Abstract
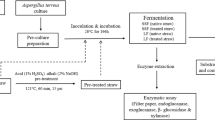
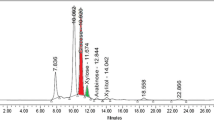
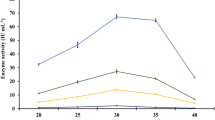
In order to improve the process feasibility and cost function of bioethanol production, higher titres of biomass saccharifying cellulases need to be produced on commercial scale. Diverse lignocellulosic substrates including abundantly accessible paddy straw can be utilized as carbonaceous substrates for the production of cellulose hydrolysing enzymes. This study aims to meliorate milled and sequential acid/alkali pre-treated paddy straw as inducer-substrates for the synthesis of fungal secretomes from Penicillium mallochii repertoires under solid state and liquid shake flask fermentation (SSF and LSF, respectively). The analysis of enzymatic activities of the respective secretomes reinforced the cellulolytic potential of P. mallochii where the maximum cellulase production (Filter paper cellulase: 76.43, Carboxymethyl cellulase: 130.29, Avicelase: 18.6 and β-glucosidase: 83.59 U L−1) was exhibited under LSF conditions using pre-treated paddy straw as the inducer. The disorganisation of the cellulosic structure via hydrogen bond disruption, as indicated by FTIR analysis, after acid/alkali pretreatment allowed a better establishment of fungus on the substrate, thereby facilitating higher cellulase production. A maximum ethanol content of 11.63 g L−1 was obtained at 48 h of simultaneous saccharification and fermentation using pre-hydrolysed paddy straw at 10% (w/v) solid loading and a cellulase dosage of 25 FPU gds−1. These results affirm the utilization of paddy straw for cellulase and ethanol production in an integrated bioprocess that can further be explored and optimized for scale-up studies to cater industrial applications.
Graphical Abstract







Similar content being viewed by others
Data availability
The data used to support the findings of this study are included within the article.
References
Alengebawy A, Ran Y, Ghimire N, Osman AI, Ai P (2023) Rice straw for energy and value-added products in China: a review. Environ Chem Lett 21(5):2729–2760. https://doi.org/10.1007/s10311-023-01612-3
Anu KA, Singh D, Kumar V, Singh B (2022) Production of cellulolytic enzymes by Myceliophthora thermophila and their applicability in saccharification of rice straw. Biomass Convers Biorefin 12:2649–2662. https://doi.org/10.1007/s13399-020-00783-1
Aruwajoye GS, Sewsynker-Sukai Y, Kana EG (2020) Valorisation of cassava peels through simultaneous saccharification and ethanol production: effect of prehydrolysis time, kinetic assessment and preliminary scale up. Fuel 278:1–11. https://doi.org/10.1016/j.fuel.2020.118351
Barbosa FC, Silvello MA, Goldbeck R (2020) Cellulase and oxidative enzymes: new approaches, challenges and perspectives on cellulose degradation for bioethanol production. Biotechnol Lett 42:875–884. https://doi.org/10.1007/s10529-020-02875-4
Binod P, Sindhu R, Singhania RR, Vikram S, Devi L, Nagalakshmi S, Kurien N, Sukumaran RK, Pandey A (2010) Bioethanol production from rice straw: an overview. Bioresour Technol 101:4767–4774. https://doi.org/10.1016/j.biortech.2009.10.079
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chen J, Zhang B, Luo L, Zhang F, Yi Y, Shan Y, Lü X (2021) A review on recycling techniques for bioethanol production from lignocellulosic biomass. Renew Sustain Energy Rev 149:111370. https://doi.org/10.1016/j.rser.2021.111370
Cuong TT, Le HA, Khai NM, Hung PA, Linh LT, Thanh NV, Tri ND, Huan NX (2021) Renewable energy from biomass surplus resource: potential of power generation from rice straw. Sci Rep 11:792. https://doi.org/10.1038/s41598-020-80678-3
David AN, Sewsynker-Sukai Y, Sithole B, Gueguim Kana EB (2020) Development of a green liquor dregs pretreatment for enhanced glucose recovery from corn cobs and kinetic assessment on various bioethanol fermentation types. Fuel 274:117797. https://doi.org/10.1016/j.fuel.2020.117797
de Barros EM, Carvalho VM, Rodrigues THS, Rocha MVP, Gonçalves LRB (2017) Comparison of strategies for the simultaneous saccharification and fermentation of cashew apple bagasse using a thermotolerant Kluyveromyces marxianus to enhance cellulosic ethanol production. Chem Eng J 307:939–947. https://doi.org/10.1016/j.cej.2016.09.006
El-Shishtawy RM, Mohamed SA, Asiri AM, Gomaa ABM, Ibrahim IH, Al-Talhi HA (2015) Saccharification and hydrolytic enzyme production of alkali pre-treated wheat bran by Trichoderma virens under solid state fermentation. BMC Biotechnol 15:1–13. https://doi.org/10.1186/s12896-015-0158-4
Goldbeck R, Ramos MM, Pereira GA, Maugeri-Filho F (2013) Cellulase production from a new strain Acremonium strictum isolated from the Brazilian Biome using different substrates. Bioresour Technol 128:797–803. https://doi.org/10.1016/j.biortech.2012.10.034
Grandmontagne D, Navarro D, Neugnot-Roux V, Ladevèze S, Berrin JG (2021) The secretomes of Aspergillus japonicus and Aspergillus terreus supplement the Rovabio® enzyme cocktail for the degradation of soybean meal for animal feed. J Fungi 7:1–15. https://doi.org/10.3390/jof7040278
Hans M, Kumar S, Chandel AK, Polikarpov I (2019) A review on bioprocessing of paddy straw to ethanol using simultaneous saccharification and fermentation. Process Biochem 85:125–134. https://doi.org/10.1016/j.procbio.2019.06.019
He Y, Pang Y, Liu Y, Li X (2008) Wang K (2008) Physicochemical characterization of rice straw pretreated with sodium hydroxide in the solid state for enhancing biogas production. Energy Fuels 22:2775–2781. https://doi.org/10.1021/ef8000967
** X, Song J, Liu GQ (2020) Bioethanol production from rice straw through an enzymatic route mediated by enzymes developed in-house from Aspergillus fumigatus. Energy. https://doi.org/10.1016/j.energy.2019.116395
Kaur A, Kuhad RC (2019) Valorization of rice straw for ethanol production and lignin recovery using combined acid-alkali pre-treatment. Bioenergy Res 12:570–582. https://doi.org/10.1007/s12155-019-09988-3
Kaur G, Taggar MS, Kalia A (2023) Cellulase-immobilized chitosan-coated magnetic nanoparticles for saccharification of lignocellulosic biomass. Environ Sci Pollut Res 30:111627–111647. https://doi.org/10.1007/s11356-023-27919-w
Kaur G, Kaur J, Taggar MS (2024a) Applications of biomass-derived materials for energy production. Biomass energy for sustainable development. CRC Press, pp 127–143
Kaur G, Krishania M, Taggar MS, Kalia A (2024b) Adsorptive removal of inhibitors from paddy straw hydrolysate using surfactant-modified bentonite clay for fermentative xylitol production. Biomass Convers Biorefin 14(1):1317–1328. https://doi.org/10.1007/s13399-023-04618-7
Kaur G, Taggar MS, Kalia A, Krishania M, Singh A (2024c) Fungal secretomes of Aspergillus terreus repertoires cultivated on native and acid/alkali treated paddy straw for cellulase and xylanase production. BioEnergy Res 17:145–159. https://doi.org/10.1007/s12155-023-10637-z
Kaur G, Taggar MS, Kalia A, Kaur J (2024d) Fungal cellulolytic enzyme complex immobilized on chitosan-functionalised magnetic nanoparticles for paddy straw saccharification. Process Saf Environ Prot 185:533–544. https://doi.org/10.1016/j.psep.2024.03.009
Khan MH, Ali S, Fakhru’l-Razi A, Alam Z (2007) Use of fungi for the bioconversion of rice straw into cellulase enzyme. J Environ Sci Health B 42:381–396. https://doi.org/10.1080/03601230701312647
Laltha M, Sewsynker-Sukai Y, Kana EG (2022) Simultaneous saccharification and citric acid production from paper wastewater pretreated banana pseudostem: optimization of fermentation medium formulation and kinetic assessment. Bioresour Technol 361:127700. https://doi.org/10.1016/j.biortech.2022.127700
Le M, Martínez Y, de Armas AC, González E (2020) Ethanol production from sugarcane straw using different configurations of fermentation and techno-economical evaluation of the best schemes. Renew Energy 156:377–388. https://doi.org/10.1016/j.renene.2020.04.091
Le TM, Tran UP, Duong YH, Nguyen KT, Tran VT, Le PK (2022) Development of a paddy-based biorefinery approach toward improvement of biomass utilization for more bioproducts. Chemosphere 289:133249. https://doi.org/10.1016/j.chemosphere.2021.133249
Logeswaran J, Shamsuddin AH, Silitonga AS, Mahlia TMI (2020) Prospect of using rice straw for power generation: a review. Environ Sci Pollut Res 27:25956–25969. https://doi.org/10.1007/s11356-020-09102-7
Maleki M, Ariaeenejad S, Salekdeh GH (2022) Efficient saccharification of ionic liquid-pretreated rice straw in a one-pot system using novel metagenomics derived cellulases. Bioresour Technol 345:1–11. https://doi.org/10.1016/j.biortech.2021.126536
Mandels M, Sternberg D (1976) Recent advances in cellulase technology. Ferment Technol 54:256–286
Mandels M, Weber J (1976) The production of cellulases. Adv Chem 95:391–414
Miah R, Siddiqa A, Tuli JF, Barman NK, Dey SK, Adnan N, Yamada M, Talukder AA (2017) Inexpensive procedure for measurement of ethanol: application to bioethanol production process. Adv Microbiol 7:743–748. https://doi.org/10.4236/aim.2017.711059
Nelson N (1944) The colorimetric determination of reducing sugars. J Biol Chem 153:375–379
Ningthoujam R, Jangid P, Yadav VK, Sahoo DK, Patel A, Dhingra HK (2023) Bioethanol production from alkali-pretreated rice straw: effects on fermentation yield, structural characterization, and ethanol analysis. Front Bioeng Biotechnol 11:1243856
Paschos T, Louloudi A, Papayannakos N, Kekos D, Mamma D (2022) Potential of barley straw for high titer bioethanol production applying pre-hydrolysis and simultaneous saccharification and fermentation at high solid loading. Biofuels 13:467–473. https://doi.org/10.1080/17597269.2020.1760688
Phitsuwan P, Permsriburasuk C, Baramee S, Teeravivattanakit T, Ratanakhanokchai K (2017) Structural analysis of alkaline pretreated rice straw for ethanol production. Int J Polym Sci 17:4676969. https://doi.org/10.1155/2017/4876969
Rajamani S, Kolla SSN, Gudivada R, Raghunath R, Ramesh K, Jadhav SA (2023) Valorization of rice husk to value-added chemicals and functional materials. Int J Environ Res 17(1):22. https://doi.org/10.1007/s41742-023-00512-2
Santos F, Machado G, Faria D, Lima J, Marçal N, Dutra E, Souza G (2017) Productive potential and quality of rice husk and straw for biorefineries. Biomass Convers Biorefin 7:117–126. https://doi.org/10.1007/s13399-016-0214-x
Sawasdee A, Haosagul S, Pisutpaisal N (2023) Biogas production from co-digestion between rice straw and food waste with pilot scale. Int J Environ Res 17(2):31. https://doi.org/10.1007/s41742-023-00523-z
Sewsynker-Sukai Y, Kana EG (2018) Simultaneous saccharification and bioethanol production from corn cobs: process optimization and kinetic studies. Bioresour Technol 262:32–41. https://doi.org/10.1016/j.biortech.2018.04.056
Singh B, Bala A, Anu A, Kumar V, Singh D (2022) Biochemical properties of cellulolytic and xylanolytic enzymes from Sporotrichum thermophile and their utility in bioethanol production using rice straw. Prep Biochem Biotechnol 52:197–209. https://doi.org/10.1080/10826068.2021.1925911
Singh S, Kaur G, Singh DP, Arya S, Krishania M (2024) Exploring rice straw’s potential from a sustainable biorefinery standpoint: towards valorization and diverse product production. Process Saf Environ Prot 184:314–333. https://doi.org/10.1016/j.psep.2024.01.105
Siqueira JGW, Rodrigues C, de Souza Vandenberghe LP, Woiciechowski AL, Soccol CR (2020) Current advances in on-site cellulase production and application on lignocellulosic biomass conversion to biofuels: a review. Biomass Bioenerg 132:105419. https://doi.org/10.1016/j.biombioe.2019.105419
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D, Crocker DLAP (2010) Determination of structural carbohydrates and lignin in biomass. Lab Anal Proced 1617:1–16 (TP-510-42618)
Sreejith RP, Sankar M, Sukumaran RK, Sivaraman S (2022) Rapid estimation of the chemical composition of rice straw using FTIR spectroscopy: a chemometric investigation. Biomass Convers Biorefin 9:1–19. https://doi.org/10.1007/s13399-022-03508-8
Sriariyanun M, Kitiborwornkul N, Tantayotai P, Rattanaporn K, Show PL (2022) One-pot ionic liquid-mediated bioprocess for pretreatment and enzymatic hydrolysis of rice straw with ionic liquid-tolerance bacterial cellulase. Bioengineering 9(1):17. https://doi.org/10.3390/bioengineering9010017
Srivastava N, Singh R, Srivastava M, Syed A, Pal DB, Bahkali AH, Mishra PK, Gupta VK (2022) Impact of mixed lignocellulosic substrate and fungal consortia to enhance cellulase production and its application in NiFe2O4 nanoparticles mediated enzymatic hydrolysis of wheat straw. Bioresour Technol 345:126560. https://doi.org/10.1016/j.biortech.2021.126560
Srivastava N, Singh R, Verma B, Rai AK, Tripathi SC et al (2023) Microbial cellulase production and stability investigations via graphene like carbon nanostructure derived from paddy straw. Int J Biol Macromol 237:124033. https://doi.org/10.1016/j.ijbiomac.2023.124033
Sun S, Huang Y, Sun R, Tu M (2016) The strong association of condensed phenolic moieties in isolated lignins with their inhibition of enzymatic hydrolysis. Green Chem 18:4276–4286. https://doi.org/10.1039/C6GC00685J
Sutaoney P, Rai SN, Sinha S, Choudhary R, Gupta AK, Singh SK, Banerjee P (2024) Current perspective in research and industrial applications of microbial cellulases. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2024.130639
Taiwo AE, Tom-James A, Falowo OA, Okoji A, Adeyi O, Olalere AO, Eloka-Eboka A (2022) Techno-economic analysis of cellulase production by Trichoderma reesei in submerged fermentation processes using a process simulator. S Afr J Chem Eng 42:98–105. https://doi.org/10.1016/j.sajce.2022.08.001
Tareen AK, Punsuvon V, Sultan IN, Khan MW, Parakulsuksatid P (2021) Cellulase addition and pre-hydrolysis effect of high solid fed-batch simultaneous saccharification and ethanol fermentation from a combined pretreated oil palm trunk. ACS Omega 6:26119–26129. https://doi.org/10.1021/acsomega.1c03111
Toan NS, Nguyen TDP, Thu TTN, Lim DT, Dong PD, Gia NT, Khoo KS, Kit WC, Show PL (2021) Soil mineralization as effects of plant growth promoting bacteria isolated from microalgae in wastewater and rice straw application in a long-term paddy rice in Central Viet Nam. Environ Technol Innov 24:101982. https://doi.org/10.1016/j.eti.2021.101982
Toan NS, Phuong NTD, Thuy PT, Dong PD, Gia NT, Thu TTN, Show PL (2022) Effects of burning rice straw residue on-field on soil organic carbon pools: environment-friendly approach from a conventional rice paddy in central Viet Nam. Chemosphere 294:133596. https://doi.org/10.1016/j.chemosphere.2022.133596
Yan H, Zhang Q, Wang Y, Cui X, Liu Y, Yu Z, Xu S, Ruan R (2023) Rice straw as microalgal biofilm bio-carrier: effects of indigenous microorganisms on rice straw and microalgal biomass production. J Environ Manage 341:118075. https://doi.org/10.1016/j.jenvman.2023.118075
Zhang Y, **n D, Wen P, Chen X, Jia L, Lu Z, Zhang J (2023) Comparison of alkaline sulfite pretreatment and acid sulfite pretreatment with low chemical loading in saccharification of poplar. Appl Biochem Biotechnol 195:4414–4428. https://doi.org/10.1007/s12010-023-04351-x
Funding
This work was supported by the Department of Science and Technology, Government of India under DST-INSPIRE (Innovation in Science Pursuit for Inspired Research) Fellowship program (Grant Number IF190819).
Author information
Authors and Affiliations
Contributions
GK: Investigation, data curation, formal analysis, resources, software, writing- original draft; MST: Conceptualization, supervision, resource acquisition, validation, writing- review and editing; AK: resource acquisition, supervision, and writing: review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, G., Taggar, M.S. & Kalia, A. Bio-valorising Paddy Straw as an Inducer-Substrate for Ethanol Production using Fungal Secretome of Penicillium mallochii. Int J Environ Res 18, 68 (2024). https://doi.org/10.1007/s41742-024-00621-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41742-024-00621-6