samples of patients by multiplex fluorescent-bead-based technology (Luminex 200, Austin, TX, USA) using two commercial Luminex screening assay kits: a customized Luminex Assay kit from R&D Systems (Lilles, France) for MMP-1, resistin, Collagen IA1, PDGF-BB and TIMP-1 and the Bio-Plex Pro TGF-β assay from Bio–Rad Laboratories (Marnes-la-Coquette, France).
In brief, the samples were diluted before incubation with specific antibody-coated fluorescent beads according to the manufacturer’s recommendations. After washing, 50 beads were analyzed with the Luminex 200™ analyzer and Bio-Plex Manager software version 6 (Bio–Rad Laboratories), and the analyte concentrations of the samples were estimated through the serial dilution of cytokine standards (MMP-1 sensitivity < 3 pg/mL; resistin sensitivity < 20 ng/mL; Col IA1 sensitivity < 100 pg/mL; PDGF-BB sensitivity < 50 pg/mL; TIMP-1 sensitivity < 800 pg/mL; and TGF-β1 sensitivity < 15 pg/mL).
Statistical analysis
Quantitative variables are described as the means (standard error) or median (interquartile range) when appropriate. Categorical variables are described as total numbers (percentages).
Univariate analyses were performed to compare fibrosis markers in the serum samples according to fibrosis stage using Wilcoxon tests53 or according to the profile of mutation using Kruskal–Wallis tests54 and Steel–Dwass55 tests for post-hoc pairwise comparison. Proportions were compared using the chi-square test of independence. The data distributions were visualized in violin plots.
A first exploratory multivariate analysis was performed using FAMD, with the R package FactoMineR56 to analyze the variability of the data taking into account both the mutation profile and serum marker levels. Logistic regressions were then used to identify factors associated with advanced fibrosis. The candidate variables were MMP-1, resistin, Col IA1, PDGF-BB, TIMP-1, TGF- β1 and mutation profiles. The variables identified by univariate regressions (p value < 0.20) were then introduced in a multivariate analysis (stepwise backward logistic regression using Akaike Information Criterion for model selection). The regression formula used for this purpose also allowed us to identify factors independently associated with advanced fibrosis. Finally, we assessed the Spearman correlations between the serum levels of liver markers according to the mutation profile using the R package corrplot.
A p value lower than 0.05 was considered statistically significant. Statistical analyses were performed using R version 3.6.2.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
World Health Organization. Hepatitis B. https://www.who.int/news-room/fact-sheets/detail/hepatitis-b (2021).
Wu, J.-F. & Chang, M.-H. Natural history of chronic hepatitis B virus infection from infancy to adult life: The mechanism of inflammation triggering and long-term impacts. J. Biomed. Sci. 22, 92 (2015).
Chang, M.-L. & Liaw, Y.-F. Hepatitis B flares in chronic hepatitis B: Pathogenesis, natural course, and management. J. Hepatol. 61, 1407–1417 (2014).
Liaw, Y.-F. & Chu, C.-M. Hepatitis B virus infection. Lancet 373, 582–592 (2009).
Funk, M. L., Rosenberg, D. M. & Lok, A. S. F. World-wide epidemiology of HBeAg-negative chronic hepatitis B and associated precore and core promoter variants. J. Viral Hepat. 9, 52–61 (2002).
Kitab, B. et al. Variability in the precore and core promoter regions of HBV strains in Morocco: Characterization and impact on liver disease progression. PLoS ONE 7, e42891 (2012).
Zarski, J.-P. et al. Characteristics of patients with chronic hepatitis B in France: Predominant frequency of HBe antigen negative cases. J. Hepatol. 45, 355–360 (2006).
Carman, W. F. et al. Mutation preventing formation of hepatitis B e antigen in patients with chronic hepatitis B infection. Lancet 2, 588–591 (1989).
Ozasa, A. et al. Influence of genotypes and precore mutations on fulminant or chronic outcome of acute hepatitis B virus infection. Hepatolology. 44, 326–334 (2006).
Tong, M. J., Blatt, L. M., Kao, J.-H., Cheng, J. T. & Corey, W. G. Basal core promoter T1762/A1764 and precore A1896 gene mutations in hepatitis B surface antigen-positive hepatocellular carcinoma: A comparison with chronic carriers. Liver Int. 27, 1356–1363 (2007).
Buckwold, V. E., Xu, Z., Chen, M., Yen, T. S. & Ou, J. H. Effects of a naturally occurring mutation in the hepatitis B virus basal core promoter on precore gene expression and viral replication. J. Virol. 70, 5845–5851 (1996).
Liu, S. et al. Associations between hepatitis B virus mutations and the risk of hepatocellular carcinoma: A meta-analysis. J. Natl. Cancer Inst. 101, 1066–1082 (2009).
Chen, C.-H. et al. Clinical significance of hepatitis B virus (HBV) genotypes and precore and core promoter mutations affecting HBV e antigen expression in Taiwan. J. Clin. Microbiol. 43, 6000–6006 (2005).
Bai, X. et al. Temporal acquisition of sequential mutations in the enhancer II and basal core promoter of HBV in individuals at high risk for hepatocellular carcinoma. Carcinogenesis 32, 63–68 (2011).
Liao, Y. et al. Precore mutation of hepatitis B virus may contribute to hepatocellular carcinoma risk: Evidence from an updated meta-analysis. PLoS ONE 7, e38394 (2012).
Yang, Y., Sun, J.-W., Zhao, L.-G., Bray, F. & **ang, Y.-B. Quantitative evaluation of hepatitis B virus mutations and hepatocellular carcinoma risk: A meta-analysis of prospective studies. Chin. J. Cancer Res. 27, 497–508 (2015).
Ducancelle, A. et al. Different precore/core mutations of hepatitis B interact with, limit, or favor liver fibrosis severity. J. Gastroenterol. Hepatol. 31, 1750–1756 (2016).
Mansouri, A., Gattolliat, C.-H. & Asselah, T. Mitochondrial dysfunction and signaling in chronic liver diseases. Gastroenterology 155, 629–647 (2018).
Friedman, S. L. Mechanisms of hepatic fibrogenesis. Gastroenterology 134, 1655–1669 (2008).
Ming, D. et al. Elevated TGF-β1/IL-31 pathway is associated with the disease severity of hepatitis B virus-related liver cirrhosis. Viral Immunol. 28, 209–216 (2015).
Meng, Z. et al. High serum resistin associates with intrahepatic inflammation and necrosis: An index of disease severity for patients with chronic HBV infection. BMC Gastroenterol. 17, 6 (2017).
Murawaki, Y., Ikuta, Y., Idobe, Y. & Kawasaki, H. Serum matrix metalloproteinase-1 in patients with chronic viral hepatitis. J. Gastroenterol. Hepatol. 14, 138–145 (1999).
Bedossa, P. Liver fibrosis in hepatitis B: A dynamic process. Gastroenterol. Clin. Biol. 34(Suppl 2), S103-108 (2010).
Liu, T., Wang, X., Karsdal, M. A., Leeming, D. J. & Genovese, F. Molecular serum markers of liver fibrosis. Biomark. Insights 7, 105–117 (2012).
Horowitz, J. M. et al. Evaluation of hepatic fibrosis: A review from the society of abdominal radiology disease focus panel. Abdom. Radiol. N. Y. 42, 2037–2053 (2017).
Soresi, M., Giannitrapani, L., Cervello, M., Licata, A. & Montalto, G. Non invasive tools for the diagnosis of liver cirrhosis. World J. Gastroenterol. 20, 18131–18150 (2014).
Diang, X.-C. et al. Association between serum platelet-derived growth factor BB and degree of liver damage, fibrosis and hepatitis B e antigen (HBeAg) status in CHB patients. Hepatogastroenterology. 59, 2357–2360 (2012).
Zhang, B.-B. et al. Diagnostic value of platelet derived growth factor-BB, transforming growth factor-beta1, matrix metalloproteinase-1, and tissue inhibitor of matrix metalloproteinase-1 in serum and peripheral blood mononuclear cells for hepatic fibrosis. World J. Gastroenterol. 9, 2490–2496 (2003).
Zhu, C. L., Li, W. T., Li, Y. & Gao, R. T. Serum levels of tissue inhibitor of metalloproteinase-1 are correlated with liver fibrosis in patients with chronic hepatitis B. J. Dig. Dis. 13, 558–563 (2012).
Leroy, V. et al. Circulating matrix metalloproteinases 1, 2, 9 and their inhibitors TIMP-1 and TIMP-2 as serum markers of liver fibrosis in patients with chronic hepatitis C: Comparison with PIIINP and hyaluronic acid. Am. J. Gastroenterol. 99, 271–279 (2004).
Bertolani, C. et al. Resistin as an intrahepatic cytokine: overexpression during chronic injury and induction of proinflammatory actions in hepatic stellate cells. Am. J. Pathol. 169, 2042–2053 (2006).
Border, W. A. & Ruoslahti, E. Transforming growth factor-beta in disease: The dark side of tissue repair. J. Clin. Invest. 90, 1–7 (1992).
Bai, Q. et al. HBV promotes the proliferation of hepatic stellate cells via the PDGF-B/PDGFR-β signaling pathway in vitro. Int. J. Mol. Med. 30, 1443–1450 (2012).
Ross, R. Platelet-derived growth factor. Lancet 1, 1179–1182 (1989).
Akpolat, N., Yahsi, S., Godekmerdan, A., Demirbag, K. & Yalniz, M. Relationship between serum cytokine levels and histopathological changes of liver in patients with hepatitis B. World J. Gastroenterol. 11, 3260–3263 (2005).
Tsushima, H. et al. Reduced plasma transforming growth factor-beta1 levels in patients with chronic hepatitis C after interferon-alpha therapy: Association with regression of hepatic fibrosis. J. Hepatol. 30, 1–7 (1999).
Kanzler, S. et al. Prediction of progressive liver fibrosis in hepatitis C infection by serum and tissue levels of transforming growth factor-beta. J. Viral Hepat. 8, 430–437 (2001).
Flisiak, R., Pytel-Krolczuk, B. & Prokopowicz, D. Circulating transforming growth factor beta(1) as an indicator of hepatic function impairment in liver cirrhosis. Cytokine 12, 677–681 (2000).
Haghgoo, S. M., Sharafi, H. & Alavian, S. M. Serum cytokines, adipokines and ferritin for non-invasive assessment of liver fibrosis in chronic liver disease: A systematic review. Clin. Chem. Lab. Med. 57, 577–610 (2019).
Zhou, J. et al. Serum platelet-derived growth factor BB levels: A potential biomarker for the assessment of liver fibrosis in patients with chronic hepatitis B. Int. J. Infect. Dis. 49, 94–99 (2016).
El-Bassiouni, N. E. et al. Role of fibrogenic markers in chronic hepatitis C and associated hepatocellular carcinoma. Mol. Biol. Rep. 39, 6843–6850 (2012).
Kantor, E. D., Lampe, J. W., Kratz, M. & White, E. Lifestyle factors and inflammation: Associations by body mass index. PLoS ONE 8, e67833 (2013).
Siervo, M. et al. Body mass index is directly associated with biomarkers of angiogenesis and inflammation in children and adolescents. Nutrition 28, 262–266 (2012).
Yoshida, S. et al. Extrahepatic platelet-derived growth factor-β, delivered by platelets, promotes activation of hepatic stellate cells and biliary fibrosis in mice. Gastroenterology 147, 1378–1392 (2014).
Kajihara, M. et al. Evaluation of platelet kinetics in patients with liver cirrhosis: Similarity to idiopathic thrombocytopenic purpura. J. Gastroenterol. Hepatol. 22, 112–118 (2007).
Bayliss, J. et al. Deep sequencing shows that HBV basal core promoter and precore variants reduce the likelihood of HBsAg loss following tenofovir disoproxil fumarate therapy in HBeAg-positive chronic hepatitis B. Gut 66, 2013–2023 (2017).
Neuman, M. G. et al. Kinetics of serum cytokines reflect changes in the severity of chronic hepatitis C presenting minimal fibrosis. J. Viral Hepat. 9, 134–140 (2002).
Revill, P. A. et al. The evolution and clinical impact of hepatitis B virus genome diversity. Nat. Rev. Gastroenterol. Hepatol. 17, 618–634 (2020).
Bedossa, P. & Poynard, T. An algorithm for the grading of activity in chronic hepatitis C The METAVIR Cooperative Study Group. Hepatology 24, 289–293 (1996).
Rousselet, M.-C. et al. Sources of variability in histological scoring of chronic viral hepatitis. Hepatology 41, 257–264 (2005).
Villeneuve, J.-P. et al. Selection of a hepatitis B virus strain resistant to adefovir in a liver transplantation patient. J. Hepatol. 39, 1085–1089 (2003).
Friedt, M. et al. Mutations in the basic core promotor and the precore region of hepatitis B virus and their selection in children with fulminant and chronic hepatitis B. Hepatol. Baltim. Md 29, 1252–1258 (1999).
Wilcoxon, F. Individual comparisons by ranking methods. Biom. Bull. 1, 80–83 (1945).
Kruskal, W. H. & Wallis, W. A. Use of ranks in one-criterion variance analysis. J. Am. Stat. Assoc. 47, 583–621 (1952).
Steel, R. G. D. A rank sum test for comparing all pairs of treatments. Technometrics 2, 197–207 (1960).
Lê, S., Josse, J. & Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 25, 1 (2008).
Acknowledgements
The authors thank the 14 participating virology laboratories (Pontchaillou Hospital, Rennes; Amiens University Hospital, Amiens; Bichat Claude Bernard Hospital, Paris; Beaujon Hospital, Paris; Saint Louis Hospital, Paris; Saint Antoine University Hospital, Paris; Hôtel Dieu Hospital, Paris; Nancy Hospital, Nancy; Grenoble University Hospital, Grenoble; Dupuytren Hospital, Limoges; University Hospital of Paris Seine-Saint-Denis, Avicenne and Bobigny; Robert Debré Hospital, Reims, Henri Mondor Hospital, Créteil). The authors thank Michael Wood for proofreading the English manuscript.
Author information
Authors and Affiliations
Contributions
C.L., P.J., and A.D., conceived and designed the project. A.D. was involved in acquisition of data. C.L., S.B., and A.P. conducted most of the experiments and analyzed data. M.R. performed statistical analysis. C.L. and M.R. created the figures and tables. A.D. was involved in study supervision. C.L. wrote the draft of manuscript. M.R., S.B., H.L.G.G., J.B., F.L.F., and A.D. helped critically reviewing the manuscript. All authors participated in discussing and revising this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lefeuvre, C., Roux, M., Blanchard, S. et al. Analysis of hepatic fibrosis markers in the serum of chronic hepatitis B patients according to basal core promoter/precore mutants. Sci Rep 12, 10261 (2022). https://doi.org/10.1038/s41598-022-14285-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-14285-9
- Springer Nature Limited