Abstract
Phosphorylation modification is required for the modulation of phytochrome B (phyB) thermal reversion, but the kinase(s) that phosphorylate(s) phyB and the biological significance of the phosphorylation are still unknown. Here we report that FERONIA (FER) phosphorylates phyB to regulate plant growth and salt tolerance, and the phosphorylation not only regulates dark-triggered photobody dissociation but also modulates phyB protein abundance in the nucleus. Further analysis indicates that phosphorylation of phyB by FER is sufficient to accelerate the conversion of phyB from the active form (Pfr) to the inactive form (Pr). Under salt stress, FER kinase activity is inhibited, leading to delayed photobody dissociation and increased phyB protein abundance in the nucleus. Our data also show that phyB mutation or overexpression of PIF5 attenuates growth inhibition and promotes plant survival under salt stress. Together, our study not only reveals a kinase that controls phyB turnover via a signature of phosphorylation, but also provides mechanistic insights into the role of the FER-phyB module in coordinating plant growth and stress tolerance.







Similar content being viewed by others
Data availability
All materials in this study are available from the corresponding author upon request. The raw data of BSA sequencing have been deposited in the NCBI BioProject database under accession number PRJNA914087. RNA-seq data have been deposited in the NCBI GEO under accession number GSE188335. Arabidopsis reference genome (TAIR10) was used in this study. Source data are provided with this paper.
References
Kami, C., Lorrain, S., Hornitschek, P. & Fankhauser, C. Light-regulated plant growth and development. Curr. Top. Dev. Biol. 91, 29–66 (2010).
Nagy, F. & Schafer, E. Phytochromes control photomorphogenesis by differentially regulated, interacting signaling pathways in higher plants. Annu. Rev. Plant Biol. 53, 329–355 (2002).
Rockwell, N. C., Su, Y. S. & Lagarias, J. C. Phytochrome structure and signaling mechanisms. Annu. Rev. Plant Biol. 57, 837–858 (2006).
Al-Sady, B., Ni, W., Kircher, S., Schafer, E. & Quail, P. H. Photoactivated phytochrome induces rapid PIF3 phosphorylation prior to proteasome-mediated degradation. Mol. Cell 23, 439–446 (2006).
Yamaguchi, R., Nakamura, M., Mochizuki, N., Kay, S. A. & Nagatani, A. Light-dependent translocation of a phytochrome B-GFP fusion protein to the nucleus in transgenic Arabidopsis. J. Cell Biol. 145, 437–445 (1999).
Bae, G. & Choi, G. Decoding of light signals by plant phytochromes and their interacting proteins. Annu. Rev. Plant Biol. 59, 281–311 (2008).
Huang, H. et al. PCH1 regulates light, temperature, and circadian signaling as a structural component of phytochrome B-photobodies in Arabidopsis. Proc. Natl Acad. Sci. USA 116, 8603–8608 (2019).
Sweere, U. et al. Interaction of the response regulator ARR4 with phytochrome B in modulating red light signaling. Science 294, 1108–1111 (2001).
Mira-Rodado, V. et al. Functional cross-talk between two-component and phytochrome B signal transduction in Arabidopsis. J. Exp. Bot. 58, 2595–2607 (2007).
Galvao, R. M. et al. Photoactivated phytochromes interact with HEMERA and promote its accumulation to establish photomorphogenesis in Arabidopsis. Genes Dev. 26, 1851–1863 (2012).
Chen, M. et al. Arabidopsis HEMERA/pTAC12 initiates photomorphogenesis by phytochromes. Cell 141, 1230–1240 (2010).
Medzihradszky, M. et al. Phosphorylation of phytochrome B inhibits light-induced signaling via accelerated dark reversion in Arabidopsis. Plant Cell 25, 535–544 (2013).
Nito, K., Wong, C. C., Yates, J. R. 3rd & Chory, J. Tyrosine phosphorylation regulates the activity of phytochrome photoreceptors. Cell Rep. 3, 1970–1979 (2013).
Reed, J. W., Nagpal, P., Poole, D. S., Furuya, M. & Chory, J. Mutations in the gene for the red/far-red light receptor phytochrome B alter cell elongation and physiological responses throughout Arabidopsis development. Plant Cell 5, 147–157 (1993).
Jung, J.-H. et al. Phytochromes function as thermosensors in Arabidopsis. Science 354, 886–889 (2016).
Legris, M. et al. Phytochrome B integrates light and temperature signals in Arabidopsis. Science 354, 897–900 (2016).
Campos, M. L. et al. Rewiring of jasmonate and phytochrome B signalling uncouples plant growth-defense tradeoffs. Nat. Commun. 7, 12570 (2016).
Zhao, C., Zhang, H., Song, C., Zhu, J. K. & Shabala, S. Mechanisms of plant responses and adaptation to soil salinity. Innovation 1, 100017 (2020).
Franck, C. M., Westermann, J. & Boisson-Dernier, A. Plant malectin-like receptor kinases: from cell wall integrity to immunity and beyond. Annu. Rev. Plant Biol. 69, 301–328 (2018).
Duan, Q. et al. FERONIA controls pectin- and nitric oxide-mediated male-female interaction. Nature 579, 561–566 (2020).
Escobar-Restrepo, J. M. et al. The FERONIA receptor-like kinase mediates male-female interactions during pollen tube reception. Science 317, 656–660 (2007).
Li, C. et al. Glycosylphosphatidylinositol-anchored proteins as chaperones and co-receptors for FERONIA receptor kinase signaling in Arabidopsis. eLife 4, e06587 (2015).
Stegmann, M. et al. The receptor kinase FER is a RALF-regulated scaffold controlling plant immune signaling. Science 355, 287–289 (2017).
Zhu, S. et al. The RALF1-FERONIA complex phosphorylates eIF4E1 to promote protein synthesis and polar root hair growth. Mol. Plant 13, 698–716 (2020).
Liao, H., Tang, R., Zhang, X., Luan, S. & Yu, F. FERONIA receptor kinase at the crossroads of hormone signaling and stress responses. Plant Cell Physiol. 58, 1143–1150 (2017).
Zhao, C. et al. Leucine-rich repeat extensin proteins regulate plant salt tolerance in Arabidopsis. Proc. Natl Acad. Sci. USA 115, 13123–13128 (2018).
Zhao, C. et al. The LRXs-RALFs-FER module controls plant growth and salt stress responses by modulating multiple plant hormones. Natl Sci. Rev. 8, nwaa149 (2021).
Huq, E., Al-Sady, B. & Quail, P. H. Nuclear translocation of the photoreceptor phytochrome B is necessary for its biological function in seedling photomorphogenesis. Plant J. 35, 660–664 (2003).
Gigli-Bisceglia, N., van Zelm, E., Huo, W., Lamers, J. & Testerink, C. Arabidopsis root responses to salinity depend on pectin modification and cell wall sensing. Development 149, dev200363 (2022).
Wagner, D., Tepperman, J. M. & Quail, P. H. Overexpression of phytochrome B induces a short hypocotyl phenotype in transgenic Arabidopsis. Plant Cell 3, 1275–1288 (1991).
Horner, M., Yousefi, O. S., Schamel, W. W. A. & Weber, W. Production, purification and characterization of recombinant biotinylated phytochrome B for extracellular optogenetics. Bio Protoc. 10, e3541 (2020).
Su, Y. S. & Lagarias, J. C. Light-independent phytochrome signaling mediated by dominant GAF domain tyrosine mutants of Arabidopsis phytochromes in transgenic plants. Plant Cell 19, 2124–2139 (2007).
Wang, Y., Bian, Z., Pan, T., Cao, K. & Zou, Z. Improvement of tomato salt tolerance by the regulation of photosynthetic performance and antioxidant enzyme capacity under a low red to far-red light ratio. Plant Physiol. Biochem. 167, 806–815 (2021).
Mo, W. et al. PHYTOCHROME-INTERACTING FACTOR-LIKE14 and SLENDER RICE1 interaction controls seedling growth under salt stress. Plant Physiol. 184, 506–517 (2020).
Junior, C. A. S., D’Amico‐Damião, V. & Carvalho, R. F. Phytochrome type B family: the abiotic stress responses signaller in plants. Ann. Appl. Biol. 178, 135–148 (2020).
Feng, W. et al. The FERONIA receptor kinase maintains cell-wall integrity during salt stress through Ca2+ signaling. Curr. Biol. 28, 666–675.e5 (2018).
Vaahtera, L., Schulz, J. & Hamann, T. Cell wall integrity maintenance during plant development and interaction with the environment. Nat. Plants 5, 924–932 (2019).
Kwon, C. T. et al. Functional deficiency of phytochrome B improves salt tolerance in rice. Environ. Exp. Bot. 148, 100–108 (2018).
Yang, T., Lv, R., Li, J., Lin, H. & **, D. Phytochrome A and B negatively regulate salt stress tolerance of Nicotiana tobacum via ABA-jasmonic acid synergistic cross-talk. Plant Cell Physiol. 59, 2381–2393 (2018).
Banani, S. F., Lee, H. O., Hyman, A. A. & Rosen, M. K. Biomolecular condensates: organizers of cellular biochemistry. Nat. Rev. Mol. Cell Biol. 18, 285–298 (2017).
Wang, X. et al. A photoregulatory mechanism of the circadian clock in Arabidopsis. Nat. Plants 7, 1397–1408 (2021).
Chen, D. et al. Integration of light and temperature sensing by liquid-liquid phase separation of phytochrome B. Mol. Cell 82, 3015–3029.e6 (2022).
**ao, Y. et al. Mechanisms of RALF peptide perception by a heterotypic receptor complex. Nature 572, 270–274 (2019).
Haruta, M., Sabat, G., Stecker, K., Minkoff, B. B. & Sussman, M. R. A peptide hormone and its receptor protein kinase regulate plant cell expansion. Science 343, 408–411 (2014).
Chen, J. et al. FERONIA interacts with ABI2-type phosphatases to facilitate signaling cross-talk between abscisic acid and RALF peptide in Arabidopsis. Proc. Natl Acad. Sci. USA 113, E5519–E5527 (2016).
Minkoff, B. B. et al. A cell-free method for expressing and reconstituting membrane proteins enables functional characterization of the plant receptor-like protein kinase FERONIA. J. Biol. Chem. 292, 5932–5942 (2017).
Guo, H. et al. FERONIA receptor kinase contributes to plant immunity by suppressing jasmonic acid signaling in Arabidopsis thaliana. Curr. Biol. 28, 3316–3324.e6 (2018).
Duan, Q., Kita, D., Li, C., Cheung, A. Y. & Wu, H. M. FERONIA receptor-like kinase regulates RHO GTPase signaling of root hair development. Proc. Natl Acad. Sci. USA 107, 17821–17826 (2010).
Zhang, X., Yang, Z., Wu, D. & Yu, F. RALF-FERONIA signaling: linking plant immune response with cell growth. Plant Commun. 1, 100084 (2020).
Kim, J. I. et al. Phytochrome phosphorylation modulates light signaling by influencing the protein-protein interaction. Plant Cell 16, 2629–2640 (2004).
Ryu, J. S. et al. Phytochrome-specific type 5 phosphatase controls light signal flux by enhancing phytochrome stability and affinity for a signal transducer. Cell 120, 395–406 (2005).
Shalitin, D. et al. Regulation of Arabidopsis cryptochrome 2 by blue-light dependent phosphorylation. Nature 417, 758–763 (2002).
Viczian, A. et al. Differential phosphorylation of the N-terminal extension regulates phytochrome B signaling. New Phytol. 225, 1635–1650 (2020).
Wang, J. et al. A DNA methylation reader-chaperone regulator-transcription factor complex activates OsHKT1;5 expression during salinity stress. Plant Cell 32, 3535–3558 (2020).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Allen, J. J. et al. A semisynthetic epitope for kinase substrates. Nat. Methods 4, 511–516 (2007).
Qian, C. et al. Dual-source nuclear monomers of UV-B light receptor direct photomorphogenesis in Arabidopsis. Mol. Plant 9, 1671–1674 (2016).
Du, Z., Zhou, X., Ling, Y., Zhang, Z. & Su, Z. agriGO: a GO analysis toolkit for the agricultural community. Nucleic Acids Res. 38, W64–W70 (2010).
Acknowledgements
We thank H. Yang (Shanghai Normal University, China) for providing phyBpro::phyB-Myc plasmid, R. Fang (Institute of Microbiology, CAS, China) for providing OsphyB mutant, and J. Clark Lagarias (University of California, Davis, USA) for providing YHB seeds. This work was supported by National Natural Science Foundation of China Grant No. 32070295 (to C.Z.), Shanghai Pujiang Program Grant No. 20PJ1414800 (to C.Z.), Shanghai Agriculture Applied Technology Development Program Grant No. X20200101 (to C.Z.), and Strategic Priority Research Program of the Chinese Academy of Sciences Grant No. XDA27040104 (to J.-K.Z.).
Author information
Authors and Affiliations
Contributions
X.L. and C.Z. conceived and designed the experiments; X.L., W.J., Y.L., H.N., C.L., J.L. and M.L. performed experiments; X.L., L.C., L.P. and B.Z. conducted bioinformatic analysis; X.L. and R.L. performed proteomics analysis; L.T., H.W., J.Y., P.W., H.L., J.-K.Z. and C.Z. analysed data; X.L., J.-K.Z. and C.Z. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks Ferenc Nagy, Eunkyoo Oh, Christa Testerink and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 slrx620 mutation suppresses the phenotypes of lrx345 mutant.
a, Phenotype of wild type, lrx345, and slrx620 mutant grown on 1/2 MS medium with or without NaCl (120 mM) under white light conditions (16 h light/8 h dark cycle). b, Quantification of the survival rate of seedlings grown on NaCl medium. Values are the means ± SD of three biological replicates. c, Representative images of wild type, lrx345, and slrx620 mutant grown on soils for 22 days under LD conditions. Scale bar, 1 cm. d, Quantification of the petiole length of plants shown in c. Values are the means ± SD (n = 11 seedlings). e, RT–qPCR analysis of the transcript levels of PDF1.2 and PDF1.3 genes in each genotype. Values are the means ± SD of three biological replicates. f, Identification of mutations in slrx620 mutant by bulked segregant analysis (BSA). Red asterisk indicates the location of the mutated gene that is associated with the slrx620 mutant phenotypes. g, Diagram illustrating the genomic structure of phyB and the mutation site in the slrx620 mutant. h. Hypocotyl growth of seedlings grown under white light conditions for 4 days. Scale bar, 2 mm. i, Quantification of the hypocotyl length of the seedlings shown in h. Values are the means ± SD (n = 49-63 seedlings). Asterisks in b, d and e indicate statistically significant differences (**P < 0.01, Student’s t test, two-sided). Different letters in i indicate statistically significant differences (P < 0.01, one-way ANOVA).
Extended Data Fig. 2 phyB is involved in FER-mediated regulation of plant growth and salt tolerance.
a, Representative images of wild type, fer-4, phyB-9, and phyB-9 fer-4 siliques. Scale bar, 0.5 mm. b, Representative images of wild type, fer-4, phyB-9 fer-4, and 35 S::phyB-GFP/phyB-9 fer-4 grown on soils for 22 days under white light conditions (16 h light/8 h dark cycle). Scale bar, 1 cm. c, Quantification of the petiole length of plants shown in b. Values are the means ± SD (n = 21 plants). d, Phenotype of wild type, fer-4, phyB-9 fer-4, and 35 S::phyB-GFP/phyB-9 fer-4 grown on 1/2 MS media supplemented with or without NaCl (120 mM) under white light conditions. e, Quantification of the survival rate of seedlings grown on NaCl medium. Values are the means ± SD of three biological replicates. f, Phenotype of wild type, phyB-9, and 35 S::phyB-GFP/phyB-9 transgenic seedlings grown on 1/2 MS medium with or without NaCl (120 mM) under white light conditions. g, Quantification of the survival rate of seedlings grown on NaCl medium. Values are the means ± SD of three biological replicates. h, Representative images of wild type, fer-4, phyB-9, phyB-9 fer-4, 35 S::phyB-GFP/phyB-9, and 35 S::phyB-GFP/phyB-9 fer-4 seedlings grown on soils for 35 days under white light conditions. Scale bar, 5 cm. i. Quantification of the plant height as shown in h. Values are the means ± SD (n = 18-25 plants). Different letters in c, e, g and i indicate statistically significant differences (P < 0.01, one-way ANOVA).
Extended Data Fig. 3 phyB mutation suppresses the salt-hypersensitivity of the lrx345 and fer-4 mutants under monochromatic red light illumination.
a, Phenotype of each genotype grown on 1/2 MS media supplemented with or without NaCl (120 mM) under continuous red light. b, Quantification of the survival rate of seedlings grown on NaCl medium as shown in a. Values are the means ± SD of three biological replicates. c, Phenotype of wild type, lrx345, and fer-4 seedlings grown on 1/2 MS and 1/2 MS + NaCl (120 mM) media under continuous high red-light intensity (~15 μmol m-2 sec-1) or continuous low red-light intensity (~1 μmol m-2 sec-1). d, Quantification of the survival rate of seedlings grown on NaCl medium as shown in c. Values are the means ± SD of three biological replicates. e, Fluence rate response curves to analyze hypocotyl elongation inhibition under different red light fluence rates at 22 °C. Wild type and fer-4 mutant were grown on 1/2 MS medium without sucrose in dark or continuous red light for 4 days. Relative hypocotyl length of the seedings grown under different red light fluence rates was calculated by comparing with that under dark conditions. Values are the means ± SD (n = 32-41 seedlings). Asterisks in b and d indicate statistically significant differences (**P < 0.01, Student’s t test, two-sided).
Extended Data Fig. 4 Transcriptomic profiling reveals that FER regulates a subset of gene expression via phyB.
a, Venn diagrams showing the overlapped genes that were up-regulated in fer-4 mutant compared with wild type, but down-regulated in phyB-9 fer-4 compared with fer-4 mutant. b, Heat map of the 272 overlapped genes shown in a. c, GO enrichment analysis for the overlapped genes shown in a. Categories that were significantly enriched for these overlapped genes were selected and shown. Significance was determined using Student’s t test (two-sided). d, Venn diagrams showing the overlapped genes that were down-regulated in fer-4 mutant compared with wild type, but up-regulated in phyB-9 fer-4 compared with fer-4 mutant. e, Heat map of the 135 overlapped genes shown in d. f, GO enrichment analysis for the overlapped genes shown in d. Categories that were significantly enriched for these overlapped genes were selected and shown. Significance was determined using Student’s t test (two-sided).
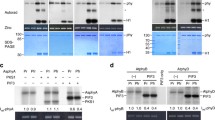
Extended Data Fig. 5 Identification of the phosphorylation sites of phyB by FER.
a, Co-localization analysis of FER and phyB in tobacco leaves under dark and white light conditions. Scale bar, 10 μm. b, Split luciferase complementation assay showing the interaction of THE1 with phyB. Fluorescence was detected at 48 h after infiltration of the indicated constructs. c, Mass spectrometry analysis of the phosphorylation of phyBN and phyBNAA after incubation with FERCD. In the left panel, the two peaks represent nonphosphorylated phyBN and the biotin-tagged nonphosphorylated phyBN. In the middle and right panels, the newly developed peaks represent phosphorylated phyBN. d, The phosphorylation sites of phyB after incubation with FERCD were identified by LC–MS analysis. PH indicates the phosphorylated residues. e, In vitro kinase assay showing the phosphorylation of phyBN and phyBNS86A by FERCD. It should be noted that, due to the addition of a small amount of FERCD in the reaction buffer, FERCD band was not clearly detected in CBB. The experiments in a and e were repeated independently for at least twice with similar results.
Extended Data Fig. 6 Identification of the phosphorylation sites of phyB by FER in Arabidopsis.
a,b, In vivo phosphorylations of S106 (a) and S227 (b) residues in phyB were identified by using LC–MS assay. PH indicates the phosphorylated residues. c,d, Relative phosphopeptide signals of the peptides containing phosphorylated S106 (c) or phosphorylated S227S234 (d) residues in wild type and fer-4 seedlings based on LC–MS data. e, Analysis of the phosphorylation of phyB in wild type and fer-4 seedlings grown on 1/2 MS medium with or without NaCl (120 mM). Total proteins were extracted from seven-day-old etiolated seedlings. phyB-GFP protein was immunoprecipitated with anti-GFP magarose beads, and immunoblotting assays were performed using anti-phosphoserine and anti-GFP antibodies. The experiment was repeated independently for twice with similar results. f, Relative phosphopeptide signals of the phyB peptides containing phosphorylated S94S106 residues in Arabidopsis seedlings treated with or without NaCl (150 mM). Values in c, d and f are the means ± SD of two biological replicates. Asterisks in c and f indicate statistically significant differences (*P < 0.05, Student’s t test, two-sided).
Extended Data Fig. 7 Phosphorylation of Ser106 and Ser227 is required for the regulation of photobody dissociation under dark conditions.
a, Protein abundance of phyB in wild type (Col-0), 35 S::phyB-GFP/phyB-9 (WT), and 35 S::phyBS106AS227A-GFP/phyB-9 (AA) transgenic plants was analyzed using anti-phyB antibody. b, Hypocotyl phenotype of four-day-old seedlings grown on 1/2 MS medium under white light conditions (16 h light/8 h dark cycle). Scale bar, 2 mm. c, Quantification of the hypocotyl length of the seedlings shown in b. Values are the means ± SD (n = 50-55 seedlings). d, Time-course analysis of the hypocotyl length of seedlings before and after dark treatment (n = 33 seedlings). e, Protein abundance of phyB in each genotype was analyzed by using anti-phyB antibody. DD represents 35 S::phyBS106DS227D-GFP/phyB-9 transgenic plants. f, Photobody dissociation under dark conditions. Five-day-old seedlings were exposed to white light for 5 h (ZT5) after dawn, and then the seedlings were treated in darkness for 1 h and 3 h before observation of photobodies. Scale bar, 4 μm. g, Quantification of the diameter of phyB photobodies shown in f. Values are the means ± SD (n > 100 bodies from 30-40 cells). h, Hypocotyl length of four-day-old seedlings grown on 1/2 MS medium under white light conditions. Values are the means ± SD (n = 54 seedlings). i, Protein abundance of phyB in each genotype was analyzed using anti-phyB antibody. j, Hypocotyl phenotype of four-day-old seedlings grown on 1/2 MS medium under white light conditions. Scale bar, 2 mm. k, Quantification of the hypocotyl length of the seedlings shown in j. Values are the means ± SD (n = 38-53 seedlings). Asterisks in g indicate statistically significant differences (**P < 0.01, Student’s t test, two-sided). Different letters in c, h, and k indicate statistically significant differences (P < 0.01, one-way ANOVA). The experiments in a, e and i were repeated independently for at least three times with similar results.
Extended Data Fig. 8 Phosphorylation of phyB is not required for far-red light-triggered photoconversion.
a, Spectral analysis of the photoswitching of wild type phyBN, phyBNS106AS227A, phyBNS86A, and phyBNS86AS106AS227A after irradiance with far-red light. Proteins were exposed to red light illumination (~17 μmol m-2 sec-1) for 5 min and then exposed to far-red light illumination (~5 μmol m-2 sec-1) for 5 min. Red lines indicate absorbance spectra after red light illumination and blue lines indicate absorbance spectra after far-red light illumination. The absorption spectra of phyBN were measured using Nanodrop 2000C spectrophotometer. b, Quantification of the absorption spectra of the wild type phyBN and point-mutated phyB under red light and far-red light illumination. Y axis represents Pfr/Pr ratio. Values are the means ± SD of three independent replicates. Different letters indicate statistically significant differences (p < 0.01, one-way ANOVA). c, Recombinant His-FERCD and His-FERCDK565R were detected by Coomassie Brilliant Blue (CBB) staining. His-FERCD was treated with or without λPPase. The experiments were repeated independently for three times with similar results.
Extended Data Fig. 9 phyB-PIFs module is required for the regulation of plant survival under salt stress.
a, The survival rate of wild type, 35 S::phyB-GFP/phyB-9 and 35 S::phyBS106AS227A-GFP/phyB-9 (AA) seedlings grown on 1/2 MS medium supplemented with NaCl (120 mM) under white light conditions (16 h light/8 h dark cycle). Values are the means ± SD of three independent replicates. b, RT–qPCR analysis of the transcript level of PIF5 gene in wild type and 35 S::PIF5-YFP transgenic plants. ACTIN8 was used as an internal control. c, RT–qPCR analysis of the transcript level of PIF3 gene in wild type and PIF3OE/fer-4 seedlings. d, Phenotypes of wild type, fer-4, PIF3OE/WT, and PIF3OE/fer-4 seedlings grown on 1/2 MS medium with or without NaCl (120 mM) under white light conditions. e, Quantification of the survival rate of seedlings grown on NaCl medium. Values in a, b, c and e are the means ± SD of three independent replicates. Different letters in a and e indicate statistically significant differences (p < 0.01, one-way ANOVA). Asterisks in c indicate statistically significant differences (**P < 0.01, Student’s t test, two-sided).
Supplementary information
Supplementary Information
Supplementary Tables 2 and 3.
Supplementary Table 1
List of differentially expressed genes.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Unprocessed western blots and gels.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 3
Unprocessed western blots.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 4
Unprocessed western blots and gels.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 5
Unprocessed western blots.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 6
Unprocessed western blots.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 5
Unprocessed western blots and gels.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 6
Unprocessed western blots.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 7
Unprocessed western blots.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 8
Unprocessed gels.
Source Data Extended Data Fig. 9
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Jiang, W., Li, Y. et al. FERONIA coordinates plant growth and salt tolerance via the phosphorylation of phyB. Nat. Plants 9, 645–660 (2023). https://doi.org/10.1038/s41477-023-01390-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-023-01390-4
- Springer Nature Limited