Abstract
Acquisition of a lipid-laden phenotype by immune cells has been defined in infectious diseases and atherosclerosis but remains largely uncharacterized in cancer. Here, in breast cancer models, we found that neutrophils are induced to accumulate neutral lipids upon interaction with resident mesenchymal cells in the premetastatic lung. Lung mesenchymal cells elicit this process through repressing the adipose triglyceride lipase (ATGL) activity in neutrophils in prostaglandin E2-dependent and -independent manners. In vivo, neutrophil-specific deletion of genes encoding ATGL or ATGL inhibitory factors altered neutrophil lipid profiles and breast tumor lung metastasis in mice. Mechanistically, lipids stored in lung neutrophils are transported to metastatic tumor cells through a macropinocytosis–lysosome pathway, endowing tumor cells with augmented survival and proliferative capacities. Pharmacological inhibition of macropinocytosis significantly reduced metastatic colonization by breast tumor cells in vivo. Collectively, our work reveals that neutrophils serve as an energy reservoir to fuel breast cancer lung metastasis.







Similar content being viewed by others
Data availability
RNA-seq data are deposited in ArrayExpress and are available under accession code E-MTAB-9128. For lung metastasis-free survival analysis of patients with breast cancer, the published microarray dataset was used (GSE2603). For correlation analysis of signature genes in breast cancer lung metastasis samples, the published microarray dataset (GSE14018) was used. All other data supporting the findings of this study are available within the article and its supplementary information files and on reasonable request from the corresponding author. A Nature Research Reporting Summary for this article is available as a supplementary information file. Source data are provided with this paper.
References
Liu, Y. & Cao, X. Characteristics and significance of the pre-metastatic niche. Cancer Cell 30, 668–681 (2016).
Peinado, H. et al. Pre-metastatic niches: organ-specific homes for metastases. Nat. Rev. Cancer 17, 302–317 (2017).
Wculek, S. K. & Malanchi, I. Neutrophils support lung colonization of metastasis-initiating breast cancer cells. Nature 528, 413–417 (2015).
Coffelt, S. B. et al. IL-17-producing γδ T cells and neutrophils conspire to promote breast cancer metastasis. Nature 522, 345–348 (2015).
Albrengues, J. et al. Neutrophil extracellular traps produced during inflammation awaken dormant cancer cells in mice. Science 361, eaao4227 (2018).
Spiegel, A. et al. Neutrophils suppress intraluminal NK cell–mediated tumor cell clearance and enhance extravasation of disseminated carcinoma cells. Cancer Discov. 6, 630–649 (2016).
Massagué, J. & Obenauf, A. C. Metastatic colonization by circulating tumour cells. Nature 529, 298–306 (2016).
Dupuy, F. et al. PDK1-dependent metabolic reprogramming dictates metastatic potential in breast cancer. Cell Metab. 22, 577–589 (2015).
Christen, S. et al. Breast cancer-derived lung metastases show increased pyruvate carboxylase-dependent anaplerosis. Cell Rep. 17, 837–848 (2016).
Loo, J. M. et al. Extracellular metabolic energetics can promote cancer progression. Cell 160, 393–406 (2015).
Nieman, K. M. et al. Adipocytes promote ovarian cancer metastasis and provide energy for rapid tumor growth. Nat. Med. 17, 1498–1503 (2011).
Messmer, M. N., Netherby, C. S., Banik, D. & Abrams, S. I. Tumor-induced myeloid dysfunction and its implications for cancer immunotherapy. Cancer Immunol. 64, 1–13 (2015).
Heckmann, B. L., Zhang, X., **e, X. & Liu, J. The G0/G1 switch gene 2 (G0S2): regulating metabolism and beyond. Biochim. Biophys. Acta 1831, 276–281 (2013).
Padmanabha Das, K. M. et al. Hypoxia-inducible lipid droplet-associated protein inhibits adipose triglyceride lipase. J. Lipid Res. 59, 531–541 (2018).
Munir, R., Lisec, J., Swinnen, J. V. & Zaidi, N. Lipid metabolism in cancer cells under metabolic stress. Br. J. Cancer 120, 1090–1098 (2019).
Vallochi, A. L., Teixeira, L., Oliveira, K. D. S., Maya-Monteiro, C. M. & Bozza, P. T. Lipid droplet, a key player in host–parasite interactions. Front. Immunol. 9, 1022 (2018).
Chistiakov, D. A., Melnichenko, A. A., Myasoedova, V. A., Grechko, A. V. & Orekhov, A. N. Mechanisms of foam cell formation in atherosclerosis. J. Mol. Med. 95, 1153–1165 (2017).
Missaglia, S., Coleman, R. A., Mordente, A. & Tavian, D. Neutral lipid storage diseases as cellular model to study lipid droplet function. Cells 8, 187 (2019).
Nielsen, T. S., Jessen, N., Jørgensen, J. O., Møller, N. & Lund, S. Dissecting adipose tissue lipolysis: molecular regulation and implications for metabolic disease. J. Mol. Endocrinol. 52, R199–R222 (2014).
Zhang, X. H. et al. Latent bone metastasis in breast cancer tied to Src-dependent survival signals. Cancer Cell 16, 67–78 (2009).
Charoentong, P. et al. Pan-cancer immunogenomic analyses reveal genotype-immunophenotype relationships and predictors of response to checkpoint blockade. Cell Rep. 18, 248–262 (2017).
Nakanishi, M. & Rosenberg, D. W. Multifaceted roles of PGE2 in inflammation and cancer. Semin. Immunopathol. 35, 123–137 (2013).
Sugimoto, Y. & Narumiya, S. Prostaglandin E receptors. J. Biol. Chem. 282, 11613–11617 (2007).
Dennis, E. A. & Norris, P. C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 15, 511–523 (2015).
Wang, D. & Dubois, R. N. Eicosanoids and cancer. Nat. Rev. Cancer 10, 181–193 (2010).
Zhang, X. et al. Inhibition of intracellular lipolysis promotes human cancer cell adaptation to hypoxia. Elife 6, e31132 (2017).
Knight, M., Braverman, J., Asfaha, K., Gronert, K. & Stanley, S. Lipid droplet formation in Mycobacterium tuberculosis infected macrophages requires IFN-γ/HIF-1α signaling and supports host defense. PLoS Pathog. 14, e1006874 (2018).
Palazon, A., Goldrath, A. W., Nizet, V. & Johnson, R. S. HIF transcription factors, inflammation, and immunity. Immunity 41, 518–528 (2014).
Passegue, E., Wagner, E. F. & Weissman, I. L. JunB deficiency leads to a myeloproliferative disorder arising from hematopoietic stem cells. Cell 119, 431–443 (2004).
den Brok, M. H., Raaijmakers, T. K., Collado-Camps, E. & Adema, G. J. Lipid droplets as immune modulators in myeloid cells. Trends Immunol. 39, 380–392 (2018).
Veglia, F. et al. Fatty acid transport protein 2 reprograms neutrophils in cancer. Nature 569, 73–78 (2019).
Stewart, T. J. & Abrams, S. I. Altered immune function during long-term host-tumor interactions can be modulated to retard autochthonous neoplastic growth. J. Immunol. 179, 2851–2859 (2007).
DeBerardinis, R. J., Lum, J. J., Hatzivassiliou, G. & Thompson, C. B. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 7, 11–20 (2008).
Pascual, G. et al. Targeting metastasis-initiating cells through the fatty acid receptor CD36. Nature 541, 41–45 (2017).
Zhang, M. et al. Adipocyte-derived lipids mediate melanoma progression via FATP proteins. Cancer Discov. 8, 1006–1025 (2018).
Wang, Y. Y. et al. Mammary adipocytes stimulate breast cancer invasion through metabolic remodeling of tumor cells. JCI insight 2, e87489 (2017).
Adrover, J. M., Nicolas-Avila, J. A. & Hidalgo, A. Aging: a temporal dimension for neutrophils. Trends Immunol. 37, 334–345 (2016).
Schild, T., Low, V., Blenis, J. & Gomes, A. P. Unique metabolic adaptations dictate distal organ-specific metastatic colonization. Cancer Cell 33, 347–354 (2018).
Flaherty, S. E. III, Grijalva, A., Xu, X., Ables, E., Nomani, A. & Ferrante, A. W. Jr. A lipase-independent pathway of lipid release and immune modulation by adipocytes. Science 363, 989–993 (2019).
Lim, J. P. & Gleeson, P. A. Macropinocytosis: an endocytic pathway for internalising large gulps. Immunol. Cell Biol. 89, 836–843 (2011).
Commisso, C. et al. Macropinocytosis of protein is an amino acid supply route in Ras-transformed cells. Nature 497, 633–637 (2013).
Kim, S. M. et al. PTEN deficiency and AMPK activation promote nutrient scavenging and anabolism in prostate cancer cells. Cancer Discov. 8, 866–883 (2018).
Rejman, J., Bragonzi, A. & Conese, M. Role of clathrin- and caveolae-mediated endocytosis in gene transfer mediated by lipo- and polyplexes. Mol. Ther. 12, 468–474 (2005).
Shaul, M. E. & Fridlender, Z. G. Tumour-associated neutrophils in patients with cancer. Nat. Rev. Clin. Oncol. 16, 601–620 (2019).
Minn, A. J. et al. Genes that mediate breast cancer metastasis to lung. Nature 436, 518–524 (2005).
Spiegelman, B. M. & Flier, J. S. Obesity and the regulation of energy balance. Cell 104, 531–543 (2001).
Lehuede, C., Dupuy, F., Rabinovitch, R., Jones, R. G. & Siegel, P. M. Metabolic plasticity as a determinant of tumor growth and metastasis. Cancer Res. 76, 5201–5208 (2016).
Zhang, Y. & Commisso, C. Macropinocytosis in cancer: a complex signaling network. Trends Cancer 5, 332–334 (2019).
Ohs, I. et al. Restoration of natural killer cell antimetastatic activity by IL12 and checkpoint blockade. Cancer Res. 77, 7059–7071 (2017).
Basnet, H. Flura-seq identifies organ-specific metabolic adaptations during early metastatic colonization. Elife 8, e43627 (2019).
Breitkopf, S. B. et al. A relative quantitative positive/negative ion switching method for untargeted lipidomics via high resolution LC-MS/MS from any biological source. Metabolomics 13, 30 (2017).
Acknowledgements
We thank S.I. Abrams (Roswell Park Comprehensive Cancer Center) for providing the AT3 breast cancer cell line, Y. Kang (Princeton University) for providing the MDA-4175 breast cancer cell line and R.A. Weinberg (Whitehead Institute for Biomedical Research) for providing the lentiviralvector expressing mouse Csf3. This work was supported by grants from the National Institutes of Health (R00-CA188093 to G.R.; R37-CA237307 to G.R.; P30-CA034196 to G.R., L.D.S. and S.L.; R35−GM133562 to S.L.; and R24-OD026440 and R01-AI132963 to L.D.S.), the U.S. Department of Defense (W81XWH-18-1-0013 to G.R.), Leukemia Research Foundation New Investigator Grant (to S.L.) and The Jackson Laboratory Director’s Innovation Fund (19000-17-31 to S.L.). Q.L. is supported by the Pyewacket Fund at The Jackson Laboratory. We acknowledge critical comments from E. T. Liu, N. A. Rosenthal, K. Seburn and C. Robinett. We also thank G. Stafford for RNA-seq analysis and W. Schott for cell sorting, as well as The Jackson Laboratory Scientific Service for assistance.
Author information
Authors and Affiliations
Contributions
G.R., P.L. and M.L. conceived the project, designed the study and performed the data analysis. P.L., M.L., J.S., Z.G., L.H. and Q.L. performed the experiments. L.D.S. provided critical assistance on experimental design. X.H.-F.Z. provided essential experimental materials. B.L. and X.H.-F.Z. provided assistance on clinical dataset analysis and experimental design; X.C. and S.L. performed the clinical dataset analysis and provided assistance on statistical analysis. G.R., P.L., M.L. and L.D.S. interpreted the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Peer reviewer reports are available. Zoltan Fehervari was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
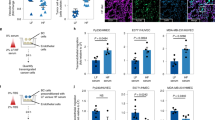
Extended Data Fig. 1 Lung neutrophils acquire a lipid-laden phenotype in the orthotopic 4T1 tumor model.
a–d, Transcriptional profiles of BM, PB and lung neutrophils sorted from the orthotopic 4T1 tumor-bearing BALB/cJ mice (n = 3). Volcano plots showing fold-change and p-value for the comparison of lung versus BM neutrophils (a), and PB versus BM neutrophils (b) based on the RNA-seq data. P values were determined by unpaired two-tailed t-test and smaller than 0.05 were considered significant. A schematic diagram showing the major pathway and key factors in lipid metabolism (c). A heat map depicts expression levels of the major lipid metabolic genes (d). e, Upper: a diagram showing the pre-metastatic and metastatic stages in the orthotopic 4T1 tumor model; lower: Representative immunostaining of neutrophils (Ly6G) in lung sections of naïve and 4T1 tumor-bearing BALB/cJ mice (n = 4 from 2 independent experiments). Scale bars, 20 μm. f, Intracellular lipids in neutrophils detected by BODIPY 493/503 staining under microscope (n = 4 mice from 2 independent experiments). Scale bars, 5 μm. g, The flow cytometry gating strategy is shown: neutrophils were identified as the Ly6G+Ly6Clow cell population which was gated on CD45+CD11b+ cells. h, Measurement of total lipid contents in neutrophils isolated from naïve and 4T1 tumor-bearing mice by BODIPY 493/503 staining and flow cytometry (n = 6). i, Cellular TG contents in neutrophils (n = 5). In f and i, neutrophils were isolated from the indicated tissues and organs of orthotopic 4T1 tumor-bearing mice at the pre-metastatic stage. n represents biologically independent animals. Data are mean ± s.e.m. and P values were determined by one-way ANOVA with Tukey’s multiple comparisons test (h and i). ns, not significant.
Extended Data Fig. 2 Up-regulation of genes encoding ATGL inhibitory factors led to reduced TG hydrolysis in pre-metastatic lung-infiltrating neutrophils.
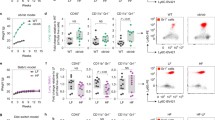
a–h, Levels of total triglyceride (TG) (a), cholesteryl ester (CE) (b), phosphatidylethanolamine (PE) (c), phosphatidylcholine (PC) (d), phosphatidylserine (PS) (e), phosphatidylinositol (PI) (f), ceramides (Cer) (g) and phosphatidylglycerol (PG) (h) in PB and lung neutrophils isolated from the pre-metastatic stage of orthotopic 4T1 tumor-bearing mice (n = 5), were determined by liquid chromatography-mass spectrometry. Relative levels of these lipid were also shown in Fig. 1f. The exact values of specific chemical species for each lipid type were provided in Source Data file. i-l, Neutrophils were isolated from the BM, PB and lung tissues of orthotopic 4T1 tumor-bearing mice (n = 5) at the pre-metastatic stage and compared for relative mRNA expression (to Rps18) of Hilpda, Cidec, G0s2, Atgl and Abhd5 by qRT-PCR (i); HILPDA, CIDEC and G0S2 protein expression by Western blotting with GAPDH as a loading control (j); relative TG hydrolase activity (k); and cellular lipase activity (l). n represents biologically independent animals. Data are mean ± s.e.m. and P values were determined by one-way ANOVA with Tukey’s multiple comparisons test (i and l) or unpaired two-tailed t-test (k). ns, not significant.
Extended Data Fig. 3 Neutrophil-specific genetic ablation of Hilpda or Cidec did not lead to significant changes on lung metastases of breast cancer in vivo.
a–f, The neutrophil-targeting cKO mice (a-c, Hilpda-cKO; d–f, Cidec-cKO), and their wild type littermates were orthotopically implanted with AT3-g-csf cells. On day 15, the relative lipid levels in lung neutrophils were determined by BODIPY 493/503 staining and flow cytometry (a and d) (n = 5 per group), and the resected primary tumors were weighed (b and e). At the end point (day 30), spontaneous lung metastases of WT and cKO mice were quantified (c and f). n = 13 (WT group) and 14 (Hilpda-cKO group) for b, c; n = 15 (WT group) and 14 (Cidec-cKO group) for e, n = 10 (WT group) and 12 (Cidec-cKO group) for f. g, A schematic diagram showing the modified experimental lung metastasis model employed in this study. Mice were first orthotopically implanted with non-labeled AT3-g-csf cells to induce a neutrophil-high host condition. Luciferase-labeled AT3-Luc cells were then implanted intravenously on day 10, a time point within the pre-metastatic stage. On day 15, the primary tumors were resected. At the end point, the lung metastatic progression of AT3-Luc cells was detected by ex vivo BLI. h, Following the above modified experimental lung metastasis model, the lung metastatic progression of AT3-Luc cells in WT and Cidec-cKO recipient mice was determined by ex vivo BLI. Representative BLI images of the harvested lungs are shown and red lines indicate blank wells without lung tissues (left) (n = 11). n represents biologically independent animals. Data are mean ± s.e.m. and P values were determined by unpaired two-tailed t-test. ns, not significant.
Extended Data Fig. 4 Tumor cells absorb lipids from lipid-laden lung neutrophils.
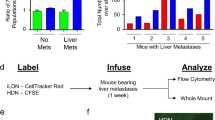
a, Representative images from 2 biologically independent experiments showing lipid transfer from BODIPY FL C16-loaded neutrophils to 4T1-mCherry cells. Arrowheads indicate neutrophils. Scale bars, 20 μm. b, 4T1-mCherry or AT3-mCherry cells were mono-cultured or co-cultured with their respective tumor-bearing mice-derived neutrophils that were pre-loaded with BODIPY FL C16. Then the intensity of BODIPY in tumor cells was examined by flow cytometry. Representative of 3 biologically independent experiments. c, The cellular TG contents of AT3-mCherry cells after monoculture or co-culture with neutrophils. Data are mean ± s.d. from 3 biologically independent cell cultures. d, Measurement of the lipids in early lung-colonizing AT3-mCherry cells in G-CSF-pretreated mice without and with anti-Ly6G-based neutrophil depletion (see Methods) (n = 5). Data are mean ± s.e.m. e, The lipid transfer from BODIPY FL C16-loaded lung neutrophils to the indicated tumor cells in vivo as determined by flow cytometry (see Methods) (n = 4). Representative flow cytometry histograms are shown. f, As depicted in the left panel, AT3-mCherry or 4T1-mCherry cells were mono-cultured or co-cultured with PB or lung neutrophils. In one experimental group, lung neutrophils were pre-activated by phorbol 12-myristate 13-acetate (PMA) and hydrogen peroxide (H2O2). The total lipid levels in tumor cells were determined by BODIPY 493/503 staining and flow cytometry. Data are mean ± s.d. from 5 (4T1-mCherry) or 3 (AT3-mCherry) biologically independent cell cultures. Neutrophils used throughout this figure were isolated from the pre-metastatic stage of 4T1 tumor-bearing mice or AT3-g-csf tumor-bearing mice. n represents biologically independent animals. P values were determined by one-way ANOVA with Tukey’s multiple comparisons test. ns, not significant.
Extended Data Fig. 5 Neutrophil-derived lipids enhance the proliferative capacity of metastatic tumor cells.
a, Oxygen consumption rates (OCR) of AT3 cells, upon mono-culture or co-culture with PB or lung neutrophils (left). The amount of OCR derived from fatty acid oxidation was quantified as the magnitude of the response to etomoxir (right). b–d, Measurement of the proliferative capacities of the indicated tumor cells without and with co-culture with PB or lung neutrophils (see Methods). e–g, Comparison of the metastatic colonization potentials of the indicated tumor cells (e, AT3-Luc; f, MCF7-Luc; and g, MDA-4175-Luc) without and with co-culture with PB or lung neutrophils (see Methods). Representative BLI images of the recipient mice and quantification of BLI signals within the lung areas are shown (n = 5 for e and n = 6 for f, g). h, i, PB and lung neutrophils were isolated from AT3-g-csf tumor-bearing WT or Atgl-cKO mice (pre-metastatic stage). The intracellular lipids in neutrophils were detected by BODIPY 493/503 staining under microscope (representative of n = 5 mice). Scale bars, 5 μm (h). The cellular TG contents of neutrophils were measured (n = 5) (i). j, 4T1-Luc cells, upon co-culture with PB or lung neutrophils isolated from AT3-g-csf tumor-bearing WT or Atgl-cKO mice (pre-metastatic stage), were intravenously injected into NSG mice (see Methods). Representative BLI images of the recipient mice and quantification of BLI signals within the lung areas are shown (n = 6). k–m, As depicted in k, the effects of EIPA treatment on lung colonization by 4T1 tumor cells (l) and on primary tumor growth (m) were determined (see Methods) (n = 10). Neutrophils throughout this figure were isolated from the pre-metastatic stage of 4T1 tumor-bearing mice (c, d, f, g) or AT3-g-csf tumor-bearing mice (a, b and e), except otherwise stated. n represents biologically independent animals. Data are mean ± s.d. from 6 (a) or 4 (b–d) biologically independent cell cultures, and mean ± s.e.m. for e-g, i, j, l, m. P values were determined by two-way ANOVA with Sidak’s multiple comparisons test (b–d, mono-culture versus lung Neu co-culture), one-way ANOVA with Tukey’s multiple comparisons test (a, e–g, i and j) or unpaired two-tailed t-test (l, m). ns, not significant.
Extended Data Fig. 6 Both lipid- and neutrophil-associated gene expression signatures are related with lung metastasis in human breast cancer patients.
Kaplan–Meier plots of lung metastasis-free survival of breast cancer patients, stratified by expression of indicated gene signature sets in their primary tumors (GEO accession number: GSE2603, n = 82 patients). A risk score was calculated for each sample which was defined as a linear combination of expression values of genes in one signature set weighted by their estimated Cox model regression coefficients. If the risk score for one sample was in the top 20th percentile of the risk scores, then it was classified into high-risk group, otherwise into low-risk group. P values were calculated by a 2-sided log-rank test. The used gene signature sets are derived from Gene Ontology (GO): Biological Process of MsigDB v.7.1, including GO_POSITIVE_REGULATION_OF_LIPID_CATABOLIC_PROCESS (M14107, 25 genes); GO_POSITIVE_REGULATION_OF_LIPID_TRANSPORT (M11731, 61 genes); GO_LIPID_OXIDATION (M15880, 101 genes); GO_NEUTROPHIL_MIGRATION (M25402, 119 genes); GO_REGULATION_OF_NEUTROPHIL_CHEMOTAXIS (M29283, 30 genes); and GO_NEUTROPHIL_EXTRAVASATION (M24616, 13 genes).
Extended Data Fig. 7 A lung MC → neutrophil → tumor cell metabolic axis in the lung metastatic niche.
A schematic diagram depicts how lung neutrophils are stimulated by lung resident MCs to accumulate lipids, and in turn transport their stored lipids to metastatic tumor cells for survival and proliferation leading to accelerated lung metastasis in a breast cancer model.
Supplementary information
Source data
Source Data Fig. 1
Statistical source data
Source Data Fig. 1
Uncropped immunoblots
Source Data Fig. 2
Statistical source data
Source Data Fig. 3
Statistical source data
Source Data Fig. 4
Statistical source data
Source Data Fig. 5
Statistical source data
Source Data Fig. 6
Statistical source data
Source Data Fig. 7
Statistical source data
Source Data Extended Data Fig. 1
Statistical source data
Source Data Extended Data Fig. 2
Statistical source data
Source Data Extended Data Fig. 2
Uncropped immunoblots
Source Data Extended Data Fig. 3
Statistical source data
Source Data Extended Data Fig. 4
Statistical source data
Source Data Extended Data Fig. 5
Statistical source data
Rights and permissions
About this article
Cite this article
Li, P., Lu, M., Shi, J. et al. Lung mesenchymal cells elicit lipid storage in neutrophils that fuel breast cancer lung metastasis. Nat Immunol 21, 1444–1455 (2020). https://doi.org/10.1038/s41590-020-0783-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-020-0783-5
- Springer Nature America, Inc.
This article is cited by
-
Neutrophils in Cancer immunotherapy: friends or foes?
Molecular Cancer (2024)
-
Multi-model analysis of gallbladder cancer reveals the role of OxLDL-absorbing neutrophils in promoting liver invasion
Experimental Hematology & Oncology (2024)
-
Mechanisms underlying neutrophils adhesion to triple-negative breast cancer cells via CD11b-ICAM1 in promoting breast cancer progression
Cell Communication and Signaling (2024)
-
Role of Exosomes in Cancer and Aptamer-Modified Exosomes as a Promising Platform for Cancer Targeted Therapy
Biological Procedures Online (2024)
-
Extramedullary hematopoiesis in cancer
Experimental & Molecular Medicine (2024)