Abstract
Vγ9Vδ2 T cells are promising candidates for cellular tumor immunotherapy. Due to their HLA-independent mode of action, allogeneic Vγ9Vδ2 T cells can be considered for clinical application. To apply allogeneic Vγ9Vδ2 T cells in adoptive immunotherapy, the methodology used to obtain adequate cell numbers with optimal effector function in vitro needs to be optimized, and clinical safety and efficacy also need to be proven. Therefore, we developed a novel formula to improve the expansion of peripheral γδ T cells from healthy donors. Then, we used a humanized mouse model to validate the therapeutic efficacy of expanded γδ T cells in vivo; furthermore, the expanded γδ T cells were adoptively transferred into late-stage liver and lung cancer patients. We found that the expanded cells possessed significantly improved immune effector functions, including proliferation, differentiation, and cancer cell killing, both in vitro and in the humanized mouse model. Furthermore, a phase I clinical trial in 132 late-stage cancer patients with a total of 414 cell infusions unequivocally validated the clinical safety of allogeneic Vγ9Vδ2 T cells. Among these 132 patients, 8 liver cancer patients and 10 lung cancer patients who received ≥5 cell infusions showed greatly prolonged survival, which preliminarily verified the efficacy of allogeneic Vγ9Vδ2 T-cell therapy. Our clinical studies underscore the safety and efficacy of allogeneic Vγ9Vδ2 T-cell immunotherapy, which will inspire further clinical investigations and eventually benefit cancer patients.
Similar content being viewed by others
Introduction
Cancer is one of the leading lethal diseases worldwide because of its high incidence and mortality. In 2018, 18.1 million new cancer cases and 9.6 million cancer deaths were estimated to occur worldwide.1 Therefore, develo** new antitumor strategies for reducing mortality rates and improving patients’ quality of life are urgently needed. In this respect, the introduction of checkpoint inhibitors to unleash the activity of tumor-reactive T cells has been a milestone in cancer immunotherapy.2 Among various newly emerging treatment strategies, adoptive immune cell transfer therapy (ACT) has attracted great attention. ACT potentially includes αβ T-cell (e.g., CD8+ T cells and CAR-T cells),3,4,5 natural killer (NK) cell-6,7,8 and gamma delta (γδ) T-cell-based9,10,11,12,13,14,15 immunotherapy.
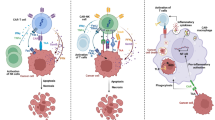
γδ T cells, specifically the Vγ9Vδ2 subset, which is the dominate subset among γδ T cells in human peripheral blood, recognize target cells in a human leukocyte antigen (HLA)-independent manner. Moreover, γδ T cells can directly kill target cells without the involvement of dendritic cells (DCs) and perform dual functional roles in antitumor and anti-infective immunity. Vγ9Vδ2 T cells recognize pyrophosphates secreted by many microbes or overproduced by malignant cells in the context of butyrophilin 3A1 molecules.16,17 Importantly, endogenous production of pyrophosphates can be stimulated by nitrogen-containing aminobisphosphonates, such as zoledronate (ZOL), leading to potent activation of γδ T cells.18 Such advantages enable γδ T cells to rapidly respond against malignant transformation and pathogenic stress.19 For instance, γδ T cells are the earliest producers of IFN-γ in the tumor microenvironment20 and during spinal cord injury.21 γδ T cells utilize a variety of surface receptors and cytokines, such as NKG2D, TRAIL, FASL, TNF-α, IFN-γ, Granzyme B, and perforin, to initiate cytotoxicity against cancer cells.11,12,13,In vivo evaluation of the antitumor activity of Vγ9Vδ2 T cells using humanized mice The establishment of humanized mouse model and related experimental procedures referred to our previously published work in Cancer Cell14,15,23 Prior to 2014, a number of clinical trials were conducted using autologous γδ T cells; however, only limited responses were reported.12,26,27 The main obstacles related to autologous γδ T-cell therapy include the difficulty of expansion, limited cell purity, and impaired cell functions. To obtain adequate cell numbers and optimal effector functions, we developed a NF consisting of ZOL (which selectively activates Vγ9Vδ2 γδ T cells), IL-2, IL-15, and vitamin C (Patent#: PCT/CN2019/075491) to better expand Vγ9Vδ2 T cells from healthy donors in vitro instead of using a gene modification-based methodology. Our results clearly indicated that the NF could promote γδ T-cell proliferation and differentiation, evidenced by significantly increased cell numbers and Ki-67+ proliferating cell numbers, a higher percentage of S-phase cells with a reduced percentage of G1-phase cells, and an elevated percentage of terminally differentiated effector memory (EMRA) cells. Moreover, new formula-expanded γδ T cells (NF cells) had a strikingly lower apoptosis rate than OF-expanded cells. In addition, NF cells expressed significantly higher levels of costimulatory molecules, which implied that the NF-expanded γδ T cells might promote antigen presentation to conventional CD4 and CD8 T cells, and thus correlate with superior antitumor activity. The substantially stronger cellular energy metabolism capability of NF cells was associated with their superior antitumor activity as well. Importantly, NF cells also expressed considerably higher levels of effector molecules (IFN-γ and TNF-α) and exhibited increased degranulation (induction of CD107a expression) associated with enhanced cytotoxicity against various cancer cell lines in vitro. Together, these results demonstrated that NF cells had optimal immune effector functions, as well as stronger in vitro antitumor activity than OF cells. Furthermore, our preclinical in vivo experiments showed that adoptive transfer of NF cells significantly inhibited tumor growth in humanized mice transplanted with human lung tumor cells and prolonged tumor-bearing mouse survival from <3 months to as long as 8 months. It should be mentioned here that the humanized mouse model was established according to our published protocols38,39 and mice that survived GVHD caused by huPBMC pre-engraftment were used for experiments in our work. Notably, we observed that only NF cells visually colocalized with inoculated tumors, implying NF cells had a better ability to migrate to the tumor site than did OF cells. Although OF cells prolonged mouse survival as well, the inhibition of tumor growth was not as efficient as that induced by NF cells, as indicated by the significantly prolonged overall survival of NF-cell-treated mice. It should be remarked here that the variability in outcomes among the five mice per group could be partially related to the differences in immune reconstitution among individual mice or the differences in the antitumor activity of infused γδ T cells derived from different donors. Nevertheless, based on the present in vivo work, which was repeated once previously, we can conclude that NF cells have stronger antitumor activities than OF cells. In addition, although the Vγ9Vδ2 T cells used for adoptive transfer were not 100% pure (≥90%), no allogeneic effects were observed in our mouse experiments. Such results provided scientific support for the initiation of our clinical trials. From our clinical trial investigations, we can draw some clinically important conclusions. First, allogeneic Vγ9Vδ2 T cells (NF cells) are clinically safe. This conclusion is strongly supported by our observation of 414 allogeneic NF-cell infusions in 132 cancer patients, which showed no significant adverse effects (e.g., immune rejection, cytokine storm, or GVHD effects). We emphasized here that the majority of patients received only 1–4 rounds of cell therapy and that the data obtained were thus only used to evaluate the clinical safety of allogeneic Vγ9Vδ2 T cells. It should be mentioned here that we measured cytokine release at 24 h post cell transfer therapy as well and found no evidence of cytokine storm after treatment with allogeneic Vγ9Vδ2 T cells. Nevertheless, a better time point for measuring cytokine release syndrome in patients with solid tumors would be ~1 week post cell therapy. Then, according to clinical observations, cell therapy could improve the quality of life in all patients, including pain relief, appetite, and sleep quality. In our clinical study, among 132 cancer patients, only 8 liver and 10 lung cancer patients received ≥5 cell infusions; therefore, the obtained clinical data were used to preliminarily analyze the efficacy of allogeneic Vγ9Vδ2 T-cell therapy. NF-cell infusions (≥5) prolonged survival in seven of the eight liver cancer patients and eight of the ten lung cancer patients to ≥10 months. Quite excitingly, in our latest follow-up in June 2020, we observed that three liver and two lung cancer patients were still alive (corresponding to survival times between 30 and 35 months) with normal lives. This clearly demonstrates that allogeneic Vγ9Vδ2 T-cell therapy prolonged the survival of most late-stage cancer patients who received ≥5 cell infusions in this small cohort (18 patients) investigation, showing the clinical efficacy of this therapy. In addition, follow-up study of 18 patients preliminarily implied that liver cancer patients might have better clinical responses than lung cancer patients. However, such observations need to be verified by further increasing the patient numbers in extended clinical studies. In conclusion, we developed a NF for the in vitro expansion of Vγ9Vδ2 T cells. The NF could significantly enhance cellular immune functions, including proliferation, differentiation, cellular energy metabolism, effector molecule expression, and cytotoxicity against cancer cell lines, while reducing the apoptotic rate in vitro. Moreover, an in vivo experiment using humanized mice firmly validated the superior antitumor cytotoxicity of NF cells compared with OF cells, which significantly prolonged mouse survival from <3 months to as long as 8 months. Most importantly, our clinical trials for the first time provided scientific evidence that allogeneic Vγ9Vδ2 T cells are clinically safe and preliminarily demonstrated therapeutic efficacy in solid tumor patients. Based on this work, we expect more clinical trials using allogeneic Vγ9Vδ2 T cells to treat malignant tumors to be conducted, which will eventually benefit tumor patients.
Data availability
Detailed clinical data sharing is not applicable to this article as clinical and commercial applications are ongoing.
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Wei, S. C., Duffy, C. R. & Allison, J. P. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov. 8, 1069–86 (2018).
Brudno, J. N. & Kochenderfer, J. N. Chimeric antigen receptor T-cell therapies for lymphoma. Nat. Rev. Clin. Oncol. 15, 31–46 (2018).
Mikkilineni, L. & Kochenderfer, J. N. Chimeric antigen receptor T-cell therapies for multiple myeloma. Blood 130, 2594–602 (2017).
Neelapu, S. S. et al. Chimeric antigen receptor T-cell therapy-assessment and management of toxicities. Nat. Rev. Clin. Oncol. 15, 47–62 (2018).
Yuan, Hu,Z. T. & Zhang, Cai Natural killer cell-based immunotherapy for cancer: advances and prospects. Engineering 5, 106–14 (2019).
Cheng, M. et al. Natural killer cell lines in tumor immunotherapy. Front. Med. 6, 56–66 (2012).
Romee, R. et al. Cytokine-induced memory-like natural killer cells exhibit enhanced responses against myeloid leukemia. Sci. Transl. Med. 8, 357ra123 (2016).
Alnaggar, M. et al. Allogenic V9V2 T cell as new potential immunotherapy drug for solid tumor: a case study for cholangiocarcinoma. J. Immunother. Cancer 7, 36 (2019).
Kakimi, K. et al. gammadelta T cell therapy for the treatment of non-small cell lung cancer. Transl. Lung Cancer Res. 3, 23–33 (2014).
Wu, Y. L. et al. gamma delta T cells and their potential for immunotherapy. Int J. Biol. Sci. 10, 119–48 (2014).
Lo Presti, E. et al. Current advances in gammadelta T cell-based tumor immunotherapy. Front. Immunol. 8, 1401 (2017).
Silva-Santos, B., Mensurado, S. & Coffelt, S. B. gammadelta T cells: pleiotropic immune effectors with therapeutic potential in cancer. Nat. Rev. Cancer 19, 392–404 (2019).
Sebestyen, Z. et al. Translating gammadelta (gammadelta) T cells and their receptors into cancer cell therapies. Nat. Rev. Drug Discov. 19, 169–84 (2020).
Vantourout, P. & Hayday, A. Six-of-the-best: unique contributions of gammadelta T cells to immunology. Nat. Rev. Immunol. 13, 88–100 (2013).
Gu, S. et al. Butyrophilin3A proteins and Vgamma9Vdelta2 T cell activation. Semin Cell Dev. Biol. 84, 65–74 (2018).
Yang, Y. et al. A structural change in butyrophilin upon phosphoantigen binding underlies phosphoantigen-mediated Vgamma9Vdelta2 T cell activation. Immunity 50, 1043–53. e5 (2019).
Roelofs, A. J. et al. Peripheral blood monocytes are responsible for gammadelta T cell activation induced by zoledronic acid through accumulation of IPP/DMAPP. Br. J. Haematol. 144, 245–50 (2009).
Wang, P. & Malkovsky, M. Different roles of the CD2 and LFA-1 T-cell co-receptors for regulating cytotoxic, proliferative, and cytokine responses of human V gamma 9/V delta 2 T cells. Mol. Med. 6, 196–207 (2000).
Gao, Y. et al. γδ T cells provide an early source of interferon γ in tumor immunity. J. Exp. Med. 198, 433 (2003).
Sun, G. et al. gammadelta T cells provide the early source of IFN-gamma to aggravate lesions in spinal cord injury. J. Exp. Med. 215, 521–35 (2018).
**ang, Z. & Tu, W. Dual face of Vγ9Vδ2-T cells in tumor immunology: anti- versus pro-tumoral activities. Front. Immunol. 8, 1–13. (2017).
Bonneville, M., O’Brien, R. L. & Born, W. K. Gammadelta T cell effector functions: a blend of innate programming and acquired plasticity. Nat. Rev. Immunol. 10, 467–78 (2010).
Tosolini, M. et al. Assessment of tumor-infiltrating TCRVgamma9Vdelta2 gammadelta lymphocyte abundance by deconvolution of human cancers microarrays. Oncoimmunology 6, e1284723 (2017).
Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 21, 938–45 (2015).
Zhao, Y., Niu, C. & Cui, J. Gamma-delta (gammadelta) T cells: friend or foe in cancer development? J. Transl. Med. 16, 3 (2018).
Hoeres, T. et al. Improving the efficiency of Vgamma9Vdelta2 t-cell immunotherapy in cancer. Front. Immunol. 9, 800 (2018).
Tan, W. K. et al. Expansion of Gamma Delta T Cells-A Short Review on Bisphosphonate and K562-Based Methods. J. Immunologic Sci. 2, 6–12 (2018).
Kouakanou, L. et al. Vitamin C promotes the proliferation and effector functions of human γδ T cells. Cell. Mol. Immunol. https://doi.org/10.1038/s41423-019-0247-8 (2019).
Meraviglia, S. et al. In vivo manipulation of Vgamma9Vdelta2 T cells with zoledronate and low-dose interleukin-2 for immunotherapy of advanced breast cancer patients. Clin. Exp. Immunol. 161, 290–7 (2010).
Kondo, M. et al. Zoledronate facilitates large-scale ex vivo expansion of functional gammadelta T cells from cancer patients for use in adoptive immunotherapy. Cytotherapy 10, 842–56 (2008).
Chargui, J. et al. Bromohydrin pyrophosphate-stimulated Vgamma9delta2 T cells expanded ex vivo from patients with poor-prognosis neuroblastoma lyse autologous primary tumor cells. J. Immunother. 33, 591–8 (2010).
Qaqish, A. et al. Adoptive transfer of phosphoantigen-specific gammadelta T cell subset attenuates mycobacterium tuberculosis infection in nonhuman primates. J. Immunol. 198, 4753–63 (2017).
**ao, L. et al. Large-scale expansion of Vgamma9Vdelta2 T cells with engineered K562 feeder cells in G-Rex vessels and their use as chimeric antigen receptor-modified effector cells. Cytotherapy 20, 420–35 (2018).
Fisher, J. & Anderson, J. Engineering approaches in human gamma delta T cells for cancer immunotherapy. Front. Immunol. 9, 1409 (2018).
Capsomidis, A. et al. Chimeric antigen receptor-engineered human gamma delta T cells: enhanced cytotoxicity with retention of cross presentation. Mol. Ther. 26, 354–65 (2018).
Peters, C., Kouakanou, L. & Kabelitz, D. A comparative view on vitamin C effects on alphabeta- versus gammadelta T-cell activation and differentiation. J. Leukoc. Biol. 107, 1009–22 (2020).
**ang, Z. et al. Targeted activation of human Vgamma9Vdelta2-T cells controls epstein-barr virus-induced B cell lymphoproliferative disease. Cancer Cell 26, 565–76 (2014).
Tu, W. et al. The aminobisphosphonate pamidronate controls influenza pathogenesis by expanding a gammadelta T cell population in humanized mice. J. Exp. Med. 208, 1511–22 (2011).
Chi, H. Regulation and function of mTOR signalling in T cell fate decisions. Nat. Rev. Immunol. 12, 325–38 (2012).
Cheng, M. et al. NK cell-based immunotherapy for malignant diseases. Cell. Mol. Immunol. 10, 230–52 (2013).
Bollino, D. & Webb, T. J. Chimeric antigen receptor-engineered natural killer and natural killer T cells for cancer immunotherapy. Transl. Res. 187, 32–43 (2017).
Hu, Y., Tian, Z. G. & Zhang, C. Chimeric antigen receptor (CAR)-transduced natural killer cells in tumor immunotherapy. Acta Pharm. Sin. 39, 167–76 (2018).
Mehta, R. S. & Rezvani, K. Chimeric antigen receptor expressing natural killer cells for the immunotherapy of cancer. Front Immunol. 9, 283 (2018).
Harrer, D. C. et al. RNA-transfection of gamma/delta T cells with a chimeric antigen receptor or an alpha/beta T-cell receptor: a safer alternative to genetically engineered alpha/beta T cells for the immunotherapy of melanoma. BMC Cancer 17, 551 (2017).
Nakajima, J. et al. A phase I study of adoptive immunotherapy for recurrent non-small-cell lung cancer patients with autologous gammadelta T cells. Eur. J. Cardiothorac. Surg. 37, 1191–7 (2010).
Qian, L. et al. Dual effects of cellular immunotherapy in inhibition of virus replication and prolongation of survival in HCV-positive hepatocellular carcinoma patients. J. Immunol. Res. 2016, 6837241 (2016).
Kobayashi, H. et al. Complete remission of lung metastasis following adoptive immunotherapy using activated autologous gammadelta T-cells in a patient with renal cell carcinoma. Anticancer Res. 30, 575–9 (2010).
Bennouna, J. et al. Phase-I study of Innacell gammadelta, an autologous cell-therapy product highly enriched in gamma9delta2 T lymphocytes, in combination with IL-2, in patients with metastatic renal cell carcinoma. Cancer Immunol. Immunother. 57, 1599–609 (2008).
Kobayashi, H. et al. Phase I/II study of adoptive transfer of gammadelta T cells in combination with zoledronic acid and IL-2 to patients with advanced renal cell carcinoma. Cancer Immunol. Immunother. 60, 1075–84 (2011).
Kobayashi, H. et al. Safety profile and anti-tumor effects of adoptive immunotherapy using gamma-delta T cells against advanced renal cell carcinoma: a pilot study. Cancer Immunol. Immunother. 56, 469–76 (2007).
Abe, Y. et al. Clinical and immunological evaluation of zoledronate-activated Vgamma9gammadelta T-cell-based immunotherapy for patients with multiple myeloma. Exp. Hematol. 37, 956–68 (2009).
Mohme, M., Riethdorf, S. & Pantel, K. Circulating and disseminated tumour cells - mechanisms of immune surveillance and escape. Nat. Rev. Clin. Oncol. 14, 155–67 (2017).
Chang, C. H. et al. Metabolic competition in the tumor microenvironment is a driver of cancer progression. Cell 162, 1229–41 (2015).
Acknowledgements
We thank all patients and investigators involved in the study. This work was supported by the Key Program of the National Natural Science Foundation of China (31830021); Major International Joint Research Program of China (31420103901); “111 project” (B16021); Scientific and Technological Plan of Guangdong Province (201704KW010) (Z.Y.); Fundamental Research Funds for the Central Universities, Natural Science Foundation of Guangdong Province, China (2020A1515010132) (Y.W.); and General Research Fund, Research Grants Council of Hong Kong (17122519, 17121214, 17115015, and 17126317) (W.T.), Hong Kong SAR, China. This work was also partially supported by the National Natural Science Foundation of China (31570898); the Natural Science Foundation of Guangdong Province, China (2016A030313112) (Z.X.); grant Ka 502/19–1 from the German Research Council (Deutsche Forschungsgemeinschaft); and the Cluster of Excellence ExC 306 “Inflammation-at-Interfaces” (Deutsche Forschungsgemeinschaft) (D.K.). Y.H. was supported by the China Postdoctoral Science Foundation (2017M622898). Y.X. was supported by the Postdoctoral Fund of the First Affiliated Hospital of **an University (809008). L.K. was supported by a long-term fellowship from the German Academic Exchange Service (DAAD). C.P. is the recipient of a grant from the Erich und Gertrud Roggenbruck Foundation.
Author information
Authors and Affiliations
Contributions
Protocol design: Z.Y., Y.W., Y.X., Z.X., M.A., W.T., and D.K. In vitro experiments: Y.X., J.H., L.K., Y.H., L.L., C.P., J.W.L., and Y.C. Animal experiments: J.Y., Z.X., Y.X., J.H., and W.T. Clinical therapy for patients: M.A., K.X., and J.C. Immune function testing and statistical analysis: Y.X., J.X.L., J.H., Y.W., Q.W., M.L., J.W.L., J.L.H., and Y.C. Cell culture and quality control for treatment: Y.L., X.W., Y.X., M.A., J.H., M.L., Q.W., and Y.W. Patient recruitment: M.A., K.X., Y.X., Y.W., Z.Y., J.C., and M.L. Patient follow-up: Y.X., M.A., Y.W., Z.Y., M.L., and Q.W. Manuscript writing: Y.W., Z.Y., Y.X., Y.H., and D.K. Manuscript revision: Y.W., Z.Y., Y.H., D.K., W.T., Y.X., and Z.X. All authors contributed to confirmation of the clinical protocol, data analyses, results and discussion and to manuscript proof-reading, and all authors approved the final version.
Corresponding authors
Ethics declarations
Competing interests
A PCT patent (PCT/CN2019/075491) for the new formula for γδ T-cell expansion was filed. D.K. is a member of the Scientific Advisory Boards of Incysus, Imcheck, and Lava Therapeutics.
Consent for publication
All authors who participated in this research agreed to publish this work.
Ethics approval and consent to participate
The study protocol received ethical approval from the Regional Ethics Committee of Guangzhou Fuda Cancer Hospital, China. Written informed consent was obtained from the participants in accordance with the Declaration of Helsinki and ClinicalTrials.gov (NCT03183232, NCT03183219, NCT03183206, and NCT03180437). All animal studies were approved and performed in compliance with the guidelines for the use of experimental animals by the Committee on the Use of Live Animals in Teaching and Research, University of Hong Kong.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xu, Y., **ang, Z., Alnaggar, M. et al. Allogeneic Vγ9Vδ2 T-cell immunotherapy exhibits promising clinical safety and prolongs the survival of patients with late-stage lung or liver cancer. Cell Mol Immunol 18, 427–439 (2021). https://doi.org/10.1038/s41423-020-0515-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-020-0515-7
- Springer Nature Limited