Abstract
Expanded newborn screening facilitates early identification and intervention of patients with inborn errors of metabolism (IEMs), There is a lack of disease spectrum data for many areas in China. To determine the disease spectrum and genetic characteristics of IEMs in **'an city of Shaanxi province in northwest China, 146152 newborns were screening by MSMS from January 2014 to December 2019 and 61 patients were referred to genetic analysis by next generation sequencing (NGS) and validated by Sanger sequencing. Seventy-five newborns and two mothers were diagnosed with IEMs, with an overall incidence of 1:1898 (1:1949 without mothers). There were 35 newborns with amino acidemias (45.45%, 1:4176), 28 newborns with organic acidurias (36.36%, 1:5220), and 12 newborns and two mothers with FAO disorders (18.18%; 1:10439 or 1:12179 without mothers). Phenylketonuria and methylmalonic acidemia were the two most common disorders, accounting for 65.33% (49/75) of all confirmed newborn. Some hotspot mutations were observed for several IEMs, including PAH gene c.728G>A for phenylketonuria; MMACHC gene c.609G>A and c.567dupT, MMUT gene c.323G>A for methylmalonic acidemia and SLC25A13 gene c.852_855del for citrin deficiency. Our study provides effective clinical guidance for the popularization and application of expanded newborn screening, genetic screening, and genetic counseling of IEMs in this region.
Similar content being viewed by others
Introduction
Inborn errors of metabolism (IEMs) are a phenotypically and genetically heterogeneous group of disorders caused by defective enzymes, cofactors, and transporters in metabolic pathways, leading to the accumulation of toxic substrates, the production of by-products, and reduced levels of products, which result in a series of clinical manifestations1. Since Garrod first proposed the concept of “inborn errors of metabolism” in 1909, more than 1000 different IEMs have been identified. While individual rare, the overall incidence has been shown to be upwards of 1 in 8002, IEMs can occur in any ethnic group and across every age. Clinical manifestations are complex and often non-specific, including acute metabolic crisis, epilepsy, metabolic acidosis, severe low blood sugar, high blood ammonia, and multiple organ damage. Some of them are amenable to treatment, if not treated timely, may cause irreversible physical disabilities, mental retardation, and even early death3.
Newborn screening (NBS) for IEMs can improve their prognosis and quality of life through early diagnosis and early intervention, recognizing as a huge success in field of public health of the last 50 years4. In the early 2000s, tandem mass spectrometry (MSMS) was introduced into many newborn screening programs, this technology allows inexpensive simultaneous detect dozens of amino acids and acylcarnitine profile in a single test, with commendable analytical accuracy and precision. Consequently, MS/MS has been applied wildly for the screening of IEMs that include most amino acid disorders, organic acidemias, and fatty acid oxidation defects 1,5.
In mainland China, MS/MS-based NBS was first piloted in Shanghai in 2003. There were 116000 newborns screened from 2003 to 2007, twenty IEMs were identified yielding an incidence of 1:58006. However, subsequent studies have shown that the incidence, disease spectrum, and genetic characteristics of IEMs varied greatly in different regions of China, especially between the north and south. Furthermore, although MS/MS has been applied in China for many years, there are neither corresponding technical specifications in China nor expanded NBS data for many areas. Therefore, it is necessary to accumulate data on the clinical practice of MSMS and to study the IEMs disease spectrum and gene mutations in different regions to guide expanded NBS.
Expanded NBS program was begun in 2014 in ** normally at present. The longest follow-up period is approximately 6 years.
Discussion
There are no national detailed expanded NBS standards in China due to regional differences in the application of MSMS and lack of sufficient and valid studies. Some work has been done on the epidemiology, technical aspects and clinical validity of MS/MS screening in several provinces and cities in China, indicated that NGS combined with MS/MS is an enhanced plan for IEMs as well as disease spectrum and genetic characteristic of IEMs vary dramatically in different regions9,10,11,12,12. Genetic analysis showed that the most common mutation was c.728G>A in the PAH gene, which is in consistent with previous reports22,23.
Methylmalonic acidemia was the second most common IEM in this region and also had the highest incidence of organic acidemias (1:6960), which was slightly lower than reported in **ing city (1:5590)10 and Henan province (1:6032)24, but significantly higher than in Zhejiang province (1:46500)25, Jiangsu province (1:35734)9, Quanzhou city (1:121515)11, and Taiwan (1:101625)20 in southern China. Thus, our research provides further evidence that the incidence of MMA in northern China is higher than that in southern China. However, 21 patients were diagnosed with methylmalonic acidemia in this study, 52.4% of patients with isolated MMA and 47.6% with combined MMA with homocysteinemia, which were different from most reported conclusions that combined MMA with homocysteinemia caused by the MMACHC gene mutation is the common type of MMA in the mainland of China 26,27. It is worth noting that our study was performed at a single center, may result in selection bias. Therefore, it is necessary to continuously increase the samples from different cities in this region in order to obtain more objective and reliable information about the disease spectrum of MMA. c.609G>A and c.567dupT in the MMACHC gene were two common mutations, which is consistent with previous reports26. Similar to the report by Hu et al.28, in our population, c.323G>A was the most prevalent mutation in the MMUT gene. However, previous other studies have suggested that c.729_730insTT is the most common MMUT mutation in China29,30. This difference should be taken into account the small number of cases in this study and the impact of geographical differences.
Citrin deficiency was another common amino acid disorder detected in this study with an incidence of 1:36538, comparable with those reported in **ing city (1:39556)10 and Quanzhou city (1:36455)11. The incidence in our study was significantly higher than the calculated incidence based on the carrier rate of 1:940 in the SLC25A13 gene in northern China31. However, a recent study conducted by Lin et al.32 on genetic screening for 29364 newborns at six NBS centers in southern, northern, and border regions of China, showed that the carrying rate of SLC25A13 gene mutations were basically the same in these three regions (1/42, 1/43, and 1/56, respectively). Therefore, more data should be available to determine the true incidence and regional differences of CD. Nevertheless, a large number of studies have shown that c.852_855del in the SLC25A13 gene is a hotspot mutation in Chinese patients with CD, with no regional differences in its incidence. The four patients diagnosed with CD in this study carried c.852_855del heterozygous mutations in the SLC25A13 gene. But unfortunately, CD as an autosomal recessive disorder, we did not detect other mutations in these four patients, which may be related to the limited detection ability of target NGS technology to detect gene intron regions, regulatory regions, large fragment deletions / duplications and complicated structural rearrangements. In recent years, with the reduction in cost and widely application of NGS, newborn genetic screening has received more and more attention33. In terms of the above cases, biochemical screening is still irreplaceable. Of course, considering the previous reported cases of false negative results with MSMS screening, combining newborn metabolic and genetic screening may be a better choice.
In our study, fatty acid oxidation disorders accounted for only 18.18% of all confirmed cases, including PCD, SCADD, MCADD, and VLCADD, with 3 infants of each identified. However, if maternal carnitine deficiency was included in the analysis, it could be concluded that carnitine deficiency was more common in the region. c.1400C>G in the SLC22A5 gene for PCD and c.1031A>G in the ACADS gene for SCADD were common mutations, consistent with previous reports10,11,12,34. However, of the 3 cases diagnosed with MCADD, 2 cases with dizygotic twins carried a homozygous c.985A>G (66.7%) mutation in the ACADM gene, so we think that the mutation frequency of c.985A>G seems to be overestimated in this study. c.985A>G is a known high-frequency mutation of MCADD in Caucasian populations, involved in approximately 90% of cases35, but it has not been found in Japan36, South Korea37 and Taiwan20.
In this study, there was one patient confirmed with HCS by expanded NBS. This disease is a type of multiple carboxylase deficiency (MCD) caused by abnormal biotin metabolism. The clinical manifestations are non-specific, and usually mainly include nervous system and skin damage. The results of MSMS showed elevated 3-hydroxypentanoylcarnitine. In mainland China, except for 3 cases of MCD confirmed in 1861262 newborns screened in Zhejiang province25, there were no patient reported in other regions by expanded NBS9,10,11. In addition, Han et al.30 screened 18303 patients with suspected IEMs by MSMS, 24 patients were diagnosed with HCS. Likewise, a comparative study of selective screening and expanded NBS in Asian countries suggested that larger number of patients with MCD were identified via selective screening, whereas very few were discovered with expanded NBS and it is easier to detect when dietary biotin insufficiency38. Therefore, the diagnosis of MCD depends more on the identification of clinicians, and of course, early diagnosis through expanded NBS can lead to effective intervention before the onset of clinical symptoms, thus reducing the risk of a poor prognosis.
This study had several limitations of note. With regard to MSMS screening efficiency, like most newborn screening centers in China, we did not use any second tier testing so that we had a high positive rate of initial screening. The Region4Stork (R4S) collaborative project has provided an optimized tool for laboratory management, clinical validation and interpretation of results of newborn screening by MS/MS, which effectively reduced the false positive rate and promoted the improvement of screening efficiency39,40. However, this system has not been widely used in China, and we still use traditional interpretation rules, which may also result in a high positive rate of initial screening. The artificial intelligence interpretation model based on localized big data under study is expected to further promote the interpretation of newborn screening results by MSMS. In addition, due to the lack of relevant screening policies and the inadequacy of education on expanded NBS, nearly 10% of the infants in this study were not recalled for secondary testing. Although we did not find false negative cases during the study period, which was related to the strict cut-off value we set, it also caused a high initial screening positive rate, which in turn caused a psychological and economic burden on neonatal guardians. The identification of false negative results mainly relies on child care management system. In this way, the current imperfect child care network may also lead to failure to effectively identify false negative cases. Therefore, how to further optimize the MSMS screening system and relevant policy support to improve screening efficiency will become the focus of our following work. Besides, a potential source of bias in sample acquisition may exit due to its single-center design, thus, our findings may not accurately reflect the prevalence of IEMs in Shaanxi province and even northwest China. However, considering the lack of massive information on expanded NBS in this region, so the incidence and disease spectrum of IEMs, including amino acid disorders, organic acidemia and fatty acid oxidation disorders derived from this study still have important reference value for the implementation of expanded NBS by MSMS in Northwest China.
Conclusions
We presented expanded NBS results with MS/MS in a northwestern Chinese population. The prevalence, disease spectrum, and genetic characteristics of confirmed IEMs were initially clarified. Our experience showed that IEMs were not uncommon in northwest China, PKU and MMA were the most common IEMs in this region, which provided effective clinical guidance for the popularization and application of MS/MS with genetic analysis for NBS and diagnosis of IEMs. In addition, the mutational hotspots we found could be potential candidates for gene screening, this will be of value for genetic counseling and genetic diagnosis of IEMs. Of course, we also need to accumulate more clinical data to standardize and optimize expanded NBS with MSMS, and the combination of MS/MS with gene screening is necessary in future.
Materials and methods
Subjects and study protocol
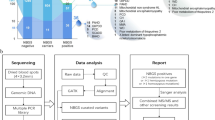
From January 2014 to December 2019, A total of 146152 newborns were referred to expanded NBS by MS/MS at the Neonatal Screening Center of Northwest Women's and Children's Hospital in **'an, Shaanxi, China. The baseline characteristics of the enrolled infants is shown in Table 1. The study protocol was reviewed and approved by the Ethics Committee of Northwest Women's and Children's Hospital. Written informed contents were obtained from all the infants' parents.
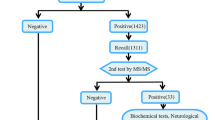
All procedures were in accordance with the technical standards for newborn screening of the Ministry of Health of China (2010). In brief, blood samples were collected from the heel stick at more than 72 h after birth, spotted onto a Whatman 903 filter paper, air-dried at room temperature, and then sent to the laboratory for MSMS analysis. Blood specimens of preterm (< 32 weeks of gestational age) or low birth weight neonates (< 1000 g) were similarly assayed, but second specimens of these infants were collected and assayed again after one month of life. Newborns with positive initial screening results were recalled for repeated test, if the second test was still positive, the infant was transferred to a specialist for further diagnosis, treatment, and follow up. During the study period, the panel of screened disorders via MSMS was not officially regulated in China, and all disorders recommended in the US panel were screened for in our center.
Expanded NBS by MSMS
Dried blood spot (DBS) samples were pre-processed following the instruction of the NeoBase non-derivative MSMS Kit (PerkinElmer, Waltham, United States). Each 3.2-mm diameter blood DBS sample was added to a PerkinElmer 96-well microtiter plate with 100 µL of extraction buffer containing amino acid and acylcarnitine internal standards. After incubation at 45 °C for 45 min with shaking in a PerkinElmer shaking incubator, 80 µL was transferred to a new microtiter plate and placed in an ACQUITY TQD mass spectrometer (Waters, Milford, MA, USA) for quantitative analysis10,12. The analytes included 11 amino acids and 31 acylcarnitine. the 11 amino acids were alanine (Ala), citrulline (Cit), arginine (Arg), glycine (Gly), leucine/isoleucine (Leu/lle/Pro-OH), methionine (Met), ornithine (Orn), phenylalanine (Phe), Valine (Val), Proline (Pro), and tyrosine (Tyr); the 31 acylcarnitine were C0, C2, C3, C3DC+C4OH, C4, C4DC+C5OH, C5, C5:1, C5DC+C6OH, C6, C6DC, C8, C8:1, C10, C10:1, C10:2, C12, C12:1, C14, C14:1, C14:2, C14OH, C16, C16:1, C16:1OH, C16OH, C18, C18:1, C18:1OH, C18:2, and C18OH.
The reference values were initially set by determining 3000 normal-term infants (0.5th–99.5th percentile), and then modified over time as more samples were analyzed and more clinical data were available to minimize both false positives and negatives. Each IEM had one or more indicators including metabolites and ratios, and the positive rules of IEMs are set by three experienced physicians (including two metabolic disease specialists and a laboratory technician). When MSMS results met the positive rules of IEMs, they were considered as positive. All the positive rules and disorder lists of IEMs were shown in Supplementary material.
Genetic analysis
Genetic analysis was performed by amplicon target capture and next generation sequencing (NGS) technology (Uni-Medica, Shenzhen, China). Peripheral blood or DBS were collected from patients diagnosed with one kind of IEM by clinical and biochemical tests. Genomic DNA was extracted using Peripheral Blood or Dry Blood Spot Nucleic Acid Extraction Kits (Uni-Medica, Shenzhen, China) according to the manufacturer's protocol. The Qubit dsDNA HS assay (Invitrogen, Carlsbad, CA, USA) was used to quantify DNA concentrations. Then, the sequencing library was established using multiplex polymerase chain reaction (PCR) (Uni-Medica, Shenzhen, China) and quantified by Qubit and quantitative PCR. Library dilution and denaturation was performed following the kit instructions and sequenced on an Illumina HiSeq X10 (Illumina, San Diego, CA) platform. The data were analyzed using the Uni-Medical GTS Analysis System. Pathogenicity analysis of each variant was performed to comply with the ACMG guidelines7. Next, all variants identified by NGS were further validated by Sanger sequencing of the parents.
The panel contained 95 genes related to IEMs (PAH, PTS, QDPR, FAH, TAT, HPD, BCKDHA, BCKDHB, DBT, CPS1, OTC, ASS1, NAGS, SLC25A13, ASL, ARG1, OAT, SLC25A15, CBS, MAT1A, PRODH, GLDC, MMACHC, MUT, MMAA, MMAB, PCCA, PCCB, IVD, GCDH, MCCC1, MCCC2, AUH, HMGCL, HLCS, BTD, ACAT1, ACADSB, ACAD8, HSD17B10, L2HGDH, SLC22A5, CPT1A, CPT2, SLC25A20, ACADS, ACADM, ACADVL, HADHA, HADHB, ETFA, ETFB, ETFDH, ETHE1, MLYCD, SMPD1, NPC1, NPC2, GBA, GLA, IDUA, IDS, GALNS, GLB1, ARSB, G6PC, SLC37A4, GAA, AGL, CYP11B1, GALC, PSAP, ARSA, ABCD1, ATP7B, GALT, ALDOB, DNAJC12, GCH1, PCBD1, MTHFR, DLD, PC, HADH, NADK2, SUCLA2, SUCLG1, LMBRD1, HCFC1, TAZ, GNMT, AHCY, MMADHC, MCEE, and, ABCD4).
Diagnosis, treatment and follow-up
In consideration of early diagnosis and intervention for positive cases, we adopted a strategy of diagnosis and treatment at the same time. Diagnosis was made by combining clinical manifestations, individualized assistant examinations and genetic testing, of which genetic testing was a very important basis. For the positive cases after recall, individualized auxiliary examinations were conducted according to disease type, which included routine blood and urine tests, liver and kidney function assessment, myocardial zymogram, blood glucose, blood ammonia and blood gas analysis. For organic acid disorders and some cases of amino acid disorders, urine gas chromatography was used for differential diagnoses.
We conducted standard treatment and follow-up for all confirmed children according to the type of disease and clinical manifestation, which mainly included: (1) combination of diet and drug therapy, such as isolated MMA, glutaric aciduria type 1 (GA-1), etc.; (2) only diet treatment, such as PKU and CD, etc.; (3) only drug treatment, such as combined MMA with homocysteinemia, PCD and holocarboxylase synthetas deficiency (HCS), etc.; and (4) only regular follow-up, such as 3MCCD and SCADD, etc. these patients were followed up every 3 ~ 6 months, including blood MSMS, urine gas chromatography, blood biochemical analysis, imaging, physical examination and development assessment, etc. All infants with negative screening results were included in the children's health care management system for follow-up.
Statistical analysis
Statistical analysis was performed using SPSS19.0. The difference of categorical data was compared using Chi-square test. The difference of measurement data was compared by t test between two groups. P < 0.05 was considered statistically significant.
Ethical approval
The study protocol was reviewed and approved by the Ethics Committee of Northwest Women's and Children's Hospital.
Informed consent
Informed consent was obtained from all families enrolled in the study.
References
Vernon, H. J. Inborn errors of metabolism: Advances in diagnosis and therapy. JAMA Pediatr. 169, 778–782 (2015).
Mak, C. M., Lee, H. C., Chan, A. Y. & Lam, C. W. Inborn errors of metabolism and expanded newborn screening: Review and update. Crit. Rev. Clin. Lab. Sci. 50, 142–162 (2013).
Saudubray, J. M. & Garcia-Cazorla, A. Inborn errors of metabolism overview: Pathophysiology, manifestations, evaluation, and management. Pediatr. Clin. N. Am. 65, 179–208 (2018).
Martinez-Morillo, E., Prieto, G. B. & Alvarez, M. F. Challenges for worldwide harmonization of newborn screening programs. Clin. Chem. 62, 689–698 (2016).
Ombrone, D., Giocaliere, E., Forni, G., Malvagia, S. & la Marca, G. Expanded newborn screening by mass spectrometry: New tests, future perspective. Mass Spectrom. Rev. 35, 71–84 (2016).
Gu, X., Wang, Z., Ye, J., Han, L. & Qiu, W. Newborn screening in China: Phenylketonuria, congenital hypothyroidism and expanded screening. Ann Acad. Med. Singapore. 37, 104–107 (2008).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Lempp, T. J. et al. Mutation and biochemical analysis of 19 probands with mut0 and 13 with mut-methylmalonic aciduria: Identification of seven novel mutations. Mol. Genet. Metab. 90, 284–290 (2007).
Yang, Y. et al. Application of next-generation sequencing following tandem mass spectrometry to expand newborn screening for inborn errors of metabolism: A multicenter study. Front Genet. 10, 86 (2019).
Yang, C. et al. Newborn screening and diagnosis of inborn errors of metabolism: A 5-year study in an Eastern Chinese population. Clin. Chim. Acta. 502, 133–138 (2020).
Lin, Y. et al. Expanded newborn screening for inherited metabolic disorders and genetic characteristics in a southern Chinese population. Clin. Chim. Acta. 494, 106–111 (2019).
Wang, T. et al. Expanded newborn screening for inborn errors of metabolism by tandem mass spectrometry in Suzhou, China: Disease spectrum, prevalence, genetic characteristics in a Chinese population. Front Genet. 10, 1 (2019).
Ma, S. et al. Expanded newborn screening for inborn errors of metabolism by tandem mass spectrometry in newborns from **nxiang City in China. J. Clin. Lab. Anal. 34, 5 (2020).
Shi, X. T. et al. Newborn screening for inborn errors of metabolism in mainland China: 30 years of experience. JIMD Rep. 6, 79–83 (2012).
Lim, J. S. et al. Inborn error of metabolism (IEM) screening in Singapore by electrospray ionization-tandem mass spectrometry (ESI/MS/MS): An 8 year journey from pilot to current program. Mol. Genet. Metab. 113, 53–61 (2014).
Yoon, H. R. et al. Screening of newborns and high-risk group of children for inborn metabolic disorders using tandem mass spectrometry in South Korea: a three-year report. Clin. Chim. Acta. 354, 167–180 (2005).
Yunus, Z. M., Rahman, S. A., Choy, Y. S., Keng, W. T. & Ngu, L. H. Pilot study of newborn screening of inborn error of metabolism using tandem mass spectrometry in Malaysia: Outcome and challenges. J. Pediatr. Endocrinol. Metab. 29, 1031–1039 (2016).
Yamaguchi, S. Newborn screening in Japan: Restructuring for the New Era. Ann. Acad. Med. Singapore 37, 13–15 (2008).
Huang, X., Yang, L., Tong, F., Yang, R. & Zhao, Z. Screening for inborn errors of metabolism in high-risk children: A 3-year pilot study in Zhejiang Province, China. BMC Pediatr. 12, 18 (2012).
Niu, D. et al. Nationwide survey of extended newborn screening by tandem mass spectrometry in Taiwan. J. Inherit. Metab. Dis. 33, S295–S305 (2010).
Lee, H. C. et al. Analysis of inborn errors of metabolism: Disease spectrum for expanded newborn screening in Hong Kong. Chin. Med. J. 124, 983–989 (2011).
Liu, N. et al. Spectrum of PAH gene variants among a population of Han Chinese patients with phenylketonuria from Northern China. BMC Med. Genet. 18, 108 (2017).
Chen, T. et al. Mutational and phenotypic spectrum of phenylalanine hydroxylase deficiency in Zhejiang Province, China. Sci Rep. 8, 17137 (2018).
Zhao, D. et al. Screening results of neonatal methylmalonic acidemia (MMA) of 349 858 cases in Henan Province. Chin. J. Birth Health Heredity. 24, 86–87 (2016).
Hong, F. et al. Screening for newborn organic aciduria in Zhejiang Province: Prevalence, outcome and follow-up. Zhejiang Da Xue Xue Bao Yi Xue Ban. 46, 240–247 (2017).
Zhou, W., Li, H., Wang, C., Wang, X. & Gu, M. Newborn screening for methylmalonic acidemia in a Chinese population: Molecular genetic confirmation and genotype phenotype correlations. Front. Genet. 9, 726 (2018).
Liu, Y. et al. Heterogeneous phenotypes, genotypes, treatment and prevention of 1 003 patients with methylmalonic acidemia in the Mainland of China. Zhonghua Er Ke Za Zhi. 56, 414–420 (2018).
Hu, S., Mei, S. Y., Bai, Y. & Kong, X. D. Analysis of MUT gene mutation and prenatal diagnosis for 20 pedigrees affected with isolated methylmalonic aciduria. Chin. J. Med. Genet. 8, 471–474 (2018).
Kang, L. et al. A study on a cohort of 301 Chinese patients with isolated methylmalonic acidemia. J. Inherit. Metab. Dis. 43, 409–423 (2020).
Han, L. et al. Spectrum analysis of common inherited metabolic diseases in Chinese patients screened and diagnosed by tandem mass spectrometry. J. Clin. Lab. Anal. 29, 162–168 (2015).
Lin, W. et al. Molecular diagnosis of pediatric patients with citrin deficiency in China: SLC25A13 mutation spectrum and the geographic distribution. Sci. Rep. 6, 1 (2016).
Lin, Y. et al. Combining newborn metabolic and genetic screening for neonatal intrahepatic cholestasis caused by citrin deficiency. J. Inherit. Metab. Dis. 43, 467–477 (2020).
Kaiser, J. Baby genome screening needs more time to gestate. Science 354, 398–399 (2016).
Zhou, W. et al. Biochemical, molecular, and clinical characterization of patients with primary carnitine deficiency via large-scale newborn screening in Xuzhou area. Front. Pediatr. 7, 50 (2019).
Giroux, S. et al. Assessment of the prevalence of the 985A>G MCAD mutation in the French-Canadian population using allele-specific PCR. Clin. Genet. 71, 569–575 (2007).
Tajima, G. et al. Screening of MCAD deficiency in Japan: 16 years’ experience of enzymatic and genetic evaluation. Mol. Genet. Metab. 119, 322–328 (2016).
Woo, H. I. et al. Clinical, biochemical and genetic analyses in two Korean patients with medium-chain acyl-CoA dehydrogenase deficiency. Korean J. Lab. Med. 31, 54–60 (2011).
Shibata, N. et al. Diversity in the incidence and spectrum of organic acidemias, fatty acid oxidation disorders, and amino acid disorders in Asian Countries: Selective screening vs expanded newborn screening. Mol. Genet. Metab. Rep. 16, 5–10 (2018).
McHugh, D. et al. Clinical validation of cutoff target ranges in newborn screening of metabolic disorders by tandem mass spectrometry: A worldwide collaborative project. Genet. Med. 13, 230–254 (2011).
Marquardt, G. et al. Enhanced interpretation of newborn screening results without analyte cutoff values. Genet. Med. 14, 648–655 (2012).
Acknowledgements
We thank all colleagues for their contributions, The authors would like to acknowledge Shenzhen Uni-medica Co.,Ltd. for technical supports in genetic analysis.
Funding
Shaanxi Key R & D Program Project (2017SF-206).
Author information
Authors and Affiliations
Contributions
R.Z. conceived and designed the research, performed the statistical analysis, and wrote the manuscript. R.Q. participated in its design and coordination and helped draft the manuscript. C.S., Y.Z., R.W., W.Y., L.Y., B.W. and N.C. conducted experiments and collected data. X.M. carried out genetic consultations. F.L. and M.F. took part in diagnosis and treatment of infants with IEMs. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, R., Qiang, R., Song, C. et al. Spectrum analysis of inborn errors of metabolism for expanded newborn screening in a northwestern Chinese population. Sci Rep 11, 2699 (2021). https://doi.org/10.1038/s41598-021-81897-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-81897-y
- Springer Nature Limited