Abstract
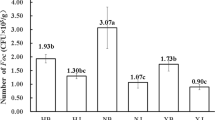
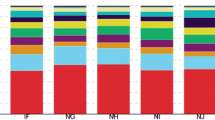
Dwarf bunt of wheat, which is caused by Tilletia controversa J.G. Kühn, is a soil-borne disease which may lead up to an 80% loss of yield together with degradation of the quality of the wheat flour by production of a fishy smell. In this study, high-throughput sequencing technology was employed to characterize the microbial composition of wheat tissues (roots, spikes, first stem under the ear, and stem base) and rhizosphere soil of wheat varieties that are resistant and susceptible to T. controversa. We observed that the soil fungal community abundance and diversity were higher in resistant varieties than in susceptible varieties in both inoculated and uninoculated wheat, and the abundances of Sordariomycetes and Mortierellomycetes increased in the resistant varieties infected with T. controversa, while the abundances of Dothideomycetes and Bacteroidia increased in the susceptible varieties. Regarding the bacteria present in wheat tissues, the abundances of Chloroflexi, Bacteroidetes, Gemmatimonadetes, Verrucomicrobia and Acidobacteria in the ear and the first stem under the ear were higher than those in other tissues. Our results indicated that the abundances of Sordariomycetes, Mortierellomycetes, Leotiomycetes, Chryseobacterium and Massilia were higher in T. controversa-infected resistant varieties than in their controls, that Dothideomycetes, Bacteroidia, Nocardioides and Pseudomonas showed higher abundances in T. controversa-infected susceptible varieties, and that Curtobacterium, Exiguobacterium, Planococcus, and Pantoea may have higher abundances in both T. controversa-infected susceptible and resistant varieties than in their own controls.
Similar content being viewed by others
Introduction
Wheat (Triticum aestivum L.) is an important food crop that humans have been consuming for 5000 years1. The quality and yield of wheat are affected by various factors2. Dwarf bunt in wheat, which is caused by Tilletia controversa J.G. Kühn, spreads through seeds or soil3 and is a disease of quarantine significance in many countries4. This disease often harms winter wheat in areas covered by snow for long periods in winter. Wheat Dwarf bunt can lead to a reduction in wheat production, degrade flour quality and produce a rotten fish odour. Resistant varieties of wheat contribute considerably to controlling this disease5. The teliospores of T. controversa have strong resistance to stress and can survive for 10 years under favourable environments6.
Plant microbiota can help to maintain the health of plants and can provide important genetic variability, which is of strong significance for plant resistance to biotic and abiotic stress7. Plant entophytic bacteria are parasitic bacteria that are widely observed in the tissues of plants in nature8. These bacteria can exist in various parts of the plant, including the aboveground, belowground, and seed parts9, and they can promote plant growth by fixing nitrogen, producing plant hormones, and improving drought resistance10,11,12; also, these bacteria can protect host plants from damage by inhibiting the growth of pathogenic bacteria13,14,15. In a recent study on the response of wheat endophytes to stripe rust, it was observed that the abundance of endophytes in roots was higher than that in stems and leaves, and the abundances of endophytes in resistant and susceptible varieties was observed to vary considerably16.
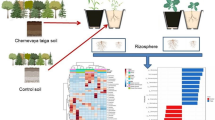
Rhizosphere soil serves as a bridge between microbes and plant roots. This soil enables materials and energy to be exchanged between plants and microbes. The rhizosphere microbiota promotes plant growth and health by enhancing plant resistance to adverse conditions or improving plant nutrient absorption17,18,19. The rhizosphere bacterial community in the soil can strongly reduce the morbidity and mortality in tobacco caused by mixed Fusarium-Alternaria disease20. Thilagam and Hemalatha21 found that plant growth-promoting rhizobacterial (PGPR) actinobacterial isolates can effectively suppress chili anthracnose. At the same time, soil microbes can also increase the resistance of plants to some microbial stresses by enhancing plant drought resistance by intercepting hormones in plants22. The rhizosphere microbiota can also affect the nutritional status of plants. The symbiotic relationship between legumes and nitrogen-fixing rhizobia is a typical example of how soil microbiota helps plants absorb nitrogen23. Some rhizosphere microbiota can promote iron and phosphorus absorption by plants through mineralization, dissolution, or the secretion of iron carriers19, 24. Similarly, plants can also affect the structure and composition of the rhizosphere microbial community. Resistant and susceptible varieties exhibit differences between their rhizosphere microbial communities. In a study of the resistance and susceptibility of watermelon to Fusarium oxysporum f. sp. niveum, An et al.25 observed that the populations of actinomycetes in the rhizosphere soil of resistant varieties are more abundant than those of susceptible varieties, but the fungal community exhibits the opposite property. Sun et al.http://drive5.com/uparse/), OTUs were clustered based on 97% similarity, and single sequences and chimaeras were removed during the clustering process. The RDP classifier (http://rdp.cme.msu.edu/) was utilized to classify and annotate each sequence. The Silva database (SSU123) was utilized to compare 16S rRNA sequences, and Unite (Release 6.0 http://unite.ut.ee/index.php) was utilized to compare the internal transcribed spacer region; the confidence threshold was 0.7. All raw paired-end Illumina sequence data have been deposited in the National Center for Biotechnology Information (NCBI) Sequence Read Archive database (https://www.ncbi.nlm.nih.gov/sra) under BioProject no. PRJNA639912.
References
Islam, S. et al. Characterization of the single-cell transcriptional landscape by highly multiplex RNA-seq. Genome. Res. 21, 1160–1167 (2011).
Shapiro, E., Biezuner, T. & Linnarsson, S. Single-cell sequencing-based technologies will revolutionize whole-organism science. Nat. Rev. Genet. 14, 618–630 (2013).
Tyler, L. J. & Jensen, N. F. Some factors that influence development of dwarf bunt in whiter wheat. Phytopathology 48, 565–571 (1958).
Goates, B. J., Peterson, G. L., Bowden, R. L. & Maddux, L. D. Analysis of induction and establishment of dwarf bunt of wheat under marginal climatic conditions. Plant Dis. 95, 478–484 (2011).
Mathre, D. E. Dwarf bunt: politics, identification, and biology. Annu. Rev. Phytopathol. 34, 67–85 (1996).
Helback, H. Domestication of Food Plants in the Old World: Joint efforts by botanists and archeologists illuminate the obscure history of plant domestication. Science 130, 365–372 (1959).
Dai, Y. et al. Wheat-associated microbiota and their correlation with stripe rust reaction. J. Appl. Microbiol. 128, 544–555 (2020).
Rosenblueth, M. & Martínez-Romero, E. Bacterial endophytes and their interactions with hosts. Mol. Plant Microbe. Interact. 19, 827–837 (2006).
Chebotar, V. K. et al. Endophytic bacteria in microbial preparations that improve plant development. Prikl. Biokhim. Mikrobiol. 51, 283–289 (2015).
Flores-Félix, J. D. et al. Plants probiotics as a tool to produce highly functional fruits: The case of phyllobacterium and vitamin C in strawberries. PLoS ONE 10, e0122281 (2015).
Qin, Y. et al. Isolation and identification of a cold-adapted bacterium and its characterization for biocontrol and plant growth-promoting activity. Ecol. Eng. 105, 362–369 (2017).
Zhang, L. et al. Deciphering the root endosphere microbiome of the desert plant Alhagi sparsifolia for drought resistance-promoting bacteria. Appl. Environ. Microbiol. 86, e02863-e2919 (2020).
El-Shatoury, S. A. et al. Biocontrol of chocolate spot disease (Botrytis cinerea) in faba bean using endophytic actinomycetes Streptomyces: a field study to compare application techniques. PeerJ. 8, e8582 (2020).
Islam, M. N., Ali, M. S., Choi, S. J., Hyun, J. W. & Baek, K. H. Biocontrol of citrus canker disease caused by Xanthomonas citri subsp. citri using an endophytic Bacillus thuringiensis. Plant Pathol. J. 35, 486–497 (2019).
Romero, F. M., Marina, M. & Pieckenstain, F. L. Novel components of leaf bacterial communities of field-grown tomato plants and their potential for plant growth promotion and biocontrol of tomato diseases. Res. Microbiol. 167(3), 222–233 (2016).
Kiani, T. et al. Isolation and characterization of culturable endophytic bacterial community of stripe rust-resistant and stripe rust-susceptible Pakistani wheat cultivars. Int. Microbiol. 22, 191–201 (2019).
Bakker, P., Pieterse, C., de Jonge, R. & Berendsen, R. L. The soil-borne legacy. Cell 172, 1178–1180 (2018).
Berendsen, R. L., Pieterse, C. M. & Bakker, P. A. The rhizosphere microbiome and plant health. Trends Plant Sci. 17, 478–486 (2012).
Bulgarelli, D., Schlaeppi, K., Spaepen, S., Ver Loren vanThemaat, E. & Schulze-Lefert, P. Structure and functions of the bacterial microbiota of plants. Annu. Rev. Plant Biol. 64, 807–838 (2013).
Santhanam, R. et al. Native root-associated bacteria rescue a plant from a sudden-wilt disease that emerged during continuous crop**. Proc. Natl. Acad. Sci. U.S.A. 112, E5013–E5020 (2015).
Thilagam, R. & Hemalatha, N. Plant growth promotion and chilli anthracnose disease suppression ability of rhizosphere soil actinobacteria. J. Appl. Microbiol. 126, 1835–1849 (2019).
Fitzpatrick, C. R. et al. Assembly and ecological function of the root microbiome across angiosperm plant species. Proc. Natl. Acad. Sci. U.S.A. 115(6), E1157–E1165 (2018).
Udvardi, M. & Poole, P. S. Transport and metabolism in legume-rhizobia symbioses. Annu. Rev. Plant Biol. 64, 781–805 (2013).
Mendes, R., Garbeva, P. & Raaijmakers, J. M. The rhizosphere microbiome: Significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol. Rev. 37, 634–663 (2013).
An, M., Zhou, X., Wu, F., Ma, Y. & Yan, P. Rhizosphere soil microorganism populations and community structures of different watermelon cultivars with differing resistance to Fusarium oxysporum f. sp. niveum. Can. J. Microbiol. 57, 355–365. https://doi.org/10.1139/w11-015 (2011).
Sun, J., Peng, M., Wang, Y., Li, W. & **a, Q. The effects of different disease-resistant cultivars of banana on rhizosphere microbial communities and enzyme activities. FEMS Microbiol. Lett. 345, 121–126 (2013).
Kuntal, B. K., Chandrakar, P., Sadhu, S. & Mande, S. S. “NetShift”: A methodology for understanding “driver microbes” from healthy and disease microbiome datasets. ISME J. 13, 442–454 (2019).
Zhou, X., Wang, J. T., Wang, W. H., Tsui, C. K. & Cai, L. Changes in bacterial and fungal microbiomes associated with tomatoes of healthy and infected by Fusarium oxysporum f. s. lycopersici. Microb. Ecol. https://doi.org/10.1007/s00248-020-01535-4 (2020).
Wei, F., Passey, T. & Xu, X. Amplicon-based metabarcoding reveals temporal response of soil microbial community to fumigation-derived products. Appl. Soil Ecol. 103, 83–92 (2016).
Wei, Z. et al. Trophic network architecture of root-associated bacterial communities determines pathogen invasion and plant health. Nat. Commun. 6, 8413 (2015).
Walitang, D. I. et al. Characterizing endophytic competence and plant growth promotion of bacterial endophytes inhabiting the seed endosphere of Rice. BMC Microbiol. 17, 209 (2017).
Rascovan, N. et al. Integrated analysis of root microbiomes of soybean and wheat from agricultural fields. Sci. Rep. 6, 28084 (2016).
Campisano, A. et al. Bacterial endophytic communities in the grapevine depend on pest management. PLoS ONE 9, e112763 (2014).
Kandel, S. L., Joubert, P. M. & Doty, S. L. Bacterial endophyte colonization and distribution within plants. Microorganisms 5, 77 (2017).
Robinson, R. J. et al. Endophytic bacterial community composition in wheat (Triticum aestivum) is determined by plant tissue type, developmental stage and soil nutrient availability. Plant Soil 405, 381–396 (2016).
Wemheuer, F. et al. Bacterial endophyte communities of three agricultural important grass species differ in their response towards management regimes. Sci. Rep. 7, 40914 (2017).
Xu, W. et al. Diversity of cultivable endophytic bacteria in mulberry and their potential for antimicrobial and plant growth-promoting activities. Microbiol. Res. 229, 126328 (2019).
Wei, F. et al. Cultivar-dependent variation of the cotton rhizosphere and endosphere microbiome under field conditions. Front. Plant Sci. 10, 1659 (2019).
Compant, S., Clément, C. & Sessitsch, A. Plant growth-promoting bacteria in the rhizo- and endosphere of plants: Their role, colonization, mechanisms involved and prospects for utilization. Soil Biol. Biochem. 42, 669–678 (2010).
Berg, G. et al. Endophytic and ectophytic potato-associated bacterial communities differ in structure and antagonistic function against plant pathogenic fungi. FEMS Microbiol. Ecol. 51, 215–229 (2005).
Zadoks, J. C., Chang, T. T. & Konzak, C. F. A decimal code for the growth stages of cereals. Weed Res. 14, 415–421 (1974).
Gao, L. et al. Development of a SCAR marker for molecular detection and diagnosis of Tilletia controversa Kühn, the causal fungus of wheat dwarf bunt. World J. Microbiol. Biotechnol. 30, 3185–3195 (2014).
Oksanen, J. et al. vegan: Community Ecology Package. R package version 2.5–5. https://CRAN.R-project.org/package=vegan (2019).
Csardi, G. & Nepusz, T. The igraph software package for complex network research. Inter. Comp. Syst. 1695, 1–9 (2006).
Clauset, A., Newman, M. E. J. & Moore, C. Finding community structure in very large networks. Phys. Rev. E. 70, 066111 (2004).
Acknowledgements
This work was supported by the Natural Science Foundation (31761143011, 31571965), the National Key Research and Development Programme of China (2018YFD0200406) and the Ministry of Agriculture and Rural Affairs of China (CARS-03).
Author information
Authors and Affiliations
Contributions
L.G. designed the research and revised the manuscript. T.S.X., W.L.J, D.D.Q. performed the experiments. T.S.X. contributed to data analyses and manuscript writing. L.G., J.M.Z., W.Q.C., and T.G.L. contributed reagents/materials/analysis tools. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xu, T., Jiang, W., Qin, D. et al. Characterization of the microbial communities in wheat tissues and rhizosphere soil caused by dwarf bunt of wheat. Sci Rep 11, 5773 (2021). https://doi.org/10.1038/s41598-021-85281-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-85281-8
- Springer Nature Limited
This article is cited by
-
Sugar accumulation stage in sugar beets is a key stage in response to continuous crop** soil microbial community assembly
Plant and Soil (2024)
-
Deciphering differences in microbial community composition and multifunctionality between healthy and Alternaria solani-infected potato rhizosphere soils
Plant and Soil (2023)