Abstract
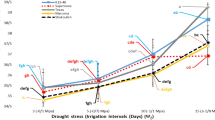
Selecting appropriate scion-rootstock combinations, which are determined by the rootstock’s root system, is vital for successful orchard management. There are usually two types of rootstocks available for pear orchards, clonal as well as seedling rootstocks. This research aimed to examine how seedlings and clonal rootstocks respond and withstand soil moisture limitations and drought stress conditions. Two pear rootstocks including ‘Dargazi’ seedlings and ‘Pyrodwarf’ clones were used. The irrigation treatments were well-watered (100% FC), moderate (60% FC), and severe drought (30% FC). The physiological and gas exchange responses were measured at 30 and 60 days after stress treatment (DAT). Results indicated that stomatal restrictions were one of the main causes of photosynthetic limitations under drought stress in the studied pear rootstocks. Photosynthesis (An) in ‘Pyrodwarf’ was more affected by the leaf ABA concentration than in ‘Dargazi’ rootstock. In 60 DAT, ‘Dargazi’ seedlings improved gas exchange parameters, and root dry weight (RDW). The increase in proline and glycine betaine and decrease in leaf water content per unit leaf area (LWCA) under drought stress showed signs of osmotic adjustment in both studied rootstocks. Although at 30 DAT, the photosynthetic restriction of ‘Pyrodwarf’ was affected by the stomatal closure, in 60 DAT the limitation to photosynthesis is maybe non-stomatal. In conclusion, these findings showed that ‘Pyrodwarf’ adapts better to moderate and shorter duration of drought stress, and ‘Dargazi’ was more adapted to severe and long periods of drought stress. This is due to the direct interaction of the rootstock and scion in resistance to drought stress. Besides it can help to select suitable rootstock for orchards faced with moderate to severe drought conditions.
Graphical Abstract






Similar content being viewed by others
Data Availability
The authors declare that all the data supporting the findings of this research are available within the article.
References
Al-Yasi H, Attia H, Alamer K, Hassan F, Esmat F, Elshazly S, Siddique KH, Hessini K (2020) Impact of drought on growth, photosynthesis, osmotic adjustment, and cell wall elasticity in Damask rose. Plant Physiol Biochem 150:133–139. https://doi.org/10.1016/j.plaphy.2020.02.038
Aliniaeifard S, Asayesh ZM, Driver J, Vahdati K (2020) Stomatal features and desiccation responses of Persian walnut leaf as caused by in vitro stimuli aimed at stomatal closure. Trees 34:1219–1232. https://doi.org/10.1007/s00468-020-01992-x
Aliniaeifard S, Van Meeteren U (2016) Stomatal characteristics and desiccation response of leaves of cut chrysanthemum (Chrysanthemum morifolium) flowers grown at high air humidity. Sci Hortic 205:84–89. https://doi.org/10.1016/j.scienta.2016.04.025
Anjum SA, Ashraf U, Tanveer M, Khan I, Hussain S, Shahzad B, Zohaib A, Abbas F, Saleem MF, Ali I (2017) Drought-induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front Plant Sci 8:69. https://doi.org/10.3389/fpls.2017.00069
Arzani K (1994) Horticultural and physiological aspects of vigor control in apricot (Prunus armeniaca L.) under orchard and controlled environment conditions. Ph.D. Thesis. Department of Plant Science, Massey University. New Zealand. http://hdl.handle.net/10179/4059
Arzani K. (2017) The potential and limiting environmental conditions on fruit trees germplasm and yield of established orchards in Iran. In: Proceedings of the First International Horticultural Science Conference of Iran (IrHC2017). September 4-7, Tarbiat Modares University (TMU), Tehran Iran, p. 110. Abstracts Book. https://www.ishs.org/symposium/643
Arzani K (2021) The national Asian pear (Pyrus serotina Rehd) project in Iran: compatibility and commercial studies of introduced cultivars. Acta Hortic 1315:91–98. https://doi.org/10.17660/ActaHortic1315:14
Asayesh ZM, Arzani K, Mokhtassi-Bidgoli A, Abdollahi H (2023) Enzymatic and non-enzymatic response of grafted and ungrafted young European pear (Pyrus communis L) trees to drought stress. Sci Hortic 310:111745. https://doi.org/10.1016/j.scienta.2022.111745
Askari-Khorsgani O, Flores F, Pessarakli M (2018) Plant signaling pathways involved in stomatal movement under drought stress conditions. Adv Plants Agric Res 8:290–297
Bates L, Waldren R, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Beis A, Patakas A (2010) Differences in stomatal responses and root-to-shoot signaling between two grapevine varieties subjected to drought. Funct Plant Biol 37:139–146. https://doi.org/10.1071/FP09034
Berdeja M, Nicolas P, Kappel C, Dai ZW, Hilbert G, Peccoux A, Lafontaine M, Ollat N, Gomes E, Delrot S (2015) Water limitation and rootstock genotype interact to alter grape berry metabolism through transcriptome reprogramming. Hortic Res 2:1–13. https://doi.org/10.1038/hortres.2015.12
Bhusal N, Han SG, Yoon TM (2019) Impact of drought stress on photosynthetic response, leaf water potential, and stem sap flow in two cultivars of bi-leader apple trees (Malus× domestica Borkh.). Sci Hortic 246:535–543. https://doi.org/10.1016/j.scienta.2018.11.021
Bolat I, Dikilitas M, Ikinci A, Ercisli S, Tonkaz T (2016) Morphological, physiological, biochemical characteristics and bud success responses of myrobolan 29 c plum rootstock subjected to water stress. Can J Plant Sci 96:485–493. https://doi.org/10.1139/cjps-2015-0260
Bolhar-Nordenkampf H, Long S, Baker N, Oquist G, Schreiber U, Lechner E (1989) Chlorophyll fluorescence as a probe of the photosynthetic competence of leaves in the field: a review of current instrumentation. Funct Ecol 3:497–514. https://doi.org/10.2307/2389624
Brodribb T (1996) Dynamics of changing intercellular CO2 concentration (ci) during drought and determination of minimum functional ci. Plant Physiol 111:179–185. https://doi.org/10.1104/pp.111.1.179
Chaves M, Zarrouk O, Francisco R, Costa J, Santos T, Regalado A, Rodrigues M, Lopes C (2010) Grapevine under deficit irrigation: hints from physiological and molecular data. Ann Bot 105:661–676. https://doi.org/10.1093/aob/mcq030
Claeys H, Inze D (2013) The agony of choice: how plants balance growth and survival under water-limiting conditions. Plant Physiol 162:1768–1779. https://doi.org/10.1104/pp.113.220921
Cochard H, Coll L, Le Roux X, Ameglio T (2002) Unraveling the effects of plant hydraulics on stomatal closure during water stress in walnut. Plant Physiol 128:282–290. https://doi.org/10.1104/pp.010400
Costa JM, Ortuno MF, Lopes CM, Chaves MM (2012) Grapevine varieties exhibiting differences in stomatal response to water deficit. Funct Plant Biol 39:179–189. https://doi.org/10.1071/FP11156
Geng DL, Lu LY, Yan MJ, Shen XX, Jiang LJ, Li HY, Wang LP, Yan Y, Xu JD, Li CY (2019) Physiological and transcriptomic analyses of roots from Malus sieversii under drought stress. J Integr Agric 18:1280–1294. https://doi.org/10.1016/S20953119(19)62571-2
Dayer S, Scharwies JD, Ramesh SA, Sullivan W, Doerflinger FC, Pagay V, Tyerman SD (2020) Comparing hydraulics between two grapevine cultivars reveals differences in stomatal regulation under water stress and exogenous ABA applications. Front Plant Sci 11:705. https://doi.org/10.3389/fpls.2020.00705
Dinh TH, Watanabe K, Takaragawa H, Nakabaru M, Kawamitsu Y (2017) Photosynthetic response and nitrogen use efficiency of sugarcane under drought stress conditions with different nitrogen application levels. Plant Prod Sci 20:412–422. https://doi.org/10.1080/1343943X.2017.1371570
Dubey R, Pandey BK, Sawant SV, Shirke PA (2023) Drought stress inhibits stomatal development to improve water use efficiency in cotton. Acta Physiol Plant 45:30. https://doi.org/10.1007/s11738-022-03511-6
Flexas J, Bota J, Loreto F, Cornic G, Sharkey T (2004) Diffusive and metabolic limitations to photosynthesis under drought and salinity in C3 plants. Plant Biol 6:269–279. https://doi.org/10.1055/s-2004-820867
Gomes FP, Oliva MA, Mielke MS, de Almeida AAF, Leite HG, Aquino LA (2008) Photosynthetic limitations in leaves of young Brazilian Green Dwarf coconut (Cocos nucifera L. ‘nana’) palm under well-watered conditions or recovering from drought stress. Environ Exp Bot 62:195–204. https://doi.org/10.1016/j.envexpbot.2007.08.006
Grieve C, Grattan S (1983) Rapid assay for determination of water-soluble quaternary ammonium compounds. Plant Soil 70:303–307. https://doi.org/10.1007/BF02374789
Hao L, Zhang Y, Wang S, Zhang W, Wang S, Xu C, Yu Y, Li T, Jiang F, Li W (2020) A constitutive and drought-responsive mRNA undergoes long-distance transport in pear (Pyrus betulaefolia) phloem. Plant Sci 293:110419. https://doi.org/10.1016/j.plantsci.2020.110419
Huang X, Li K, Xu X, Yao Z, ** C, Zhang S (2015) Genome-wide analysis of WRKY transcription factors in white pear (Pyrus bretschneideri) reveals evolution and patterns under drought stress. BMC Genom 16:1–14. https://doi.org/10.1186/s12864-015-2233-6
Hussain S, Khalid MF, Saqib M, Ahmad S, Zafar W, Rao MJ, Morillon R, Anjum MA (2018) Drought tolerance in citrus rootstocks is associated with a better antioxidant defense mechanism. Acta Physiol Plant 40:135. https://doi.org/10.1007/s11738-018-2710-z
Ikinci A, Bolat I, Ercisli S, Kodad O (2014) Influence of rootstocks on growth, yield, fruit quality and leaf mineral element contents of pear cv’.Santa Maria in semi-arid conditions. Biol Res 47:1–8. https://doi.org/10.1186/0717-6287-47-71
Inglese P, Caruso T, Gugliuzza G, Pace L (2002) Crop load and rootstock influence on dry matter partitioning in trees of early and late ripening peach cultivars. J Am Soc Hortic Sci 127:825–830. https://doi.org/10.21273/JASHS.127.5.825
Jimenez S, Dridi J, Gutierrez D, Moret D, Irigoyen JJ, Moreno MA, Gogorcena Y (2013) Physiological, biochemical and molecular responses in four Prunus rootstocks submitted to drought stress. Tree Physiol 33:1061–1075. https://doi.org/10.1093/treephys/tpt074
Kadkhodaei S, Arzani K, Yadollahi A, Karimzadeh G, Abdollahi H (2021) Genetic diversity and similarity of Asian and European pears (Pyrus spp.) revealed by genome size and morphological traits prediction. Int J Fruit Sci 21(1):619–633. https://doi.org/10.1080/15538362.2021.1908201
Karbasi M, Arzani K (2021) Asian and European pear scion-rootstock interactions and Pyrodwarf rootstock root growth and distribution. Acta Hortic. 1315:63–68. https://doi.org/10.17660/ActaHortic.2021.1315.10
Klumb EK, Rickes LN, Braga EJB, Bianchi VJ (2017) Evaluation of gas exchanges in different Prunus spp. rootstocks under drought and flooding stress. Fitotecnia • Rev Bras Frutic 39 (4) https://doi.org/10.1590/0100-29452017899
Kuecuekyumuk C (2020) Drought response of young pear trees (Pyrus comminus). Appl Ecol Environ Res 18:7769–7781. https://doi.org/10.15666/aeer/1806_77697781
Lampinen B, Shackel K, Southwick S, Olson W, DeJong T (2004) Leaf and canopy level photosynthetic responses of French prune (Prunus domestica L. ‘French’) to stem water potential-based deficit irrigation. J Hortic Sci Biotechnol 79:638–644. https://doi.org/10.1080/14620316.2004.11511819
Li F, Bao W, Wu N (2009) Effects of water stress on growth, dry matter allocation and water-use efficiency of a leguminous species, Sophora davidii. Agrofor Syst 77:193–201. https://doi.org/10.1007/s10457-008-9199-1
Liu B, Li M, Cheng L, Liang D, Zou Y, Ma F (2012) Influence of rootstock on antioxidant system in leaves and roots of young apple trees in response to drought stress. Plant Growth Regul 67:247–256. https://doi.org/10.1007/s10725-012-9683-5
Liu B, Liang J, Tang G, Wang X, Liu F, Zhao D (2019) Drought stress affects on growth, water use efficiency, gas exchange, and chlorophyll fluorescence of Juglans rootstocks. Sci Hortic 250:230–235. https://doi.org/10.1016/j.scienta.2019.02.056
Losciale P, Manfrini L, Morandi B, Novak B, Pierpaoli E, Zibordi M, Corelli Grappadelli L, Antonelli S, Galli F (2012) Water restriction effect on pear rootstocks: photoprotective processes and the possible role of photorespiration in limiting carbon assimilation. Acta Hortic 1058:237–244. https://doi.org/10.17660/ActaHortic.2014.1058.27
Lovisolo C, Perrone I, Carra A, Ferrandino A, Flexas J, Medrano H, Schubert A (2010) Drought-induced changes in development and function of grapevine (Vitis spp.) organs and their hydraulic and non-hydraulic interactions at the whole-plant level: a physiological and molecular update. Funct Plant Biol 37:98–116. https://doi.org/10.1071/FP09191
Ma X, Ma F, Li C, Mi Y, Bai T, Shu H (2010) Biomass accumulation, allocation, and water-use efficiency in 10 Malus rootstocks under two watering regimes. Agrofor Syst 80:283–294. https://doi.org/10.1007/s10457-010-9306-y
Martin-StPaul N, Delzon S, Cochard H (2017) Plant resistance to drought depends on timely stomatal closure. Ecol Lett 20(11):1437–1447 https://doi.org/10.1111/ele.12851
McAdam SA, Brodribb TJ (2015) Hormonal dynamics contribute to divergence in seasonal stomatal behavior in a monsoonal plant community. Plant Cell Environ 38:423–432. https://doi.org/10.1111/pce.12398
McAdam SA, Manzi M, Ross JJ, Brodribb TJ, Gomez-Cadenas A (2016) Uprooting an abscisic acid paradigm: shoots are the primary source. Plant Signal Behav 11:652–659. https://doi.org/10.1080/15592324.2016.1169359
Medrano H, Escalona JM, Bota J, Gulias J, Flexas J (2002) Regulation of photosynthesis of C3 plants in response to progressive drought: stomatal conductance as a reference parameter. Ann Bot 89:895–905. https://doi.org/10.1093/aob/mcf079
Meggio F, Prinsi B, Negri A, Simone Di Lorenzo G, Lucchini G, Pitacco A, Failla O, Scienza A, Cocucci M, Espen L (2014) Biochemical and physiological responses of two grapevine rootstock genotypes to drought and salt treatments. Aust. J. Grape Wine Res 20:310–323. https://doi.org/10.1111/ajgw.12071
Miranda MT, Da Silva SF, Silveira NM, Pereira L, Machado EC, Ribeiro RV (2021) Root osmotic adjustment and stomatal control of leaf gas exchange are dependent on citrus rootstocks under water deficit. J Plant Growth Regul 40:11–19. https://doi.org/10.1007/s00344-020-10069-5
Misson L, Limousin JM, Rodriguez R, Letts MG (2010) Leaf physiological responses to extreme droughts in Mediterranean Quercus ilex forest. Plant Cell Environ 33:1898–1910. https://doi.org/10.1111/j.1365-3040.2010.02193.x
Pandey P, Irulappan V, Bagavathiannan MV, Senthil-Kumar M (2017) Impact of combined abiotic and biotic stresses on plant growth and avenues for crop improvement by exploiting physio-morphological traits. Front Plant Sci 8:537. https://doi.org/10.3389/fpls.2017.00537
Pou A, Medrano H, Flexas J, Tyerman SD (2013) A putative role for TIP and PIP aquaporins in dynamics of leaf hydraulic and stomatal conductances in grapevine under water stress and re-watering. Plant Cell Environ 36:828–843. https://doi.org/10.1111/pce.12019
Qi J, Sun S, Yang L, Li M, Ma F, Zou Y (2019) Potassium uptake and transport in apple roots under drought stress. Hortic Plant J 5:10–16. https://doi.org/10.1016/j.hpj.2018.10.001
Rampino P, Pataleo S, Gerardi C, Mita G, Perrotta C (2006) Drought stress response in wheat: physiological and molecular analysis of resistant and sensitive genotypes. Plant Cell Environ 29:2143–2152. https://doi.org/10.1111/j.1365-3040.2006.01588.x
Richards L (1949) Methods of measuring soil moisture tension. Soil Sci 68:95
Rouhi V, Samson R, Lemeur R, Van Damme P (2007) Photosynthetic gas exchange characteristics in three different almond species during drought stress and subsequent recovery. Environ Exp Bot 59:117–129. https://doi.org/10.1016/j.envexpbot.2005.10.001
Santana-Vieira DDS, Freschi L, da Hora Almeida LA, de Moraes DHS, Neves DM, dos Santos LM, Bertolde FZ, dos Santos SoaresFilho W, Coelho Filho MA, da Silva Gesteira A (2016) Survival strategies of citrus rootstocks subjected to drought. Sci Rep. 6:1–12. https://doi.org/10.1038/srep38775
Singh SK, Reddy KR (2011) Regulation of photosynthesis, fluorescence, stomatal conductance and water-use efficiency of cowpea (Vigna unguiculata [L.] Walp.) under drought. J. Photochem. Photobiol. B, Biol 105:40–50. https://doi.org/10.1016/j.jphotobiol.2011.07.001
Sircelj H, Tausz M, Grill D, Batic F (2007) Detecting different levels of drought stress in apple trees (Malus domestica Borkh.) with selected biochemical and physiological parameters. Sci Hortic 113:362–369. https://doi.org/10.1016/j.scienta.2007.04.012
Sun G, Li W, Abulitifu Y (2016) Effects of regulated deficit irrigation on twig water potential of Korla fragrant pear. J Agric Sci Technol 17(8):1802–1833
Sun X, Yan H, Kang X, Ma F (2013) Growth, gas exchange, and water-use efficiency response of two young apple cultivars to drought stress in two scion-one rootstock grafting systems. Photosynthetica 51:404–410. https://doi.org/10.1007/s11099-013-0040-3
Sussmilch FC, Brodribb TJ, McAdam SA (2017) What are the evolutionary origins of stomatal responses to abscisic acid in land plants? J Integr Plant Biol 59:240–260. https://doi.org/10.1111/jipb.12523
Tang Y, Wang L, Ma C, Liu J, Liu B, Li H (2011) The use of HPLC in the determination of endogenous hormones in anthers of bitter melon. J Life Sci 5:139–142
Tuna AL, Kaya C, Ashraf M (2010) Potassium sulfate improves water deficit tolerance in melon plants grown under glasshouse conditions. J Plant Nutr 33:1276–1286. https://doi.org/10.1080/01904167.2010.484089
Wang Y, Chen ZH, Zhang B, Hills A, Blatt MR (2013) PYR/PYL/RCAR abscisic acid receptors regulate K+ and Cl− channels through reactive oxygen species-mediated activation of Ca2+ channels at the plasma membrane of intact Arabidopsis guard cells. Plant Physiol 163:566–577. https://doi.org/10.1104/pp.113.219758
Wang Z, Li G, Sun H, Ma L, Guo Y, Zhao Z, Gao H, Mei L (2018) Effects of drought stress on photosynthesis and photosynthetic electron transport chain in young apple tree leaves. Biol Open 7(11):bio035279. https://doi.org/10.1242/bio.035279
Warren C (2008) Rapid measurement of chlorophylls with a microplate reader. J Plant Nutr 31:1321–1332. https://doi.org/10.1080/01904160802135092
Wilkinson S, Davies WJ (2002) ABA-based chemical signaling: the coordination of responses to stress in plants. Plant Cell Environ 25:195–210. https://doi.org/10.1046/j.0016-8025,2001.00824.x
Wu X, Yin H, Shi Z, Chen Y, Qi K, Qiao X, Wang G, Cao P, Zhang S (2018) Chemical composition and crystal morphology of epicuticular wax in mature fruits of 35 pear (Pyrus spp) cultivars. Front Plant Sci 9:679. https://doi.org/10.3389/fpls.2018.00679
Yordanov I, Velikova V, Tsonev T (2000) Plant responses to drought, acclimation, and stress tolerance. Photosynthetica 38:171–186. https://doi.org/10.1023/A:1007201411474
Zhou S, Duursma RA, Medlyn BE, Kelly JW, Prentice IC (2013) How should we model plant responses to drought? An analysis of stomatal and non-stomatal responses to water stress. Agric for Meteorol 182:204–214. https://doi.org/10.1016/j.agrformet.2013.05.009
Zhou Y, Tian X, Yao J, Zhang Z, Wang Y, Zhang X, Li W, Wu T, Han Z, Xu X (2020) Morphological and photosynthetic responses differ among eight apple scion-rootstock combinations. Sci Hortic 261:108981. https://doi.org/10.1016/j.scienta.2019.108981
Zohouri M, Abdollahi H, Arji I, Abdossi V (2020) Variations in growth and photosynthetic parameters of some clonal semi-dwarfing and vigorous seedling pear (Pyrus spp.) rootstocks in response to deficit irrigation. Acta Sci Pol Hortorum Cultus 19(2):105–121. https://doi.org/10.24326/asphc.2020.2.11
Acknowledgements
We would like to thank Tarbiat Modares University (TMU) for its financial support. This work was supported by Ph.D. Student Grant Program by TMU. In addition, greenhouse and laboratory facilities provided by Pomology Lab., Department of Horticultural Science at TMU are acknowledged.
Author information
Authors and Affiliations
Contributions
Zeinab Maleki Asayesh: made a major contribution to data collection and statistical analysis, experimental work, writing—original draft, writing—review and editing. Kazem Arzani: managed a Ph.D. student grant from TMU authorities for the first author and supervised the design of the research and data collection, presentation, and reviewed with extensive manuscript draft revisions including the final revision of the article. Ali Mokhtassi-Bidgoli: consulted and helped with the statistical design of the experiment and data analysis. Hamid Abdollahi: advised on some data collection and reviewing the first draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. ‘Pyrodwarf’ and ‘Dargazi’ rootstocks showed different physiological and photosynthetic responses to water stress.
2. ‘Dargazi’ showed better resistance to long-duration drought stress compared to ‘Pyrodwarf’ rootstock.
3. The different root systems in the two pear rootstocks could be one of the factors involved in their differences in water stress tolerance.
4. Stomatal restrictions were one of the main causes of photosynthetic limitations under drought stress in pear rootstocks.
5. Photosynthesis (An) in ‘Pyrodwarf’ was more affected by the leaf ABA concentration than in ‘Dargazi’ rootstock.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asayesh, Z.M., Arzani, K., Mokhtassi-Bidgoli, A. et al. Gas Exchanges and Physiological Responses Differ Among ‘Pyrodwarf’ Clonal and ‘Dargazi’ Seedling Pear (Pyrus communis L.) Rootstocks in Response to Drought Stress. J Soil Sci Plant Nutr 23, 6469–6484 (2023). https://doi.org/10.1007/s42729-023-01502-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-023-01502-1