Abstract
Little is known about the significance of asymptomatic intra-cranial lesions (ICL) identified by brain MRI in coronary artery disease (CAD) patients. Silent cerebral lesions are suggested to be associated with arterial stiffness in healthy subjects. We investigated whether subclinical ICL are associated with arterial stiffness and the prognosis in CAD patients without medical history of cerebrovascular diseases. We recruited CAD patients who required percutaneous coronary intervention (PCI), did not meet exclusion criteria, and agreed with MRI before PCI. Subjects were divided into two groups according to the presence of ICL of cerebral microbleeds or lacunar infarction. Arterial stiffness was evaluated by brachial-ankle pulse wave velocity (baPWV). Clinical outcome was defined as a composite of cardiovascular death, non-fatal myocardial infarction, stroke, unstable angina and heart failure. In total, 149 patients underwent brain MRI. Patients with ICL (n=55) had significantly higher baPWV than those without ICL (1591–2204 vs. 1450–1956 cm per sec; P=0.009). A multivariate analysis showed that male sex (odds ratio (OR), 3.15; 95% confidence interval (CI), 1.38–7.20; P=0.006) and baPWV (OR, 1.001; 95% CI, 1.000–1.002; P=0.023) were predictors of ICL. In total, 12 patients experienced a cardiovascular event. The Kaplan–Meier analysis indicated a significantly higher incidence of cardiovascular events in patients with ICL (log-rank test: P=0.018). Multivariate Cox proportional hazards analyses indicated that ICL finding was a significant predictor of clinical outcome (hazard ratio, 3.41; 95% CI, 1.02–11.5; P=0.047). Patients with subclinical ICL had a higher baPWV and worse prognoses than those without ICL.
Similar content being viewed by others
Introduction
The incidence of subsequent cardiovascular events is high in patients with coronary artery disease (CAD) despite improvements in lifestyle, medication and interventions.1, 2 Atherosclerosis is a systemic disease that often involves more than one vascular site, and patients with CAD with one or more atherosclerotic lesions, so called polyvascular disease, have a poor prognosis.3, 4 In clinical practice, surrogate markers, such as intima-media thickness and ankle-brachial index, are used to predict adverse cardiovascular events. Little is known, however, about the significance of subclinical and silent intra-cranial lesions (ICL) identified on brain MRI, which cardiologists often encounter by chance in treating CAD patients.
In cerebrovascular and cardiovascular disease, there is a long subclinical period in which atherosclerotic lesions accumulate. The markers of subclinical cerebrovascular pathology that are known to increase the risk of ischemic stroke are small vessel diseases, such as lacunar infarctions.5 Recently, punctuate hemorrhagic legions in the brain parenchyma, or so called cerebral microbleeds, have been a focus of attention as another marker of subclinical cerebrovascular pathology,6 and MRI is highly sensitive for detecting these lesions. Previous studies have indicated that cerebral microbleeds presence is associated with an increased risk of stroke, both ischemic and hemorrhagic.7 There are several surrogate markers of arterial stiffness and atherosclerosis including brachial-ankle pulse wave velocity (baPWV),8, 9 and a previous study reported an association between ICL and arterial stiffness assessed by baPWV in healthy subjects,10 but little is known about the significance of subclinical ICL identified by brain MRI in CAD patients.
In the present study, we investigated whether ICL findings (cerebral microbleeds or lacunar infarction) identified on brain MRI are associated with arterial stiffness assessed by baPWV in CAD patients.
Methods
Study population
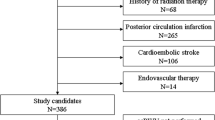
This research is an observational cohort study at Kumamoto University Hospital. We recruited CAD patients with scheduled percutaneous coronary intervention (PCI) from January 2007 to December 2014. The exclusion criteria were as follows: prior history of stroke, prior PCI procedure, past device implantation such as pacemaker, anticoagulant therapy due to diseases like atrial fibrillation, claustrophobia and patients who did not agree with MRI (Figure 1). We also excluded with collagen disease, infection, severe renal or liver dysfunction, and malignant diseases, and patients who died after admission. This study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the Human Ethics Review Committee of the Kumamoto Graduate School of Medicine, and a signed consent form was obtained from each subject. This study was registered at the UMIN protocol registration system with the identification number UMIN000019829.
Flow chart of the present study. From consecutive coronary artery disease patients, we excluded patients with prior stroke, prior PCI procedure, past device implantation, anticoagulant therapy, claustrophobia and patients who did not agree with receiving brain MRI; thus, 149 patients were analyzed and followed up. ICL, intra-cranial lesions; PCI, percutaneous coronary intervention.
Study protocol
T1-weighted, T2-weighted, fluid-attenuated inversion recovery and T2*-weighted sequences were obtained by brain MRI. Cerebral microbleeds were classified as focal, small and round-to-ovoid areas of signal loss on the T2*-weighted images (Figure 2a). Microbleeds are classified based on their location in the brain into groups that best fit their presumed etiology: cerebral amyloid angiopathy-related microbleeds and non-cerebral amyloid angiopathy-related microbleeds (deep gray matter, deep white matter and brainstem with or without lobar microbleeds). In the present study, we identified non-cerebral amyloid angiopathy-related microbleeds. In addition to microbleeds, we assessed the presence of infarcts and defined lacunar infarctions as focal lesions that were between >3 and <15 mm and seen on fluid-attenuated inversion recovery and in T1-weighted and T2-weighted sequences (Figure 2b). We defined asymptomatic cerebral microbleeds and lacunar infarction as intra-cranial lesions (ICL). The participants’ demographics and cardiovascular risk factors were assessed through structured interviews, physical examinations and laboratory examinations, including high-sensitivity C-reactive protein and baPWV. Hypertension was defined as a blood pressure of >140 systolic or >90 diastolic or the use of blood pressure-lowering medication. Dyslipidemia was defined as low-density lipoprotein ⩾140 mg dl−1, high-density lipoprotein <40 mg dl−1 or triglyceride ⩾150 mg dl−1, and diabetes was defined as a 2-h glucose tolerance test finding of at least 200 mg dl−1 or a fasting glucose level of ⩾126 mg dl−1, HbA1c ⩾6.5%, physician-diagnosed diabetes and/or the use of diabetic medication. Smoking status was determined via an interview. The presence of peripheral arterial disease (PAD) was diagnosed when the subjects had current or historical PAD symptoms like intermittent claudication, leg pain at rest and critical limb ischemia associated with a reduced ABI <0.9, or medical histories of amputation for arterial insufficiency, bypass surgery or percutaneous intervention to the extremities. Chronic kidney disease (CKD) was defined as an estimated glomerular filtration rate of <60 ml min−1 1.73 m−2. Acute coronary syndrome was defined as either an acute myocardial infarction (ST-elevation myocardial infarction or non-ST-elevation myocardial infarction) or unstable angina pectoris according to the American College of Cardiology/American Heart Association guidelines.11, 12
Representative images of cerebral microbleeds and lacunar infarction. Cerebral microbleeds on T2*-weighted MRI (a, arrowhead) in a 78-year-old man, and lacunar infarction (b, arrowhead) on T2-fluid-attenuated inversion recovery (FLAIR) MRI in a 62-year-old man. A full color version of this figure is available at the Hypertension Research journal online.
Follow-up
After the brain MRI had been performed, the patients were followed prospectively at outpatient clinics in our hospital until an endpoint occurred. The cardiovascular events were ascertained from a review of medical records and confirmed by direct contact with the patients, their families and physicians. Major adverse cardiovascular events during follow-up time were defined as a composite of cardiovascular death, non-fatal myocardial infarction, stroke, unstable angina and admission because of heart failure. Cardiovascular death was defined as death caused by myocardial infarction, congestive heart failure or documented sudden cardiac death. We used the universal definition of myocardial infarction in this study.13 The diagnosis of ischemic stroke was based on clinical and radiological evidence of stroke. For subjects who had more than 2 cardiovascular events, only the first event was considered in the analysis.
Statistical analysis
The Shapiro–Wilk test was used to assess the normal distribution of the continuous data. Continuous variables with a normal distribution were expressed as the mean±s.d., and skewed distributions were expressed as the median value with the interquartile range. Categorical data were presented as numbers and percentages. Differences between two groups were tested with Fisher's exact test for categorical variables. Differences in continuous variables were analyzed by an unpaired t test or the Mann–Whitney U test as appropriate. The odds ratio (OR) and 95% confidence interval (CI) were examined to assess the association of clinical characteristics with the ICLs using a logistic regression analysis, and Cox proportional hazard models were used to calculate the hazard ratio (HR), and factors with a P-value <0.10 was subsequently entered into multivariate analysis. We used the Kaplan–Meier method to estimate the cardiovascular event probabilities at the follow-up periods, and we used the log-rank test to compare the distributions of survival times. Statistical analyses were performed using SPSS version 22 (IBM Corp., Armonk, NY, USA). A P-value of <0.05 was considered significant.
Results
Characteristics at baseline
The study population consisted of 149 patients (Figure 1), and they were divided into two groups according to the presence of ICL: that without an ICL finding (n=94), and that with an ICL finding (n=55). The mean age of all patients was 70.8±10.0 years, and 100 of the 149 patients (67.1%) were men. The study population had a substantial burden of cardiovascular risk factors: 51.0% of the patients had diabetes, 74.5% had hypertension, 69.8% had dyslipidemia, 12.1% were current tobacco users and 28.9% had a family history of CAD. Among the 149 participants who received brain MRI before PCI, 55 patients (7.1%) had asymptomatic brain lesions of cerebral microbleeds (n=17) and lacunar infarction (n=38). Table 1a presents a comparison of the clinical characteristics of the groups. Patients with ICL were more likely to be male (80.0%; P=0.012) and had significantly higher blood pressure than those without ICL and no significant differences were observed in other clinical characteristics, including the use of statins, beta-blockers, and angiotensin-converting enzyme inhibitor or angiotensin receptor blocker on discharge. Table 1b shows the patient characteristics between subclinical ICL patients with high and low baPWV, and there observed higher age (75.0±9.3 vs. 65.9±10.1; P=0.001) and more prevalence of CKD (73.3 vs. 36.0%; P=0.007) in subclinical ICL patients with high baPWV than those with low baPWV.
Relationship between baPWV and intra-cranial findings
The baPWV among patients with and without ICL were 1898 cm s−1 (1591–2204 cm s−1) and 1756 cm s−1 (1450–1956 cm s−1; P=0.009; Figure 3a). We drew the receiver-operating curve and found a cut-off point of baPWV with ICL as 1892 cm s−1. Patients with ICL had significantly higher hs-CRP than those without ICL (1.6–9.2 mg dl−1 vs. 1.0–5.7 mg dl−1; P=0.025; Figure 3b). We performed a logistic regression analysis for the intra-cranial findings using various clinical characteristics at baseline. Univariate analysis indicated that male sex (OR, 2.71; 95% CI, 1.25–5.91; P=0.012) and baPWV (OR, 1.001; 95% CI, 1.000–1.002; P=0.005) were associated with ICL findings. The focused inclusion multivariate analysis, except for blood pressure in consideration of the internal correlation with baPWV, showed that male sex (OR, 3.15; 95% CI, 1.38–7.20; P=0.006) and baPWV (OR, 1.001; 95% CI, 1.000–1.002; P=0.023) were significant and independent predictors of ICL findings (Table 2a). We drew the receiver-operating curve of the model identified by the multivariate logistic regression analysis; the area under the curve was 0.71 (95% CI, 0.62–0.79; P<0.001).
(a) baPWV between the two groups. Patients with ICL findings identified on brain MRI had significantly higher baPWV than those without ICL (P=0.009). (b) High-sensitivity C-reactive protein (hs-CRP) between the two groups. Patients with ICL had significantly higher hs-CRP than those without ICL (P=0.025). baPWV, brachial-ankle pulse wave velocity; hs-CRP, high-sensitivity C-reactive protein; ICL, intra-cranial lesions.
Clinical outcomes
Data for 149 patients were available for an analysis of subsequent cardiovascular events. We followed patients up to 2,813 days in this study (mean: 707 days; median: 469 days). A total of 12 patients experienced a cardiovascular event, and the details of the cardiovascular events among the group with and without ICL were as follows: cardiovascular death (0 and 0%), non-fatal myocardial infarction (0 and 0%), stroke (1.8 and 0%), unstable angina (7.3 and 1.1%) and admission for heart failure (5.8 and 3.2%), respectively. We performed a Kaplan–Meier analysis among the two groups and observed a significantly higher incidence of cardiovascular events in patients with ICL (log-rank test: P=0.018; Figure 4a). As to the four groups according to the presence of ICL and high or low baPWV, there observed a tendency that patients with both ICL and high baPWV had the worst prognoses, although not significant (Figure 4b; P=0.087).
Kaplan–Meier curve of adverse cardiovascular events between the two groups. (a) Patients with ICL findings on brain MRI had worse prognoses than those without ICL (log-rank test: P=0.018). (b) Among four groups according to the presence of ICL and high or low baPWV, there observed a tendency that patients with both ICL and high baPWV had the worst prognoses, although not significant (log-rank test: P=0.087). baPWV, brachial-ankle pulse wave velocity; ICL, intra-cranial lesions. A full color version of this figure is available at the Hypertension Research journal online.
The results of the univariate and multivariate Cox proportional hazards analyses for cardiovascular events are summarized in Table 2b. The focused inclusion multivariate Cox proportional hazard analysis identified intra-cranial findings to be a significant and independent predictor of future cardiovascular events (HR, 3.41; 95% CI, 1.02–11.5; P=0.047).
Discussion
In the present study, we investigated whether ICL findings (cerebral microbleeds or lacunar infarction) identified on brain MRI are associated with arterial stiffness assessed by baPWV in CAD patients. The main findings of the present study are as follows: (1) There were many patients (55 out of 149 patients) with subclinical and silent ICLs in CAD patients without clinical evidence of cerebrovascular disease such as previous stroke. (2) Patients with ICL had significantly higher baPWV and hs-CRP than those without ICL. (3) Male sex and baPWV were significant and independent predictors of ICL findings. (4) CAD patients with ICL had a poor prognosis compared with those without ICL findings.
Cardiologists often encounter ICL findings when treating CAD patients because cardiovascular diseases often accompany cerebrovascular diseases. ICL findings such as lacunar infarction and cerebral microbleeds are common findings in stroke patients, and silent brain infarction or cerebral microbleeds are reported to be associated with an increased risk of subsequent stroke.5, 7 In healthy subjects, it was reported that assessment of arterial stiffness is useful in identifying subjects at high risk for the presence of ICL. We examined in the present study whether arterial stiffness assessed by baPWV is associated with ICL identified on brain MRI even in CAD patients, and found that CAD patients with ICL had significant higher baPWV than those without ICL. Stiffening is a major pathological manifestation of the aging of the artery, and aortic stiffness might lead to brain damage by inducing changes in the microcirculation.14 Thus, ICL would be the clinical feature with abnormal hyper-acceleration of systemic atherosclerosis determined by baPWV.
Brain MRI scans may be clinically performed to evaluate the baseline findings of intra-cranial atherosclerotic diseases, hemorrhagic lesions and even complications during procedures. In addition, cardiologists may be interested in cerebral microbleeds because dual antiplatelet therapy after PCI would increase the risk of intra-cranial hemorrhages, especially in atrial fibrillation-complicated patients who are taking antithrombotic drugs. However, there is limited evidence as to whether these ICL are potential surrogate markers for clinical outcomes in CAD patients. Extra-cardiac vessel diseases, such as the intima-media thickness of carotid arteries, have been studied to determine whether they have the potential to act as surrogate markers in the prediction of atherosclerosis, CADs and cardiovascular outcomes as well as cerebrovascular outcomes.15, 16, 17, 18 Recently, Polak et al.19 have reported that the maximum internal and mean common carotid-artery intima-media thicknesses both predict cardiovascular outcomes and have indicated that the maximum intima-media thickness of the internal carotid artery significantly improves the risk stratification of cardiovascular diseases. To date, several biomarkers have been studied to improve the predictive ability of cardiovascular diseases; however, studies have not yet been performed to determine whether asymptomatic ICL could be biomarkers of cardiovascular outcomes. Asymptomatic cerebral microbleeds and lacunar infarction, which are measured noninvasively with the T2*-weighted MRI, are independent predictors of subclinical atherosclerosis5, 6 and can predict new or subsequent cerebrovascular events.7 In the present study, we demonstrated that the asymptomatic cerebral ICL significantly predicted clinical outcomes in CAD patients according to two methods: a Kaplan–Meier analysis and multivariate Cox proportional hazards analysis. Thus, ICL could be assessed as a biomarker of adverse cardiovascular events in CAD patients. Moreover, in the present study, patients with both ICL and high baPWV had the worst prognosis, although not significant. Thus, there might be the possibility that evaluating both ICL and baPWV would further improve risk stratification after PCI. It is true that patients requiring PCI are high-risk patients with advanced atherosclerosis, but more and more careful observations might be required especially in patients with ICL findings and high baPWV.
Cerebral microbleeds and lacunar infarctions are associated with ischemic or occlusive brain diseases. Cerebral microbleeds are pathologies that have recently received substantial attention, and cardiologists are often caught in a dilemma with regard to whether dual antiplatelet therapy would be beneficial or hazardous to patients who are undergoing PCI. Evidence has suggested that non-cerebral amyloid angiopathy microbleeds are correlated with an increased risk of cerebral infarction, cerebral hemorrhage20 and acute hemorrhagic events in infarct patients,21 subsequent stroke and intra-cranial hemorrhage during warfarinization.22 However, cerebral microbleeds and lacunar infarctions are suggestive of the long-term exposure of atherosclerotic risk factors,23 and they also reflect systemic micro-vascular diseases; as a result, it makes sense that lacunar infarctions and non-cerebral amyloid angiopathy microbleeds could act as surrogate and predictive markers of cardiovascular diseases. Although the prevalence of MRI is increasing in several advanced countries, especially in Japan, it is not realistic to perform routine brain MRI on CAD patients. In the present study, the multivariate logistic regression analysis suggested that male sex and baPWV are predictive factors of an intra-cranial finding on brain MRI, and it might be possible to perform brain MRI in CAD patients with these factors.
Effect of blood pressure on the risk of stroke is suggested to be important,24 and in controlling blood pressure in patients with stroke, calcium channel blockers and ACE inhibitors or angiotensin II receptor blockers have been proposed as antihypertensive agents,25 and these drugs have been shown to reduce arterial stiffness and reserve cerebral blood flow;26 thus are thought to be suitable for patients with ICL27 and to influence on the development of ICL. However, it is necessary to investigate whether these antihypertensive agents might affect the progression of ICL in prospective studies.
The present study has several limitations. First, this study was performed at a single center and the number of study subjects was small, thus potentially making our findings less convincing. Second, we did not evaluate whether white matter lesions or cerebral lobe microbleeds could be observed in brain MRI, and we investigated only the presence of microbleeds, not the amount or number; thus, we could not evaluate the severity of the microbleeds. Third, in the present study, there were 22.8% of PAD patients and reliability of baPWV is sometimes doubtful in patients with PAD.28 In the present study, there were 32.9% patients who were prescribed aspirin before receiving brain MRI and PCI. Although there was no significant difference in the prescription rate between patients with ICL and without ICL in the present study, treatment with antiplatelet agents might have influenced cerebral microbleeds. Moreover, we did not check and evaluate IMT and carotid artery stenosis. These limitations might influence the results of our study.
In conclusion, patients with asymptomatic and subclinical intra-cranial findings in brain MRI had significantly higher baPWV and had worse prognoses than those without ICL.
References
Boden WE, O'Rourke RA, Teo KK, Hartigan PM, Maron DJ, Kostuk WJ, Knudtson M, Dada M, Casperson P, Harris CL, Chaitman BR, Shaw L, Gosselin G, Nawaz S, Title LM, Gau G, Blaustein AS, Booth DC, Bates ER, Spertus JA, Berman DS, Mancini GB, Weintraub WS . Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med 2007; 356: 1503–1516.
Kimura T, Morimoto T, Furukawa Y, Nakagawa Y, Shizuta S, Ehara N, Taniguchi R, Doi T, Nishiyama K, Ozasa N, Saito N, Hoshino K, Mitsuoka H, Abe M, Toma M, Tamura T, Haruna Y, Imai Y, Teramukai S, Fukushima M, Kita T . Long-term outcomes of coronary-artery bypass graft surgery versus percutaneous coronary intervention for multivessel coronary artery disease in the bare-metal stent era. Circulation 2008; 118: S199–S209.
Bhatt DL, Eagle KA, Ohman EM, Hirsch AT, Goto S, Mahoney EM, Wilson PW, Alberts MJ, D'Agostino R, Liau CS, Mas JL, Rother J, Smith SC Jr., Salette G, Contant CF, Massaro JM, Steg PG . Comparative determinants of 4-year cardiovascular event rates in stable outpatients at risk of or with atherothrombosis. JAMA 2010; 304: 1350–1357.
Steg PG, Bhatt DL, Wilson PW, D'Agostino R Sr., Ohman EM, Rother J, Liau CS, Hirsch AT, Mas JL, Ikeda Y, Pencina MJ, Goto S . One-year cardiovascular event rates in outpatients with atherothrombosis. JAMA 2007; 297: 1197–1206.
Vermeer SE, Longstreth WT Jr, Koudstaal PJ . Silent brain infarcts: a systematic review. Lancet Neurol 2007; 6: 611–619.
Greenberg SM, Vernooij MW, Cordonnier C, Viswanathan A, Al-Shahi Salman R, Warach S, Launer LJ, Van Buchem MA, Breteler MM . Cerebral microbleeds: a guide to detection and interpretation. Lancet Neurol 2009; 8: 165–174.
Akoudad S, Portegies ML, Koudstaal PJ, Hofman A, van der Lugt A, Ikram MA, Vernooij MW . Cerebral microbleeds are associated with an increased risk of stroke: the rotterdam study. Circulation 2015; 132: 509–516.
Schillaci G, Battista F, Settimi L, Anastasio F, Pucci G . Cardio-ankle vascular index and subclinical heart disease. Hypertens Res 2015; 38: 68–73.
Saji N, Kimura K, Yagita Y, Kawarai T, Shimizu H, Kita Y . Comparison of arteriosclerotic indicators in patients with ischemic stroke: ankle-brachial index, brachial-ankle pulse wave velocity and cardio-ankle vascular index. Hypertens Res 2015; 38: 323–328.
Ochi N, Tabara Y, Igase M, Nagai T, Kido T, Miki T, Kohara K . Silent cerebral microbleeds associated with arterial stiffness in an apparently healthy subject. Hypertens Res 2009; 32: 255–260.
Kushner FG, Hand M, Smith SC Jr., King SB 3rd, Anderson JL, Antman EM, Bailey SR, Bates ER, Blankenship JC, Casey DE Jr., Green LA, Hochman JS, Jacobs AK, Krumholz HM, Morrison DA, Ornato JP, Pearle DL, Peterson ED, Sloan MA, Whitlow PL, Williams DO . 2009 focused updates: ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2009; 54: 2205–2241.
Wright RS, Anderson JL, Adams CD, Bridges CR, Casey DE Jr., Ettinger SM, Fesmire FM, Ganiats TG, Jneid H, Lincoff AM, Peterson ED, Philippides GJ, Theroux P, Wenger NK, Zidar JP, Anderson JL, Adams CD, Antman EM, Bridges CR, Califf RM, Casey DE Jr., Chavey WE 2nd, Fesmire FM, Hochman JS, Levin TN, Lincoff AM, Peterson ED, Theroux P, Wenger NK, Wright RS . 2011 ACCF/AHA focused update of the Guidelines for the Management of Patients with Unstable Angina/Non-ST-Elevation Myocardial Infarction (updating the 2007 guideline): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines developed in collaboration with the American College of Emergency Physicians, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol 2011; 57: 1920–1959.
Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD, Katus HA, Lindahl B, Morrow DA, Clemmensen PM, Johanson P, Hod H, Underwood R, Bax JJ, Bonow RO, Pinto F, Gibbons RJ, Fox KA, Atar D, Newby LK, Galvani M, Hamm CW, Uretsky BF, Steg PG, Wijns W, Bassand JP, Menasche P, Ravkilde J, Ohman EM, Antman EM, Wallentin LC, Armstrong PW, Simoons ML, Januzzi JL, Nieminen MS, Gheorghiade M, Filippatos G, Luepker RV, Fortmann SP, Rosamond WD, Levy D, Wood D, Smith SC, Hu D, Lopez-Sendon JL, Robertson RM, Weaver D, Tendera M, Bove AA, Parkhomenko AN, Vasilieva EJ, Mendis S . Third universal definition of myocardial infarction. Circulation 2012; 126: 2020–2035.
O'Rourke MF, Hashimoto J . Mechanical factors in arterial aging: a clinical perspective. J Am Coll Cardiol 2007; 50: 1–13.
O'Leary DH, Polak JF, Kronmal RA, Savage PJ, Borhani NO, Kittner SJ, Tracy R, Gardin JM, Price TR, Furberg CD . Thickening of the carotid wall. A marker for atherosclerosis in the elderly? Cardiovascular Health Study Collaborative Research Group. Stroke 1996; 27: 224–231.
Bots ML, Hoes AW, Koudstaal PJ, Hofman A, Grobbee DE . Common carotid intima-media thickness and risk of stroke and myocardial infarction: the Rotterdam Study. Circulation 1997; 96: 1432–1437.
Chambless LE, Folsom AR, Clegg LX, Sharrett AR, Shahar E, Nieto FJ, Rosamond WD, Evans G . Carotid wall thickness is predictive of incident clinical stroke: the Atherosclerosis Risk in Communities (ARIC) study. Am J Epidemiol 2000; 151: 478–487.
Hodis HN, Mack WJ, LaBree L, Selzer RH, Liu CR, Liu CH, Azen SP . The role of carotid arterial intima-media thickness in predicting clinical coronary events. Ann Intern Med 1998; 128: 262–269.
Polak JF, Pencina MJ, Pencina KM, O'Donnell CJ, Wolf PA, D'Agostino RB Sr . Carotid-wall intima-media thickness and cardiovascular events. N Engl J Med 2011; 365: 213–221.
Bokura H, Saika R, Yamaguchi T, Nagai A, Oguro H, Kobayashi S, Yamaguchi S . Microbleeds are associated with subsequent hemorrhagic and ischemic stroke in healthy elderly individuals. Stroke 2011; 42: 1867–1871.
Fiehler J, Albers GW, Boulanger JM, Derex L, Gass A, Hjort N, Kim JS, Liebeskind DS, Neumann-Haefelin T, Pedraza S, Rother J, Rothwell P, Rovira A, Schellinger PD, Trenkler J . Bleeding risk analysis in stroke imaging before thromboLysis (BRASIL): pooled analysis of T2*-weighted magnetic resonance imaging data from 570 patients. Stroke 2007; 38: 2738–2744.
Lee SH, Ryu WS, Roh JK . Cerebral microbleeds are a risk factor for warfarin-related intracerebral hemorrhage. Neurology 2009; 72: 171–176.
Shimoyama T, Iguchi Y, Kimura K, Mitsumura H, Sengoku R, Kono Y, Morita M, Mochio S . Hypertens Res 2012; 35: 975–979.
Zhong C, Zhong X, Xu T, Peng H, Li H, Zhang M, Wang A, Xu T, Sun Y, Zhang Y . Combined effects of hypertension and heart rate on the risk of stroke and coronary heart disease: a population-based prospective cohort study among Inner Mongolians in China. Hypertens Res 2015; 38: 883–888.
Ogihara T, Kikuchi K, Matsuoka H, Fujita T, Higaki J, Horiuchi M, Imai Y, Imaizumi T, Ito S, Iwao H, Kario K, Kawano Y, Kim-Mitsuyama S, Kimura G, Matsubara H, Matsuura H, Naruse M, Saito I, Shimada K, Shimamoto K, Suzuki H, Takishita S, Tanahashi N, Tsuchihashi T, Uchiyama M, Ueda S, Ueshima H, Umemura S, Ishimitsu T, Rakugi H . The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2009). Hypertens Res 2009; 32: 3–107.
Takami T, Shigemasa M . Efficacy of various antihypertensive agents as evaluated by indices of vascular stiffness in elderly hypertensive patients. Hypertens Res 2003; 26: 609–614.
Kario K, Ishikawa J, Hoshide S, Matsui Y, Morinari M, Eguchi K, Ishikawa S, Shimada K . Diabetic brain damage in hypertension: role of renin-angiotensin system. Hypertension 2005; 45: 887–893.
Yokoyama H, Shoji T, Kimoto E, Shinohara K, Tanaka S, Koyama H, Emoto M, Nishizawa Y . Pulse wave velocity in lower-limb arteries among diabetic patients with peripheral arterial disease. J Atheroscler Thromb 2003; 10: 253–258.
Acknowledgements
We thank Rina Usui and Saki Ogata for collecting and integrating the data. This study was registered in the UMIN protocol registration system (identification number: UMIN000019829).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Tabata, N., Sueta, D., Yamashita, T. et al. Relationship between asymptomatic intra-cranial lesions and brachial-ankle pulse wave velocity in coronary artery disease patients without stroke. Hypertens Res 40, 392–398 (2017). https://doi.org/10.1038/hr.2016.159
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2016.159
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
Pepsinogen ratio and brachial-ankle pulse wave velocity: a cross-sectional study on their interrelationship in atherosclerosis
BMC Cardiovascular Disorders (2023)
-
Brachial-ankle pulse wave velocity and prognosis in patients with atherosclerotic cardiovascular disease: a systematic review and meta-analysis
Hypertension Research (2021)
-
Synergistic effect of hypertension and smoking on the total small vessel disease score in healthy individuals: the Kashima scan study
Hypertension Research (2019)
-
Inter-arm Blood Pressure Difference is Associated with Recurrent Stroke in Non-cardioembolic Stroke Patients
Scientific Reports (2019)
-
Arterial path length estimation for heart-to-brachium pulse wave velocity
Hypertension Research (2018)