Abstract
During pregnancy, myometrial phenotype is programmed into three characteristic stages referred to as the early proliferative, the midterm hypertrophic, and the late contractile stage. Increased myometrial growth in the early and midterm of pregnancy involves a complex process of cell proliferation, antiapoptosis and differentiation. We have previously demonstrated that the androgen receptor (AR) is required for myometrial cell proliferation by modulating IGF-1 signaling during early pregnancy. Here, we report that AR also exerts its antiapoptotic function in human myometrial cells. Enhanced AR expression protects, whereas AR silencing sensitizes myometrial cells to both intrinsic and extrinsic apoptotic stimuli. AR agonist inhibits, whereas AR antagonist induces myometrial cells to undergo apoptotic cell death. Gene microarray analysis confirms that the central functions of AR in myometrial cells are to regulate cell cycling and apoptosis through three major gene groups involving the epidermal growth factor (EGF) signaling, RNA splicing and DNA repair processes. AR mediates its antiapoptotic function through two distinct pathways. In the receptor-dependent pathway, AR is required for the expression of several protein factors within the EGF signaling pathway. Through the PI3K/Akt pathway, AR enhances the expression of the antiapoptotic protein Mcl-1. In the ligand-dependent pathway, AR agonist triggers the activation of Src kinase, which in turn phosphorylates STAT3 to increase Mcl-1 expression. We conclude from these results that the AR signaling exerts antiapoptotic function in myometrial cells, further supporting its key role in programming of myometrial phenotype.
Similar content being viewed by others
Main
The myometrium demonstrates remarkable plasticity during pregnancy, manifest by changes in myometrial phenotype across pregnancy starting from the early proliferative, to the midterm synthetic and the later contractile stages.1, 2 It concludes with postpartum uterine remodeling to complete the reproductive cycle following labor and returns to its nonpregnant receptive state. The proliferative stage of pregnancy involves a complex process of cell proliferation, apoptosis as well as differentiation. At this stage, the myometrial phenotype is characterized by an enhanced proliferation index for example, PCNA and BrdU incorporation.3 In addition, emerging evidence suggests that antiapoptotic mechanisms are also developed to cooperate with myometrial growth. Using a pregnant rat model, we showed high levels of the antiapoptotic proteins, Bcl2 and Bcl2L1, expressed throughout the proliferative stage.3 In addition, although there are several executioner caspases (e.g. caspase 3, 6 and 7) activated during this stage, apoptotic cell death is not observed in myometrial cells.3, 4, 5 As the early proliferative phase of pregnancy is essential to accumulate enough myometrial cell numbers and contractile capacity to engage in parturition, these observations suggest that antiapoptotic mechanisms must be coordinately developed along with cell proliferation during early pregnancy to achieve optimal myometrial growth. Thus, investigation of the antiapoptotic mechanisms of myometrial cells will contribute significantly to our understanding the physiology of uterus and the clinical management of pregnancy and labor progression.
Signaling mediated by estrogen and progesterone receptors had been demonstrated to be important for myometrial growth.6, 7, 8, 9 As transcriptional factors, these steroid receptors regulate expression of several growth factors to modulate myometrial cell growth.10, 11, 12 Growth factors such as insulin-like growth factor (IGF) and epidermal growth factor (EGF) bind their cognate tyrosine kinase receptors and trigger multiple downstream signaling cascades such as PI3K/Akt and Ras-Erk/MAPK.13, 14 Steroid receptors can also exert non-genomic actions to control cell proliferation. Ligand-activated receptors can recruit Src kinase and activate its downstream signaling cascade.15, 16 Therefore, the mutual interplay between steroid receptors and growth factors are vital for myometrial growth.
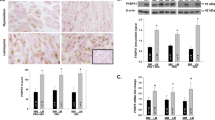
The androgen receptor (AR), a nuclear steroid receptor, is also expressed in the myometrium. Serum androgen levels start to increase progressively during the pregnant luteal phase, initiated by the luteinizing hormone surge.17 AR expression levels in the myometrium are also maintained at higher levels during early proliferative and synthetic phases of pregnancy.Supplementary Table.
UV light and anti-Fas antibody induced apoptosis
cells were serum starved for 12 h before exposured to UV light (UV Stratalinker 1800) for various doses. Cells were allowed to recover for the indicated periods of time. To induce apoptosis through the extrinsic pathway, cells were treated with anti-Fas antibody (clone CH110) at various doses and time points.20 Cells were harvested and the percentage of cells with Annexin V binding was determined by flow cytometry.20 Protein lysates was extracted for western blotting of c-PARP1 and c-CASP3.
Flow cytometry
Annexin V-PE/7-AAD Apoptosis Detection Kit (BD Pharmingen, San Diego, CA, USA) were used to detect apoptotic cell populations according to manufacturer’s protocol. Briefly, the cells (1 × 105 cells) were suspended in 100 μl binding buffer, and then treated with 5 μl of Annexin V-PE (BD Pharmingen) and 5 μl 7-AAD before analyzed on FACSCanto II flow cytometer and BD FACSDiva software v5.0.3 (Becton Dickinson, Franklin, NJ, USA). Ten thousand events are acquired for statistical analysis. Detection of apoptotic population was performed according to the manufacturer’s instructions (https://www.bdbiosciences.com/external_files/pm/doc/tds/cell_bio/live/web_enabled/6900KK_559763.pdf).
Microarray and Real-time PCR
Gene microarray analyses were performed as described before.40 Briefly, Total RNA was extracted from hTERT(shAR) and hTERT(shCTRL) cells by the mirVana RNA Isolation Kit (Ambion, Austin, TX, USA) from three independently repeated experiments. The quality and quantity of RNA were assessed with an Agilent 2100 Bioanalyzer (Caliper Technologies Corp., CA, USA). Amplified and Alexa Fluor 3 labeled RNA samples from hTERT(shCTRL) and hTERT(shAR) cells were hybridized onto the Human Agilent 4 × 44k (Agilent Technologies, Santa Clara, CA, USA), along with Alexa Fluor 5 labeled human reference RNA. Hybridization signals were analyzed following the manufacturer’s instruction. Statistical Analysis of Microarray (SAM) program (http://www-stat.stanford.edu/tibs/SAM/) was used to analyze expression differences between RNA samples from hTERT(shAR) and hTERT(shCTRL) cells. Unpaired t-tests were calculated for all probes passing filters and controlled for multiple testing by estimation of q-values using the false discovery rate method.41 IPA software (Ingenuity Systems) was used to analyze AR-regulated gene groups and signaling pathways.
Real-time PCR assays
Total RNA was extracted using Purelink RNA mini kit (Invitrogen, Burlington, ON, Canada) according to the manufacturer’s instructions. Two micrograms of total RNA was subjected to a random-primed reverse transcription using M-MLV reverse transcriptase (Invitrogen). Real-time qPCR was conducted in triplicates using Applied Biosystems7900HT with 5 ng of cDNA, 1 μM of each primer pair and SYBR Green PCR master mix (Roche, Mississauga, ON, Canada). The sequences of primers were shown below. Relative mRNA levels were normalized to GAPDH.
Statistics
Results are expressed as the mean±S.E.M. To determine differences between two groups, student t-test was carried out using GraphPad Prism (version 4) with the level of significance set at P<0.05 as *, P<0.01 as ** and P<0.001 as ***.
Abbreviations
- AR:
-
androgen receptor
- EGF:
-
epidermal growth factor
- PI3K:
-
Phosphatidylinositol-4,5-bisphosphate 3-kinase
- Akt:
-
protein kinase B
- Mcl-1:
-
induced myeloid leukemia cell differentiation protein
- Src:
-
proto-oncogene tyrosine-protein kinase
- STAT3/5:
-
signal transducer and activator of transcription 3/5
- Bcl2:
-
B-cell lymphoma 2
- Bcl-xL:
-
B-cell lymphoma-extra-large
- PARP1:
-
poly [ADP-ribose] polymerase 1
- CASP3:
-
caspase 3
- FACS:
-
fluorescence-activated cell sorting
- CPT:
-
camptothecin
- SHC:
-
Src homology 2 domain containing transforming protein
- PKC:
-
protein kinase C
- Grb2:
-
growth factor receptor-bound protein 2
- RASA:
-
Ras GTPase-activating protein
References
Lye SJ, Tsui P, Dong X, Mitchell J, Dorogin A, MacPhee D et al. Myometrial programming: a new concept underlying the regulation of myometrial function during pregnancy In: Petraglia F, Strauss III JF, Gabbe SG, Weiss G, (ed) Preterm birth—mechanisms, mediators, prevention and interventions. Informa UK Ltd: London, UK, 2007; pp 1–18.
Shynlova O, Tsui P, Jaffer S, Lye SJ . Integration of endocrine and mechanical signals in the regulation of myometrial functions during pregnancy and labour. Eur J Obstet Gynecol Reprod Biol 2009; 144: S2–10.
Shynlova O, Oldenhof A, Dorogin A, Xu Q, Mu J, Nashman N et al. Myometrial apoptosis: activation of the caspase cascade in the pregnant rat myometrium at midgestation. Biol Reprodn 2006; 74: 839–849.
Jeyasuria P, Subedi K, Suresh A, Condon JC . Elevated levels of uterine anti-apoptotic signaling may activate NFKB and potentially confer resistance to caspase 3-mediated apoptotic cell death during pregnancy in mice. Biol Reprod 2011; 85: 417–424.
Suresh A, Subedi K, Kyathanahalli C, Jeyasuria P, Condon JC . Uterine endoplasmic reticulum stress and its unfolded protein response may regulate caspase 3 activation in the pregnant mouse uterus. PLoS One 2013; 8: e75152.
Ishikawa H, Ishi K, Serna VA, Kakazu R, Bulun SE, Kurita T . Progesterone is essential for maintenance and growth of uterine leiomyoma. Endocrinology 2010; 151: 2433–2442.
Shimomura Y, Matsuo H, Samoto T, Maruo T . Up-regulation by progesterone of proliferating cell nuclear antigen and epidermal growth factor expression in human uterine leiomyoma. J Clin Endocrinol Metab 1998; 83: 2192–2198.
Burroughs KD, Fuchs-Young R, Davis B, Walker CL . Altered hormonal responsiveness of proliferation and apoptosis during myometrial maturation and the development of uterine leiomyomas in the rat. Biol Reprod 2000; 63: 1322–1330.
Yin XJ, Wang G, Khan-Dawood FS . Requirements of phosphatidylinositol-3 kinase and mammalian target of rapamycin for estrogen-induced proliferation in uterine leiomyoma- and myometrium-derived cell lines. Am JObstet Gynecol 2007; 196: 176 e171–175.
Adesanya OO, Zhou J, Bondy CA . Sex steroid regulation of insulin-like growth factor system gene expression and proliferation in primate myometrium. J Clin Endocrinol Metab 1996; 81: 1967–1974.
Rossi MJ, Chegini N, Masterson BJ . Presence of epidermal growth factor, platelet-derived growth factor, and their receptors in human myometrial tissue and smooth muscle cells: their action in smooth muscle cells in vitro. Endocrinology 1992; 130: 1716–1727.
Matsuo H, Kurachi O, Shimomura Y, Samoto T, Maruo T . Molecular bases for the actions of ovarian sex steroids in the regulation of proliferation and apoptosis of human uterine leiomyoma. Oncology 1999; 57: 49–58.
Yu L, Saile K, Swartz CD, He H, Zheng X, Kissling GE et al. Differential expression of receptor tyrosine kinases (RTKs) and IGF-I pathway activation in human uterine leiomyomas. Mol Med 2008; 14: 264–275.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 1997; 91: 231–241.
Migliaccio A, Castoria G, Di Domenico M, de Falco A, Bilancio A, Lombardi M et al. Steroid-induced androgen receptor-oestradiol receptor beta-Src complex triggers prostate cancer cell proliferation. EMBO Jl 2000; 19: 5406–5417.
Migliaccio A, Di Domenico M, Castoria G, de Falco A, Bontempo P, Nola E et al. Tyrosine kinase/p21ras/MAP-kinase pathway activation by estradiol-receptor complex in MCF-7 cells. EMBO J 1996; 15: 1292–1300.
Castracane VD, Stewart DR, Gimpel T, Overstreet JW, Lasley BL . Maternal serum androgens in human pregnancy: early increases within the cycle of conception. Hum Reprod 1998; 13: 460–464.
Liu L, Li Y, **e N, Shynlova O, Challis JR, Slater D et al. Proliferative action of the androgen receptor in human uterine myometrial cells--a key regulator for myometrium phenotype programming. J Clin Endocrinol Metab 2013; 98: 218–227.
Walters KA, Allan CM, Jimenez M, Lim PR, Davey RA, Zajac JD et al. Female mice haploinsufficient for an inactivated androgen receptor (AR) exhibit age-dependent defects that resemble the AR null phenotype of dysfunctional late follicle development, ovulation, and fertility. Endocrinology 2007; 148: 3674–3684.
Hayashi N, Peacock JW, Beraldi E, Zoubeidi A, Gleave ME, Ong CJ . Hsp27 silencing coordinately inhibits proliferation and promotes Fas-induced apoptosis by regulating the PEA-15 molecular switch. Cell Death Differ 2012; 19: 990–1002.
Feldman BJ, Feldman D . The development of androgen-independent prostate cancer. Nat Rev Cancer 2001; 1: 34–45.
Kitagawa D, Tanemura S, Ohata S, Shimizu N, Seo J, Nishitai G et al. Activation of extracellular signal-regulated kinase by ultraviolet is mediated through Src-dependent epidermal growth factor receptor phosphorylation. Its implication in an anti-apoptotic function. J Biol chemistry 2002; 277: 366–371.
Cohen NA, Stewart ML, Gavathiotis E, Tepper JL, Bruekner SR, Koss B et al. A competitive stapled peptide screen identifies a selective small molecule that overcomes MCL-1-dependent leukemia cell survival. Chem Biol 2012; 19: 1175–1186.
Jaffer S, Shynlova O, Lye S . Mammalian target of rapamycin is activated in association with myometrial proliferation during pregnancy. Endocrinology 2009; 150: 4672–4680.
Igney FH, Krammer PH . Death and anti-death: tumour resistance to apoptosis. Nat Rev Cancer 2002; 2: 277–288.
van Delft MF, Wei AH, Mason KD, Vandenberg CJ, Chen L, Czabotar PE et al. The BH3 mimetic ABT-737 targets selective Bcl-2 proteins and efficiently induces apoptosis via Bak/Bax if Mcl-1 is neutralized. Cancer Cell 2006; 10: 389–399.
Scaffidi C, Fulda S, Srinivasan A, Friesen C, Li F, Tomaselli KJ et al. Two CD95 (APO-1/Fas) signaling pathways. EMBO J 1998; 17: 1675–1687.
Jeyasuria P, Wetzel J, Bradley M, Subedi K, Condon JC . Progesterone-regulated caspase 3 action in the mouse may play a role in uterine quiescence during pregnancy through fragmentation of uterine myocyte contractile proteins. Biol Reprod 2009; 80: 928–934.
Liu H, Perlman H, Pagliari LJ, Pope RM . Constitutively activated Akt-1 is vital for the survival of human monocyte-differentiated macrophages. Role of Mcl-1, independent of nuclear factor (NF)-kappaB, Bad, or caspase activation. J Exp Med 2001; 194: 113–126.
Mills JR, Hippo Y, Robert F, Chen SM, Malina A, Lin CJ et al. mTORC1 promotes survival through translational control of Mcl-1. Proc Natl Acad Sci USA 2008; 105: 10853–10858.
Coloff JL, Macintyre AN, Nichols AG, Liu T, Gallo CA, Plas DR et al. Akt-dependent glucose metabolism promotes Mcl-1 synthesis to maintain cell survival and resistance to Bcl-2 inhibition. Cancer Res 2011; 71: 5204–5213.
Maurer U, Charvet C, Wagman AS, Dejardin E, Green DR . Glycogen synthase kinase-3 regulates mitochondrial outer membrane permeabilization and apoptosis by destabilization of MCL-1. Mol Cell 2006; 21: 749–760.
Wang JM, Chao JR, Chen W, Kuo ML, Yen JJ, Yang-Yen HF . The antiapoptotic gene mcl-1 is up-regulated by the phosphatidylinositol 3-kinase/Akt signaling pathway through a transcription factor complex containing CREB. Mol Cell Biol 1999; 19: 6195–6206.
Castoria G, Lombardi M, Barone MV, Bilancio A, Di Domenico M, Bottero D et al. Androgen-stimulated DNA synthesis and cytoskeletal changes in fibroblasts by a nontranscriptional receptor action. J Cell Biol 2003; 161: 547–556.
Epling-Burnette PK, Liu JH, Catlett-Falcone R, Turkson J, Oshiro M, Kothapalli R et al. Inhibition of STAT3 signaling leads to apoptosis of leukemic large granular lymphocytes and decreased Mcl-1 expression. J Clin Invest 2001; 107: 351–362.
Mertens HJ, Heineman MJ, Theunissen PH, de Jong FH, Evers JL . Androgen, estrogen and progesterone receptor expression in the human uterus during the menstrual cycle. Eur J Obstet Gynecol Reprod Biol 2001; 98: 58–65.
Pelletier G, Luu-The V, Li S, Labrie F . Localization and estrogenic regulation of androgen receptor mRNA expression in the mouse uterus and vagina. J Endocrinol 2004; 180: 77–85.
Weihua Z, Ekman J, Almkvist A, Saji S, Wang L, Warner M et al. Involvement of androgen receptor in 17beta-estradiol-induced cell proliferation in rat uterus. Biol Reprod 2002; 67: 616–623.
Sefton EC, Qiang W, Serna V, Kurita T, Wei JJ, Chakravarti D et al. MK-2206, an AKT inhibitor, promotes caspase-independent cell death and inhibits leiomyoma growth. Endocrinology 2013; 154: 4046–4057.
Yu Y, Liu L, **e N, Xue H, Fazli L, Buttyan R et al. Expression and function of the progesterone receptor in human prostate stroma provide novel insights to cell proliferation control. J Clin Endocrinol Metab 2013; 98: 2887–2896.
Larson SR, Zhang X, Dumpit R, Coleman I, Lakely B, Roudier M et al. Characterization of osteoblastic and osteolytic proteins in prostate cancer bone metastases. Prostate 2013; 73: 932–940.
Acknowledgements
This work is supported by Canadian Institutes of Health Research Operating Grant (MOP-111148) to XSD and SL and by a Rising Star Award (RS2013-58) from Prostate Cancer Canada to XSD.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by A Stephanou
Supplementary Information accompanies this paper on Cell Death and Disease website
Rights and permissions
Cell Death and Disease is an open-access journal published by Nature Publishing Group. This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Li, H., Li, Y., Morin, D. et al. The androgen receptor mediates antiapoptotic function in myometrial cells. Cell Death Dis 5, e1338 (2014). https://doi.org/10.1038/cddis.2014.303
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cddis.2014.303
- Springer Nature Limited