Abstract
Targeted inhibition of cyclin-dependent kinase 7 (CDK7) via its covalent inhibitor THZ1 can suppress the growth of various cancers, while its roles on colorectal cancer (CRC) remain obscure. Here we report that the expression of CDK7 is upregulated in CRC cells and tissues. THZ1 exhibits high potency and selectivity against CRC cells both in vitro and in vivo via induction of cell apoptosis rather than cell cycle disruption. Intriguingly, THZ1 treatment increases the ability of epithelial mesenchymal transition (EMT) and in vivo metastasis to liver of CRC cells. Mechanistical studies reveal that THZ1 increases the expression of Snail, while not other EMT-transcription factors, via enhancing its protein stability rather than mRNA expression or translation. By screening Snail stability related factors via qRT-PCR, results indicate THZ1 and si-CDK7 decrease the expression of protein kinase D1 (PKD1) in CRC cells. Down regulation of PKD1 mediates THZ1 up regulated Snail via dephosphorylation of Snail Ser 11 and prevention of proteasome mediated degradation. Clinical analysis confirms that CDK7 is significantly (p < 0.05) negatively correlated with the expression of mesenchymal markers including FN1, VIM, and MMP2. CRC patients whose tumors expressing less CDK7/SNAI1 or PKD1/SNAI1 showed significant (p < 0.05) poorer overall survival (OS) rate as compared with those with greater levels. Collectively, our data suggest that targeted inhibition of CDK7 can trigger the metastasis of CRC during cancer development via PKD1/Snail axis, which imposes great challenge that inhibition of CDK7 is a potential approach for cancer treatment.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is the second most common cause of cancer death in the world, which responsible for ∼655,000 deaths globally each year [1]. As the major metastasis organ, synchronous liver metastases exist in up to 20% of newly diagnosed CRC patients [2], and only 13% of 5-year survival rate of CRC patients has been reported [3]. Luckily, due to numerous studies revealing alterations in oncogenes and tumor suppressor genes contributed to the tumorigenesis and development of CRC, the clinical treatment and drug targeted therapies of CRC have been greatly improved over the last decades [4]. However, the detailed mechanisms underlying CRC progression and metastasis are still far from being completely elucidated.
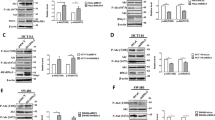
Cyclin-dependent kinases (CDKs), the critical regulatory enzymes controlling all cell cycle transitions, have been suggested as the potent therapeutic targets for cancers [5]. Among the identified CDKs, CDK7 has a general role in the phosphorylation of the RNA polymerase II carboxyterminal domain and therefore regulates the initiation and elongation of transcription [6, 7]. Although the detailed mechanisms remain poorly understood, inhibition of the CDK7 can regulate Pol II promoter occupancy [8, 9], promoter-proximal pausing [10,11,12] and mRNA stability [13]. Targeted inhibition of CDK7 via a newly developed covalent inhibitor THZ1 can negatively impact gene expression, suppress the proliferation of cancer cells, and inhibit progression of animal xenograft models [10, 12, 43]. A recent study showed that MYC mediates phosphorylation of S5 on the carboxy-terminal domain of RNA Pol II through recruitment of CDK7, which leads to the activation of targeted transcripts [44]. Whether MYC or other transcription factors are involved in CDK7 regulated transcription of PKD1 via similar approach needs further investigation.
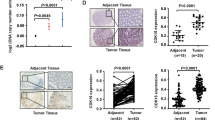
In vivo study and clinical analysis confirmed the suppression effects of CDK7 on the metastasis of CRC. We revealed that expression of CDK7 was elevated in CRC tissue as compared with the adjacent normal tissues. This was consistent with recent reports that CDK7 was increased in cancer tissues [45, 46]. We observed a strong negative correlation of CDK7 and mesenchymal markers including VIM, FN1, N-Cad, MMP2 and MMP9 expression in CRC patients. The expression of CDK7 was negatively correlated with the in vivo metastasis of CRC patients. Although there was no significant difference for the overall survival rate of patients with CDK7 expression level, CRC patients with less CDK7/SNAI1 or PKD1/SNAI1 showed poorer survival rate as compared with those with greater levels. In estrogen receptor positive (ER+) breast cancer, high-level expression of CDK7 was associated with longer survival in univariate and multivariate analyses [45]. However, patients with high expression of CDK7 exhibited worse overall survival as compared to those with low CDK7 in hepatocellular carcinoma (HCC) [47], gastric [48] and triple negative breast cancer [26]. For an unstratified cohort of 2656 breast cancer patients, no significant association was observed between CDK7 expression and recurrence-free survival [26]. It suggested that the roles of CDK7 on cancer progression might be cancer type specific.
Collectively, our data suggested that CDK7 contributes to the growth of CRC during tumorigenesis. However, CDK7 can suppress the metastasis during cancer development via a PKD1/Snail-dependent manner. The promotive effects on cancer metastasis of THZ1 in the present study imposed great challenge that targeted inhibition of CDK7 is a potential approach for cancer treatment, at least for CRC patients.
Availability of supplementary data
Detailed methods and materials, Figure S1 to S5, supplementary table S1 to S2 are attached.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA. 2016;66:7–30.
Kanas GP, Taylor A, Primrose JN, Langeberg WJ, Kelsh MA, Mowat FS, et al. Survival after liver resection in metastatic colorectal cancer: review and meta-analysis of prognostic factors. Clin Epidemiol. 2012;4:283–301.
Zullig LL, Smith VA, Jackson GL, Danus S, Schnell M, Lindquist J, et al. Colorectal cancer statistics from the veterans affairs central cancer registry. Clin Colorectal Cancer. 2016;15:e199–204.
Restifo NP, Smyth MJ, Snyder A. Acquired resistance to immunotherapy and future challenges. Nat Rev Cancer. 2016;16:121–6.
Asghar U, Witkiewicz AK, Turner NC, Knudsen ES. The history and future of targeting cyclin-dependent kinases in cancer therapy. Nat Rev Drug Discov. 2015;14:130–46.
Spangler L, Wang X, Conaway JW, Conaway RC, Dvir A. TFIIH action in transcription initiation and promoter escape requires distinct regions of downstream promoter DNA. Proc Natl Acad Sci USA. 2001;98:5544–9.
Akoulitchev S, Makela TP, Weinberg RA, Reinberg D. Requirement for TFIIH kinase activity in transcription by RNA polymerase II. Nature. 1995;377:557–60.
Bataille AR, Jeronimo C, Jacques PE, Laramee L, Fortin ME, Forest A, et al. A universal RNA polymerase II CTD cycle is orchestrated by complex interplays between kinase, phosphatase, and isomerase enzymes along genes. Mol Cell. 2012;45:158–70.
Wong KH, ** Y, Struhl K. TFIIH phosphorylation of the Pol II CTD stimulates mediator dissociation from the preinitiation complex and promoter escape. Mol Cell. 2014;54:601–12.
Kwiatkowski N, Zhang T, Rahl PB, Abraham BJ, Reddy J, Ficarro SB, et al. Targeting transcription regulation in cancer with a covalent CDK7 inhibitor. Nature. 2014;511:616–20.
Larochelle S, Amat R, Glover-Cutter K, Sanso M, Zhang C, Allen JJ, et al. Cyclin-dependent kinase control of the initiation-to-elongation switch of RNA polymerase II. Nat Struct Mol Biol. 2012;19:1108–15.
Chipumuro E, Marco E, Christensen CL, Kwiatkowski N, Zhang T, Hatheway CM, et al. CDK7 inhibition suppresses super-enhancer-linked oncogenic transcription in MYCN-driven cancer. Cell. 2014;159:1126–39.
Hong SW, Hong SM, Yoo JW, Lee YC, Kim S, Lis JT, et al. Phosphorylation of the RNA polymerase II C-terminal domain by TFIIH kinase is not essential for transcription of Saccharomyces cerevisiae genome. Proc Natl Acad Sci USA. 2009;106:14276–80.
Wang Y, Zhang T, Kwiatkowski N, Abraham BJ, Lee TI, **e S, et al. CDK7-dependent transcriptional addiction in triple-negative breast cancer. Cell. 2015;163:174–86.
Christensen CL, Kwiatkowski N, Abraham BJ, Carretero J, Al-Shahrour F, Zhang T, et al. Targeting transcriptional addictions in small cell lung cancer with a covalent CDK7 inhibitor. Cancer Cell. 2014;26:909–22.
Larochelle S, Merrick KA, Terret ME, Wohlbold L, Barboza NM, Zhang C, et al. Requirements for Cdk7 in the assembly of Cdk1/cyclin B and activation of Cdk2 revealed by chemical genetics in human cells. Mol Cell. 2007;25:839–50.
Liang YC, Tsai SH, Chen L, Lin-Shiau SY, Lin JK. Resveratrol-induced G2 arrest through the inhibition of CDK7 and p34CDC2 kinases in colon carcinoma HT29 cells. Biochem Pharmacol. 2003;65:1053–60.
Thiery JP, Acloque H, Huang RYJ, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139:871–90.
Davis FM, Stewart TA, Thompson EW, Monteith GR. Targeting EMT in cancer: opportunities for pharmacological intervention. Trends Pharmacol Sci. 2014;35:479–88.
Zhou BP, Deng J, **a W, Xu J, Li YM, Gunduz M, et al. Dual regulation of Snail by GSK-3beta-mediated phosphorylation in control of epithelial-mesenchymal transition. Nat Cell Biol. 2004;6:931–40.
Wu Y, Deng J, Rychahou PG, Qiu S, Evers BM, Zhou BP. Stabilization of snail by NF-kappaB is required for inflammation-induced cell migration and invasion. Cancer Cell. 2009;15:416–28.
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15:178–96.
Zheng H, Shen M, Zha YL, Li W, Wei Y, Blanco MA, et al. PKD1 phosphorylation-dependent degradation of SNAIL by SCF-FBXO11 regulates epithelial-mesenchymal transition and metastasis. Cancer Cell. 2014;26:358–73.
Loven J, Hoke HA, Lin CY, Lau A, Orlando DA, Vakoc CR, et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell. 2013;153:320–34.
Jiang YY, Lin DC, Mayakonda A, Hazawa M, Ding LW, Chien WW, et al. Targeting super-enhancer-associated oncogenes in oesophageal squamous cell carcinoma. Gut. 2017;66:1358–68.
Li B, Ni Chonghaile T, Fan Y, Madden SF, Klinger R, O’Connor AE, et al. Therapeutic rationale to target highly expressed CDK7 conferring poor outcomes in triple-negative breast cancer. Cancer Res. 2017;77:3834–45.
Francavilla C, Lupia M, Tsafou K, Villa A, Kowalczyk K, Rakownikow Jersie-Christensen R, et al. Phosphoproteomics of primary cells reveals druggable kinase signatures in ovarian cancer. Cell Rep. 2017;18:3242–56.
Cayrol F, Praditsuktavorn P, Fernando TM, Kwiatkowski N, Marullo R, Calvo-Vidal MN, et al. THZ1 targeting CDK7 suppresses STAT transcriptional activity and sensitizes T-cell lymphomas to BCL2 inhibitors. Nat Commun. 2017;8:14290.
Wang Y, Liu F, Mao F, Hang Q, Huang X, He S, et al. Interaction with cyclin H/cyclin-dependent kinase 7 (CCNH/CDK7) stabilizes C-terminal binding protein 2 (CtBP2) and promotes cancer cell migration. J Biol Chem. 2013;288:9028–34.
Zhang J, Zhu J, Yang L, Guan C, Ni R, Wang Y, et al. Interaction with CCNH/CDK7 facilitates CtBP2 promoting esophageal squamous cell carcinoma (ESCC) metastasis via upregulating epithelial-mesenchymal transition (EMT) progression. Tumour Biol. 2015;36:6701–14.
Roesley SN, Suryadinata R, Morrish E, Tan AR, Issa SM, Oakhill JS, et al. Cyclin-dependent kinase-mediated phosphorylation of breast cancer metastasis suppressor 1 (BRMS1) affects cell migration. Cell Cycle. 2016;15:137–51.
Cao T, **ao T, Huang G, Xu Y, Zhu JJ, Wang K, et al. CDK3, target of miR-4469, suppresses breast cancer metastasis via inhibiting Wnt/beta-catenin pathway. Oncotarget. 2017;8:84917–27.
Zheng D, Cho YY, Lau AT, Zhang J, Ma WY, Bode AM, et al. Cyclin-dependent kinase 3-mediated activating transcription factor 1 phosphorylation enhances cell transformation. Cancer Res. 2008;68:7650–60.
**ao T, Zhu JJ, Huang S, Peng C, He S, Du J, et al. Phosphorylation of NFAT3 by CDK3 induces cell transformation and promotes tumor growth in skin cancer. Oncogene. 2017;36:2835–45.
Liu F, Korc M. Cdk4/6 inhibition induces epithelial-mesenchymal transition and enhances invasiveness in pancreatic cancer cells. Mol Cancer Ther. 2012;11:2138–48.
Arsic N, Bendris N, Peter M, Begon-Pescia C, Rebouissou C, Gadea G, et al. A novel function for Cyclin A2: control of cell invasion via RhoA signaling. J Cell Biol. 2012;196:147–62.
Lehn S, Tobin NP, Berglund P, Nilsson K, Sims AH, Jirstrom K, et al. Down-regulation of the oncogene cyclin D1 increases migratory capacity in breast cancer and is linked to unfavorable prognostic features. Am J Pathol. 2010;177:2886–97.
Bhandari D, Lopez-Sanchez I, To A, Lo IC, Aznar N, Leyme A, et al. Cyclin-dependent kinase 5 activates guanine nucleotide exchange factor GIV/Girdin to orchestrate migration-proliferation dichotomy. Proc Natl Acad Sci USA. 2015;112:E4874–83.
Batlle E, Sancho E, Franci C, Dominguez D, Monfar M, Baulida J, et al. The transcription factor Snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat Cell Biol. 2000;2:84–89.
Cano A, Perez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG, et al. The transcription factor Snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol. 2000;2:76–83.
Du C, Zhang CY, Hassan S, Biswas MHU, Balaji KC. Protein kinase D1 suppresses epithelial-to-mesenchymal transition through phosphorylation of snail. Cancer Res. 2010;70:7810–9.
Nickkholgh B, Sittadjody S, Rothberg MB, Fang XL, Li KZ, Chou JW, et al. Beta-catenin represses protein kinase D1 gene expression by non-canonical pathway through MYC/MAX transcription complex in prostate cancer. Oncotarget. 2017;8:78811–24.
Nilson KA, Guo JN, Turek ME, Brogie JE, Delaney E, Luse DS, et al. THZ1 reveals roles for Cdk7 in co-transcriptional cap** and pausing. Mol Cell. 2015;59:576–87.
Posternak V, Ung MH, Cheng C, Cole MD. MYC Mediates mRNA cap methylation of canonical Wnt/beta-catenin signaling transcripts by recruiting CDK7 and RNA methyltransferase. Mol Cancer Res. 2017;15:213–24.
Patel H, Abduljabbar R, Lai CF, Periyasamy M, Harrod A, Gemma C, et al. Expression of CDK7, cyclin H, and MAT1 is elevated in breast cancer and is prognostic in estrogen receptor-positive breast cancer. Clin Cancer Res. 2016;22:5929–38.
Bartkova J, Zemanova M, Bartek J. Expression of CDK7/CAK in normal and tumor cells of diverse histogenesis, cell-cycle position and differentiation. Int J Cancer. 1996;66:732–7.
Wang C, ** H, Gao D, Wang L, Evers B, Xue Z, et al. A CRISPR screen identifies CDK7 as a therapeutic target in hepatocellular carcinoma. Cell Res. 2018;28:690.
Wang Q, Li M, Zhang X, Huang H, Huang J, Ke J, et al. Upregulation of CDK7 in gastric cancer cell promotes tumor cell proliferation and predicts poor prognosis. Exp Mol Pathol. 2016;100:514–21.
Acknowledgements
This research was supported by the National Natural Science Foundation of China (Grant No. 81673454, No. 81672608, No. 81472470, No. 31801197, and No. 81572270), the Guangdong Natural Science Funds for Distinguished Young Scholar (No. 2014A030306025), the Hunan Young Talent, China (No.2017RS3051), the Pearl River S&T Nova Program of Guangzhou (No. 1517000390), and the Fundamental Research Funds for the Central Universities (Sun Yat-sen University) (16ykpy09).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Edited by G. Del Sal
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhou, Y., Lu, L., Jiang, G. et al. Targeting CDK7 increases the stability of Snail to promote the dissemination of colorectal cancer. Cell Death Differ 26, 1442–1452 (2019). https://doi.org/10.1038/s41418-018-0222-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41418-018-0222-4
- Springer Nature Limited
This article is cited by
-
CDK7 in breast cancer: mechanisms of action and therapeutic potential
Cell Communication and Signaling (2024)
-
Transcriptional inhibition of miR-486-3p by BCL6 upregulates Snail and induces epithelial–mesenchymal transition during radiation-induced pulmonary fibrosis
Respiratory Research (2022)
-
5′-tRF-GlyGCC: a tRNA-derived small RNA as a novel biomarker for colorectal cancer diagnosis
Genome Medicine (2021)
-
Transcriptional CDK inhibitors, CYC065 and THZ1 promote Bim-dependent apoptosis in primary and recurrent GBM through cell cycle arrest and Mcl-1 downregulation
Cell Death & Disease (2021)
-
HDAC8 promotes the dissemination of breast cancer cells via AKT/GSK-3β/Snail signals
Oncogene (2020)