Abstract
Luminescence chemosensors are one of the most useful tools for the determination and imaging of small biomolecules and ions in situ in real time. Based on the unique photo-physical/-chemical properties of ruthenium(II) (Ru(II)) complexes, the development of Ru(II) complex-based chemosensors has attracted increasing attention in recent years, and thus many Ru(II) complexes have been designed and synthesized for the detection of ions and small biomolecules in biological and environmental samples. In this work, we summarize the research advances in the development of Ru(II) complex-based chemosensors for the determination of ions and small biomolecules, including anions, metal ions, reactive biomolecules and amino acids, with a particular focus on binding/reaction-based chemosensors for the investigation of intracellular analytes’ evolution through luminescence analysis and imaging. The advances, challenges and future research directions in the development of Ru(II) complex-based chemosensors are also discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Small biomolecules and ions are building blocks of living organisms and play indispensable roles in all biological processes, such as enzymatic reactions, metabolism, growth, adaptation, and various disease developments and progressions [1, 2]. Determination and monitoring of the levels of these biomolecules and ions in situ are essential for better understanding their biological roles in biomedical systems, thus contributing to early diagnosis and treatment assessment of various diseases [3,4,5]. The most common and typical methods, such as high-performance liquid chromatography (HPLC) and inductively coupled plasma-optical emission spectroscopy/mass spectrometry (ICP-OES/MS), have been developed for the determination of small biomolecules and ions in vitro [6,7,8]. Onsite determination of these analytes in situ and in vivo, using these techniques, is not possible because the sample preparation in solution is an essential step to ensure performing successful analysis [9]. Imaging technologies, such as computerized tomography (CT) [10], magnetic resonance imaging (MRI) [11] and positron emission tomography (PET), have been widely used in clinical diagnosis [12], while these technologies cannot be directly used for the determination of the concentration and/or activity of these analytes in biological samples [13, 14]. This is mainly because the contrast agents (CAs) used in these technologies are generally nonspecific; more importantly, these CAs hardly respond to small biomolecules and ions at molecular level because of their resolution and sensitivity limitations [15]. Other approaches to optical detection, such as fluorescence and phosphorescence measurements, have also been successfully developed and adopted in biomedical research and clinical diagnosis [16]. In contrast to conventional bioassay and imaging technologies, luminescence bioassay and imaging using advanced optical spectroscopic and imaging instruments are featured with high sensitivity and selectivity, fast response time and low cost, enabling their use in biological and biomedical investigations involving in vitro bioassay and in vivo luminescence bioimaging [17,18,19,20].
Chemosensors are one of the most important tools for luminescence bioassay and imaging of small biomolecules and ions in situ in real time [21,22,23]. Luminescent chemosensors are normally designed as chemical compounds that can respond to targeted analytes through a unique binding/reaction (Fig. 1) [24, 25]. Generally, the chemosensors with reaction-based sensing mechanisms have higher selectivity, and the chemosensors with binding-based sensing mechanisms feature excellent reversibility for monitoring the targeted analyte in situ. As a result of these response processes, the luminescence signals can be switched “ON” (Fig. 1A) or “OFF” (Fig. 1B). Of the luminescence switch “OFF” and “ON” responses, the emission switch “ON” chemosensors are preferable for imaging analysis because the enhancement of the luminescence intensity can be easily observed by microscopy. The emission wavelengths of the chemosensors can also be shifted after the response processes (Fig. 1C) [25,26,27], allowing ratiometric luminescence detection and imaging of targeted analytes with the potential for precise and quantitative analyses. The changes of emission signals generally correspond to the concentrations of the targeted analyte and thus can be recorded for the analyte’s determination of abundance by luminescence spectroscopes and/or microscopes. Because of the unique advantages of luminescence bioassays and imaging, enormous efforts have been devoted to the development of luminescent chemosensors for the detection of a variety of analytes in complicated biological and environmental systems in the past few decades.
As shown in Fig. 1, luminescence chemosensors generally consist of three parts, including the luminophore, response unit and spacer, which links the luminophore and response unit. Of all luminophores that are being used for the development of chemosensors, fluorescent organic dyes are most widely investigated because of their high quantum yields (ϕ) and easy modification of chemical structures [28]. Lanthanide chelates are another family of luminophores that have been successfully employed in the development of chemosensors [29, 30]. Compared with fluorescent organic dyes, lanthanide chelate luminescence has high photostability, large Stokes shift and unique line-like emissions [29]. The prolonged lifetime of lanthanide chelates (microseconds to milliseconds) enables a background-free bioassay and imaging of targeted analytes through time-gated luminescence (TGL) measurement [28, 31]. Transition metal complexes, particularly the luminescent ruthenium(II) (Ru(II)) [32], iridium(III) (Ir(III)) [33], platinum(II) (Pt(II)), gold(I) (Au(I)) [34], rhenium(I) (Re(I)) [35] and osmium(II) (Os(II)) complexes with d6, d8 and d10 electron structures, have also been studied when develo** chemosensors for biomolecule and ion detection and imaging [36,37,38,39,40]. Different from the fluorescent organic dyes that emit from excited singlet state, phosphorescence of transition metal complexes is derived from excited triplet states [41]. The excited states of these transition metal complexes are more complicated than those of fluorescent dyes and mainly include metal-to-ligand charge transfer (MLCT), intraligand charge transfer (ILCT), ligand-to-ligand charge transfer (LLCT), metal–metal-to-ligand charge transfer (MMLCT), ligand-to-metal charge transfer (LMCT), metal-to-ligand-ligand charge transfer (MLLCT) and ligand-to-metal–metal charge transfer (LMMCT) [41,42,43]. The excited state-mediated emission properties of transition metal complexes are varied upon the changes of the metal center, local environment and particularly chemical structure of ligands, enabling transition metal complexes to be designed as the chemosensors through modulating these parameters [43].
Of all transition metal complex-based luminophores, Ru(II) polypyridine complex, particularly the prototype of the Ru complex ([Ru(bpy)3]2+ (bpy: 2,2′-bipyridine)) (Fig. 2A), has been one of the most popular molecules and widely investigated in the past few decades [44]. Ru(II) polypyridine complexes have octahedral symmetry with three kinds of electronic transitions, including metal centered (MC), ligand centered (LC) and MLCT [41]. As shown in Fig. 2B, in this octahedral symmetry of Ru(II) complex, MC excited states are obtained for an electron transition from πM to σ*M orbitals, LC excited states are formed through an electron transition from πL to π*L, and MLCT excited states are produced by promotion of an electron from πM metal orbital to π*L ligand orbitals [45]. The lowest excited state MC can decay to the ground state through a fast radiationless process. In contrast, the lowest excited states LC and MLCT undergo radiative deactivation to the ground state, thus exhibiting intense luminescence at room temperature in a rigid matrix and fluid solution, respectively [41]. Consequently, the lowest excited state is 3MLCT for most luminescent Ru(II) polypyridine complexes in solution. Upon the excitation at about 450 nm (spin-allowed 1MLCT), the lowest spin-forbidden 3MLCT excited state is obtained after a fast intersystem crossing process and then emits orange to near infrared emission [46]. The 3MLCT-based emission of Ru(II) polypyridine complexes displays unique photochemical and photophysical properties, including large Stokes shift (about 150 nm), prolonged luminescence lifetime (hundreds of nanoseconds to microseconds level), high photostability and brightness by visible light excitation [41, 47].
The emission of Ru(II) complexes, including luminescence intensity and lifetime, can be fine-modulated by modifying the chemical structure of ligands, allowing the Ru(II) complexes to be used for the development of chemosensors for biomolecule and ion detection. As described in early review articles [42, 43, 48, 49], the response mechanisms of luminescent Ru(II) complex chemosensors for target analytes mainly include (1) photo-induced electron transfer (PeT), in which the Ru(II) polypyridine complexes are ideal electron donors and acceptors, (2) Förster resonance energy transfer (FRET), in which the Ru(II) complexes can serve as the energy donor and acceptor [50,51,52,53] for the energy transfer (ET), (3) distortion of ligand and the octahedral symmetry after binding/reaction with the target analyte and (4) changes of the local environment upon the analyte’s binding. In addition to the luminescence response of Ru(II) complex-based chemosensors to the analyte [40, 54, 55], other prerequisites for this family of chemosensors to be applied in bioassay and imaging include (1) capability of analyte determination in aqueous solution, (2) high sensitivity and selectivity, which allow the chemosensors to be used for targeted analyte detection even at extremely low concentration without non-specific binding of interference species, (3) low cytotoxicity, enabling targeted analyte determination and imaging with minimum perturbation to the native micro-environment and (4) high cell membrane permeability to ensure the chemosensors are easily internalized into biological tissues.
The last few years have witnessed a huge leap forward in the development of Ru(II) complexes as the chemosensors for the detection of various analytes. Based on the above-described response mechanisms, hundreds of Ru(II) complex chemosensors for colorimetric and luminescent determination of anions, metal ions, small biomolecules and biomacromolecule have been available [42, 43]. For example, the triplet nature of the emission state with long lifetime of Ru(II) complexes allows them to be used for oxygen sensing by monitoring the changes of their luminescence intensity and lifetime [56,57,58]. Some of the Ru(II) complexes have also been revealed to be nucleic acid sensitive [59, 60], thus serving as a “light switch” for DNA detection [61]. The Ru(II) complexes with dipyridophenazine (dppz) ligands (e.g., complex 1 ([Ru(phen)2(dppz)]2+) are particularly interesting (Fig. 3A) [61]. These complexes are non-luminescent (undetectably small quantum yield) in water, while the emission intensity is significantly increased when bound to DNA [62, 63], which allows the Ru(II) complexes (e.g., complex 2) to be used for imaging of DNA structure and related mitosis progression (Fig. 3A). Subsequent research has also revealed that Ru(II) complexes (e.g., complex 3) can be used for the determination of DNA mismatches and RNA (Fig. 3B) [64,65,66,67]. Since the investigation of cellular uptake and imaging of the Ru(II) complex by Barton’s group [68, 69], the application of Ru(II) complex chemosensors for sensing and imaging of intracellular biomolecules and ions has increasingly attracted interest in recent years.
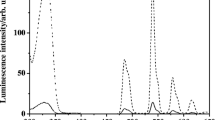
Molecular structures of examples of Ru(II) complexes 1 ([Ru(phen)2(dppz)]2+) and 2 for DNA determination and imaging (A) and complex 3 ([Ru(Me4phen)2(dppz)]2+) for detection of DNA mismatches. The luminescence spectra represent the emission of complex 3 with well-matched and mismatched DNA. Adapted with permission from Ref. [67].
In this chapter, we wish to summarize recent examples of Ru(II) complex chemosensors for the detection of small biomolecules and ions in aqueous solution, with a particular focus on binding/reaction-based chemosensors for the investigation of intracellular analytes’ evolution through luminescence imaging. Specifically, the chemosensors for the determination of reactive oxygen/nitrogen/carbonyl species (ROS/RNS/RCS), biothiols, amino acids, pH, metal ions and anions are summarized, followed by the discussion of these chemosensors for luminescence bioimaging. The advances, challenges and future research directions in the development of Ru(II) complex-based chemosensors will also be discussed.
2 Ru(II) Complex Chemosensors for Anions
In various chemical and biological processes, anions play important roles in the body, such as blood pressure stabilization, blood purification, sugar level reduction, respiration and fatigue recovery. For anion sensing, the development of Ru(II) complex-based chemosensors has attracted enormous attention in the past few decades [70,71,72]. By virtue of their abundant photo-physical and chemical properties, hundreds of Ru(II) complexes have been designed and synthesized for the detection of various anions, such as fluoride (F−) [73,74,75,76], acetate (CH3COO−) [77], cyanide (CN−)[78], phosphate (H2PO4−) [79,80,81], chloride (Cl−) and bromide (Br−). Similar to the design of other anion receptors [82], most Ru(II) complex-based chemosensors are designed using the following three response mechanisms, including (1) the binding of the Ru(II) complex’s recognition unit with anions via hydrogen bonding and deprotonation [83], electrostatic and Lewis acid–base interactions [84], (2) specific reactions of the Ru(II) complex’s recognition unit with anions [85] and (3) displacement of metal ions from heterobimetallic Ru(II) complex [86]. In the following section, the Ru(II) complex-based chemosensors for anions will be discussed according to their different response mechanisms.
2.1 Response Based on Hydrogen Bonding, Electrostatic and Lewis Acid-Base Interactions
Although most Ru(II) complex-based anion chemosensors are developed through the mechanism of hydrogen bonding and electrostatic and Lewis acid-base interactions, the colorimetric and luminescent response of these Ru(II) complexes to anions can only be obtained in organic solvents, including acetonitrile (CH3CN) and dimethyl sulfoxide (DMSO) [87, 88]. This is mainly because the hydrogen bonding, electrostatic and Lewis acid-base interactions are significantly inhibited by the water molecules and other anions in the buffer solution [89]. This sub-section will discuss some examples of Ru(II) complex chemosensors for determination of anions in aqueous solution [90,91,92].
Through modification of the [Ru(bpy)3]2+ with amide containing a calixarene moiety (binding site), Maity et al. reported a Ru(II) complex (4) for CN− and CH3COO− determination (Fig. 4) [93]. Titration of complex 4 with CN− and CH3COO− in H2O–CH3CN (95:5) resulted in a remarkable luminescence quenching and enhancement, respectively. The different response mechanisms of complex 4 to CN− and CH3COO− were investigated by 1H NMR spectra. The CN− can bind to each amide N–H and leads to the deprotonation to form HCN. The electron density on the bpy ligand increased, and thus the intramolecular quenching was enhanced. In contrast, the weak interaction of bidentate CH3COO− with two N–H protons led to the formation of electron delocalization, in which the CH3COO− pulls the electron density to itself and thus decreases the intramolecular quenching. The luminescence response of complex 4 to CN− showed a LoD of 70 ppb.
Mardanya and co-workers described a Ru(II) complex (5) with pyrene-biimidazole ligand as a chemosensor for highly selective CN− detection in both CH3CN and aqueous media (Fig. 5) [94]. The imidazole N–H protons of the coordinated ligand were found to be highly acidic with pKa1 = 5.09 and pKa2 = 8.95. Deprotonation of these two N–Hs was found through hydrogen bonding interaction with CN−, leading to the increase of electron density of the metal center. As a result, red shift of absorption and quenching of emission were obtained for complex 5 after CN− binding. Detection limit of complex 5 to CN− was determined to be 5.24 × 10–9 and 4.67 × 10–9 M for colorimetric and luminescent analyses, respectively. A similar hydrogen bonding-based interaction has been employed for the development of Ru(II) complex 6 for selective detection of thiocyanate (SCN−) (Fig. 5) [95]. In complex 6, the SCN− interacts with N–H through a 1:1 binding mode, which hinders the photo-induced electron transfer (PeT) between the long pair electron of the N atom and the Ru(II) complex. The increase of luminescence intensity was thus recorded for SCN− detection in CH3CN-HEPES buffer solution (1:1, v/v, pH = 7.2).
In an early study, Lin et al. reported a Ru(II) complex (7) for highly selective detection of F− in water by naked eye and luminescence (Fig. 6) [96]. Complex 7 was developed by incorporating a Schiff-base ligand with two bpy ligands. In the presence of F−, the conversion of quinonehydrazone moiety to azophenol could occur, resulting in a remarkable red shift of absorption spectra from 475 to 580 nm and a solution color change from orange to blue-violet. The binding of F− also led to the increase of luminescence intensity at 630 nm. Although the spectrometric responses were measured in CH3CN, the test paper prepared by staining of complex 7 also showed color changes in aqueous solution. In a later study, the same group modified the 1,10-phenanthroline-5,6-dione ligand to produce complex 8 for F− detection (Fig. 6) [97]. In the presence of F−, a similar red shift of absorption spectra (from 467 to 580 nm), color change (from yellow to magenta) and luminescence enhancement were obtained, which were attributed to the F−-mediated hydrogen bonding and deprotonation of N–H. The test papers were also prepared for F− detection in aqueous solution with ten times higher sensitivity (LoD = 1 ppm) than complex 7.
Molecular structure of Ru(II) complexes 7 and 8 as chemosensors for F−. The test paper prepared by complex 7 was then used for F− detection in aqueous solution. Adapted with permission from Ref. [96].
2.2 Response Based on Specific Reactions
Compared with the above-discussed response mechanism of hydrogen bonding and Lewis acid-base interactions, the chemosensors developed by the mechanism of chemical reaction have high sensitivity and selectivity [78, 98, 99]. The interference from water is also minimized because specific chemical reactions are involved in the sensing mechanism of these chemosenosors. Aldehyde is a strong electron-withdrawing group that can quench the MLCT emission of Ru(II) complex. In a previous study, a Ru(II) complex (9, [Ru(CHO-bpy)3]2+) with three aldehyde functionalized bpy ligands was designed and synthesized by Zhang et al. as the chemosensor for biothiol (cysteine-Cys and homocysteine-Hcy) detection in DMSO-HEPES buffer [100]. In a later study, Zhang et al. found that the nucleophilic addition of aldehyde can also be triggered by hydrogen sulfite (HSO3−) in acidic buffer [24], thus allowing complex 9 to be used as a chemosensor for HSO3− detection in phosphate-buffered saline (PBS) buffer (50 mM, pH = 5) (Fig. 7) [101]. The reaction of HSO3− and complex 9 led to the formation of 9-SO, accompanied by an increase of luminescence at 620 nm. The result of HSO3− detection in wine and sugar samples showed that complex 9 has good precision and accuracy for HSO3− in food samples. Using 5-formyl-2,2′-bipyridine as the ligand, Zhu et al. prepared a Ru(II) complex (10) as the luminescence chemosensor for CN− detection (Fig. 7) [102]. Similar to [Ru(CHO-bpy)3]2+, complex 10 in CH3CN-H2O (6:4) showed weak emission at 700 nm. The nucleophilic attack by CN− led to the formation of 10-CN, accompanied by a blue shift and increase of emission at 618 nm. Complex 10 with a detection limit of 0.75 μM was then used for the preparation of a test paper for naked eye CN− detection.
In addition to aldehyde, nucleophilic addition between the azo (N = N) group and HSO3− has recently been exploited for the development of chemosensors for HSO3− detection (Fig. 8A) [24, 85]. Owing to the PeT from the Ru(II) center to the attached azo-2,4-dinitrobenzene (DNB), Ru(II) complex 11 (Ru-azo) exhibited weak emission in 25 mM PBS buffer of pH 7.4. The HSO3− triggered reaction with the azo group led to the formation of 11-SO3 (Ru-SO3), accompanied by an enhancement in luminescence at 635 nm. More interestingly, complex 11 has a long emission lifetime of 258 ns, which enabled its use for background-free luminescence detection of HSO3− through a TGL mode. In a wine sample containing rhodamine (spiked as the artificial background signal), steady-state luminescence analysis of HSO3− failed (Fig. 8B). TGL analysis eliminated the background signals and allowed for HSO3− detection with high accuracy and precision (Fig. 8C).
Molecular structure of Ru(II) complex 11 and its reaction with HSO3− (A). Detection of HSO3− in rhodamine (RhB)-contaminated wine samples by steady-state luminescence (B) and TGL (C) analyses. Adapted with permission from Ref. [85].
2.3 Response Based on Displacement of Metal Ions
By modifying the ligands with additional coordination sites, the produced Ru(II) complexes are capable of binding to other metal ions, such as Cu2+, Co2+, Zn2+ and Hg2+, to form heterobimetallic complexes. The heterobimetallic Ru(II) complexes thus can be used as chemosensors for anion sensing through a displacement approach. Different from the hydrogen bonding-based anion sensing approach, the displacement-based response mechanism also allowed the chemosensors to be used for anion detection in water because the water molecules are not involved in the displacement processes. Among various heterobimetallic Ru(II) complexes, the one with Cu2+ (Ru(II)–Cu(II)) has been widely studied because Ru(II) complex luminescence can be quenched by Cu2+ binding through electron and energy transfers. Then, Cu2+ can be displaced in the presence of several anions, including sulfide (S2−), CN− and pyrophosphate (PPi) (Fig. 9A).
By coupling di(2-picolyl)amine (DPA) to one bpy ligand, Zhang et al. synthesized a Ru(II) complex (12) and then demonstrated the corresponding heterobimetallic Ru(II)–Cu(II) complex as the chemosensor for S2− detection (Fig. 9B) [86]. Complex 12 showed high luminescence (ϕ = 3.07%) in HEPES buffer (pH 7.2), while the luminescence was completely quenched upon binding to Cu2+ through a 1:1 coordination stoichiometry. In the presence of S2−, Cu2+ was then displaced to form the original complex 12, which was accompanied by the recovery of luminescence. This heterobimetallic Ru(II)–Cu(II) complex showed high sensitivity to S2− (LoD = 20.7 nM), allowing its use for S2− detection in three wastewater samples. In a similar work, Li et al. modified the bpy ligand with a Cu2+ receptor, 1,4,7,10-tetraazacyclododecane (cyclen), and then demonstrated the Ru(II) complex (13) for sequential Cu2+ and S2− detection (Fig. 9B) [103]. Compared with complex 12, complex 13 has better selectivity to Cu2+ binding, while the formed 13-Cu is not as sensitive as 12-Cu for S2− sensing.
The displacement approach has also been employed for the development of Ru(II) complex chemosensors for CN−. In 2017, two Ru(II) complexes (14 and 15) were synthesized by Zheng and co-workers, and the corresponding heterobimetallic Ru(II)–Cu(II) complexes were used for CN− detection in 20 mM HEPES buffer (pH 7.2) (Fig. 9B) [104]. A recovery of luminescence was observed after the displacement of Cu2+ from 14-Cu to 15-Cu to form [Cu(CN−)X]n− and complexes 14 and 15. The LoD for CN− was then determined to be 0.36 and 0.87 μM using 14-Cu and 15-Cu as the chemosensors, respectively. Similarly, Zhang et al. reported a Ru(II) complex (16) for PPi detection in 2018 (Fig. 9B) [105], in which one Cu2+ from the 16-Cu was displaced by the addition of two PPi. The chemosensor 16-Cu showed high sensitivity (LoD = 0.58 nM) for PPi detection in 10 mM HEPES buffer (pH 7.4).
3 Ru(II) Complex Chemosensors for pH
Similar to most pH sensors, Ru(II) complex chemosensors for pH have mainly been developed through the mechanism of protonation and deprotonation of several functional groups, such as imidazole [106, 107], hydroxyl (–OH) [108,109,110], carboxyl (–COOH) [111], pyridine [112, 113] and others [114]. As a result of the protonation-deprotonation process, the molecular structures and the electron density distribution of Ru(II) complex are changed, leading to the variations of absorption and emission intensity/wavelength. In this section, the progress in the development of Ru(II) complex chemosensors for pH will be briefly discussed.
As described above, deprotonation of N–H of Ru(II) complex 5’s 2,2′-biimidazole ligand occurs under basic conditions or binding with CN− [94]. In 2020, Tormo et al. also reported the use of imidazole-based ligand for the development of Ru(II) complex chemosensors for pH [115]. In later research, deprotonation of imidazole N–H under neutral and basic conditions was exploited by Yu et al. for the development of Ru(II) complex 17 ([Ru(bim)2(pip)]2+) for pH sensing and imaging (Fig. 10A) [116]. The increase of pH led to the deprotonation of all imidazole N–H from both bim and pip ligands, resulting in red shift of absorption spectra and decrease of MLCT emission. Complex 17 has a pKa of 4.49 and low cytotoxicity, enabling lysosome imaging in U251 cells. Luminescence images of U251 cells with complex 17 and LysoTracker Red showed good co-localization (Fig. 10B), and then the application of complex 17 in monitoring of intracellular pH changes was demonstrated by treating the U251 cells with lysosomal acidification inhibitor (bafilomycin A1).
Molecular structure of pH-sensitive Ru(II) complex 17 (A). Luminescence co-localization imaging of U251 cells stained with complex 17 and DYPI and LysoTracker Red (B). Adapted with permission from Ref. [116].
In 2015, Zheng and coworkers synthesized two Ru(II) complexes (18 and 19) and investigated their absorption and luminescence responses to pH (Fig. 11A) [112]. Protonation-deprotonation process occurs on the imidazole N–H and both imidazole N–H and pyridine for complexes 18 and 19, respectively. The two-step protonation-deprotonation processes resulted in complex 18 with pKa1 = 0.98 ± 0.04 and pKa2 = 8.34 ± 0.03, while the three-step protonation-deprotonation processes of complex 19 exhibited pKa1 = 1.86 ± 0.02, pKa2 = 3.43 ± 0.04 and pKa3 = 9.07 ± 0.08. Coordination of the terpyridine (tpy) ligand of complex 19 with Re(I), a heterobimetallic Ru(II)–Re(I) complex 20, was developed by Zheng et al. in 2014 (Fig. 11A) [113]. The coordination of the tpy ligand blocked the protonation-deprotonation of one pyridine; thus, a two-step protonation-deprotonation process was obtained. The pKa1 and pKa2 value of complex 20 was 1.38 ± 0.03 and 6.84 ± 0.04, respectively. Interestingly, the coordination with Re(I) significantly improved the biocompatibility, allowing complex 20 to be used for luminescence imaging in HeLa cells.
Molecular structures of pH-sensitive Ru(II) complexes 18–22 (A) and the application of complex 22 for HeLa cancer cell pH imaging (B) and discrimination from healthy HEK293 cells (C). Adapted with permission from Ref. [118].
Dinuclear Ru(II) complex 21 was then synthesized by Meng and co-workers in 2017 (Fig. 11A) [117]. Ru(II) complex 21 showed NIR emission at 760 nm with a large Stokes shift of 254 nm and lifetime (τ) 108.3 ± 0.4 ns. Different from complex 20, a three-step protonation-deprotonation process was observed on imidazole N–H at the second ligand. pKa values of complex 21 changed to pKa1 = 1.36 ± 0.02, pKa2 = 5.76 ± 0.05 and pKa3 = 9.01 ± 0.14. Through modification of the second ligand, the same group recently reported a Ru(II) complex (22) as a chemosensor for pH imaging and cancer cell discrimination (Fig. 11A) [118]. Compared with complex 20, similar photophysical properties, including intense NIR emission (~ 700 nm) and large Stokes shift (~ 200 nm) were obtained for complex 22. More importantly, the pKa2 value of complex 22 was determined to be 7.87, which is closer to the physiological value (i.e., 7.0–7.4). This allowed complex 22 to be used for luminescence imaging of intracellular pH in lysosomes (Fig. 11B). Moreover, imaging of HeLa cells showed about 13-fold higher intensity than HEK293 cells (Fig. 11C), demonstrating the “distinguishing” ability of complex 22 to identify the tumor and healthy cells.
4 Ru(II) Complex Chemosensors for Metal Ions
In addition to the anions, metal ions also play important roles in biological and environmental systems. Some metal ions, such as Cu2+, Fe3+ and Zn2+, are essential elements in the human body, while the metal ions, such as Hg2+, Cd2+ and Cr3+, are highly toxic, causing several problems for biological and environmental systems [119]. To detect these metal ions in biological and environmental systems, a number of Ru(II) complex chemosensors have been developed in the past few decades. In this section, the progress in the development of chemosensors for Cu2+, Hg2+ [120] and others will be discussed according to the types of ions.
4.1 Ru(II) Complex Chemosensors for Cu2+
As described above, the binding of Ru(II) complexes with Cu2+ could lead to the quenching of their luminescence through an excited-state electron transfer or energy transfer mechanism [86, 104, 105]. By virtue of this mechanism, a series of Ru(II) complexes have been developed as luminescence “ON–OFF” chemosensors for Cu2+ determination and imaging (Fig. 12). Complex 17 with biimidazole ligands showed good performance in pH sensing with the protonation-deprotonation mechanism [116]. In a recent study, the biimidazole-coupled phen ligand was employed as the binding site for the development of Ru(II) complex chemosensor (23) for Cu2+ detection by Li and co-workers [121]. The coordination (1:1 bonding ratio) of Cu2+ with complex 23’s biimidazole led to the formation of a stable cyclic structure. The quenched emission of complex 23 showed a linearity with Cu2+ concentration in the range of 0.25–12 μM, and the LoD was 83.3 nM. The application of complex 23 was then demonstrated by Cu2+ detection in tap and lake water samples and imaging in A549 cells. For simple modification of imidazole to pyrazol, Cu2+ “ON–OFF” Ru(II) complex 24 was then reported by **a and colleagues [122]. Different from complex 23, complex 24 coordinated with Cu2+ through a 1:2 stoichiometry. This complex showed higher sensitivity to Cu2+ (LoD = 17.8 nM) compared with complex 23. The test paper was then prepared for Cu2+ detection in river water samples. Interestingly, complex 25 with quinoline substitution showed a 1:1 stoichiometry when binding to Cu2+ in HEPES buffer (LoD = 50.67 nM) [123]. The test paper was also prepared using complex 25 as the chemosensor for Cu2+ detection. In another study, Zhang et al. reported a Ru(II) complex 26 for Cu2+ detection in aqueous solution and imaging in live pea aphids (LoD = 244 nM) [124]. Complex 26 was developed through coordination with two phen ligands and one 2-(2-hydroxyphenyl) imidazo[4,5-f][1,10]phenanthroline. The Cu2+ binding with 2-hydroxyphenyl imidazo through a 1:1 stoichiometry quenched complex 26’s emission. Similar to Cu2+-coordinated complexes 23 and 25, the Cu2+ can be displaced by the addition of EDTA (ethylene diamine tetraacetic acid), which allows the reset of the chemosensors for further detection of Cu2+.
In addition to imidazole, some other groups, such as DPA [86, 125,126,127,128], 1,8-naphthyridine [129], 1,3-benzothiazole [130], carboxyl [131] and others [132], have also been utilized as the response units for the development of Ru(II) complex chemosensors for Cu2+. For example, complex 27 reported by Ramachandran and colleagues was capable of detecting phosphate anions through C–H-anion interaction and Cu2+ through coordination with triazole, benzothiazole and the “O” linker, respectively [130] (Fig. 12). The application of this chemosensor (27) for Cu2+ detection (LoD = 700 nM) and imaging was also demonstrated by luminescence Cu2+ imaging in MCF-7 cells.
Similar to complex 15, the pyridine “linker” of dinuclear Ru(II) complex 28 has also been developed as the chemosensor for Cu2+ detection in H2O/CH3CN (1:1, v/v) [133], in HEPES buffer solution (10 mM, pH 7.4) (Fig. 13A) [134]. Complex 28 showed high luminescence (ϕ = 0.06) at 600 nm, while the Ru(II) complex’s MLCT emission was quenched after coordination of Cu2+ with imidazole and pyridine. This chemosensor showed high sensitivity (LoD = 33.3 nM) and selectivity, reversibility (in the presence of EDTA) and good biocompatibility and cell membrane permeability, enabling it to be used for luminescence imaging. In addition, the Ru(II) complex’s large two-photon absorption (TPA) cross-section enabled complex 28 to be used for TP imaging of Cu2+ in HeLa cells and zebrafish (Fig. 13B).
Molecular structure of Ru(II) complex 28 and its response mechanism to Cu2+ (A). One-photon microscopy (OPM) and two-photon microscopy (TPM) imaging of Cu2+ in cells and zebrafish (B). Adapted with permission from Ref. [134].
Although “OFF–ON” luminescence response chemosensors feature high sensitivity and selectivity, and excellent performance in luminescent bioimaging, the development of turn “ON” response chemosensors remains a challenge due to the intrinsic luminescence quenching property of Cu2+. In 2009, a phenothiazine-coupled Ru(II) complex 29 was developed by Ajayakumar as the luminescence turn “ON” chemosensor for Cu2+ detection (Fig. 14A) [135]. Complex 29 was almost non-luminescent (ϕ = 0.0035 in CH3CN) because of the PeT from electron-rich phenothiazine to the Ru(II) center. In the presence of Cu2+, the oxidation of phenothiazine to phenothiazine-5-oxide inhibited the PeT process, and thus the emission of complex 29 was switched “ON.” In another research, Zhang et al. reported a Ru(II) complex “OFF–ON” luminescence chemosensor 30 for Cu2+ detection and imaging (Fig. 14A) [136]. Complex 30 with an o-(phenylazo)aniline structure showed weak luminescence in HEPES buffer solution (20 mM, pH 7.4). Cu2+-mediated oxidative cyclization led to > 80-fold enhancement in luminescence at 599 nm. The large enhancement in luminescence allowed complex 30 for highly sensitive (LoD = 4.42 nM) and selective detection of Cu2+ in buffer and imaging of Cu2+ in pea aphids (Fig. 14B).
Molecular structure of Ru(II) complexes 29, 30 and their response reaction with Cu2+ (A). The application of Ru(II) complex 30 for Cu2+ imaging in pea aphids. Adapted with permission from Ref. [136].
Despite the quenching of most Ru(II) complexes’ emission by Cu2+ binding, the heterobimetallic Ru(II)–Cu(II) complexes provided an excellent platform for further development of “OFF–ON” response chemosensors for the detection of various analytes, such as anions [137], adenosine triphosphate (ATP) [138], amino acids [139, 140], redox biology [128] and other metal ions [141]. For example, in 2012, Wang et al. reported a Ru(II) complex for sequential detection of Cu2+ and Cr3+ in aqueous solution [141]. The coordination of a complex with Cu2+ produced the non-luminescent Ru(II)–Cu(II) complex, and this heterobimetallic complex showed high selectivity to Cr3+ in NaOAc-HOAc buffer (pH 5.6). A high sensitivity of this chemosensor to Cr3+ was also obtained with a LoD 66 nM.
4.2 Ru(II) Complex Chemosensors for Hg2+
Because of the high binding affinity of S atom with Hg2+, a series of S atom-bearing Ru(II) complexes have been developed as the chemosensors for Hg2+ detection. Previous research has revealed the colorimetric response of N-719 to Hg2+, in which the absorption spectra of N-719 were blue-shifted after binding of Hg2+ [142, 143]. The N-719 functionalized upconversion nanoparticles (UCNPs) were then prepared, and the application of the ratiometric upconversion luminescence (UCL) nanosensor for Hg2+ detection and imaging was also demonstrated [142]. In 2015, the N-719 derivative Ru(II) complex 31 was prepared by Fan and co-workers for colorimetric and luminescent determination of Hg2+ [144]. Similar to N-719, the response of complex 31 to Hg2+ was ascribed to the binding of electron-deficient Hg2+ to the electron-rich sulfur atom of NCS (thiocyante) groups. A 40-nm blue shift (from 525 to 485 nm) of absorption and a remarkable increase of luminescence at 720 nm were observed upon binding of complex 31 to Hg2+. In another study, Li et al. reported that a cyclometallated Ru(II) complex functionalized UCNPs for Hg2+ detection in water [145]. By modifying the cyclometallated Ru(II) complex with a propylsulfonate-coupled hemi-cyanine, Ru(II) complex 32 was then produced as the chemosensor for Hg2+ colorimetric analysis [194] and environmental samples [173]. Recent research found that the Ru(II) complexes’ 4,5-diamino-1,10-phenanthroline ligand can respond to RCS [200], particularly the MGO to form 2-methylpyrazino-1,10-phenanthroline ligand coordinated products [201]. Based on this reaction, Zhang et al. investigated the capability of the Ru(II) complex chemosensor for MGO determination and imaging in RAW 264.7 macrophages and flea. Recently, Zhang et al. reported a “dual-key-and-lock” Ru(II) complex chemosensor 49 for lysosomal FA determination in cancer cells and tumors (Fig. 23A) [202]. Complex 49 was designed by coupling of Ru(II) complex with a DNP quencher through an FA-responsive linker. Interestingly, the response reaction of complex 49 can only take place in the presence of FA (first “key”) under acidic conditions (second “key”), which allow FA detection specifically in lysosomes. Complex 49 has a long lifetime (τ = 330.4 ns), which facilitates the application of background-free TGL analysis in human serum samples and mouse organs. Luminescence imaging results clearly showed that complex 49 could be used for lysosomal FA detection in HeLa cells (Fig. 23B). With this FA-responsive Ru(II) complex, in vivo and ex vivo imaging results confirmed the much higher FA levels in tumor cells and tissues (Fig. 23C).
Molecular structure of Ru(II) complex 49 and its response reaction with FA (A). The luminescence imaging of intracellular FA in lysosomes (B) and ex vivo imaging of FA in different mouse organs (C). Adapted with permission from Ref. [202].
5.4 Ru(II) Complex Chemosensors for RSS
Hydrogen sulfide (H2S) is one of the major RSS in the human body and is involved in various biological processes. This endogenous gaseous molecule is produced by CBS (cystathionine β-synthase) and CSE (cystathionine γ-lyase) catalyzed reaction with thiol-containing biomolecules [203]. Recent research has also revealed that the H2S is a gasotransmitter and a regulator of critical biological processes [204, 205]. The metabolites of H2S, such as polysulfides and persulfides, are also important RSS that may have similar or divergent regulatory roles in living systems [206, 222]. In 2017, Gao et al. reported on Ru(II) complex 53 for luminescence detection of biothiols (Fig. 25) [223]. In this Ru(II) complex, two DNP quenchers were linked to two bpy ligands through a sulfonate ester bond, which enabled the quenching of MLCT emission and the “OFF–ON” response to biothiols. A morpholine moiety was conjugated to the third bpy ligand, allowing complex 53 with lysosome targeting ability. The capability of complex 53 for background-free TGL detection of biothiols was also demonstrated.
Although a number of biothiol-sensitive chemosensors have been reported, the measurement of total biothiols and determination the level of each one remains a challenge. In 2020, Liu et al. reported a “Two Birds with One Stone” Ru(II) complex 54 for the detection and discrimination of biothiols in vitro and in vivo (Fig. 26A) [224]. Complex 54 was developed through coupling of two different signaling units (Ru(II) complex and NBD) through a “luminophore-responsive linker-luminophore” approach. In the presence of GSH, the cleavage of “O” ether bond led to the formation of a luminescent Ru(II) complex and non-fluorescent NBD-SR1. In contrast, the reaction of complex 54 with Cys and Hcy led to the formation of a red-emitting Ru(II) complex and NBD-SR2 that can further undergo a five- or six-member cyclic intermediate-associated rearrangement to form corresponding green-emitting NBD-NR. This allowed for discrimination of GSH from Cys and Hcy under steady-state luminescence measurements. Moreover, under the TGL measurement model, the total biothiol concentration was obtained as elimination of the emission from NBD-NR. The GSH and Cys/Hcy concentrations were thus determined by measuring the same sample with both steady-state and TGL models. The time-gated luminescence imaging of intracellular biothiols was then demonstrated, showing that the NBD emission was eliminated after a 4-ns delay (τNBD-NR = 0.8 ns) (Fig. 26B).
Strategy for the development of Ru(II) complex 54 for biothiol detection and discrimination (A). Luminescence and TGL imaging (a, 0–12 ns; b, 0–4 ns; c, 4–12 ns) of HeLa cells incubated with complex 54 (B). Adapted with permission from Ref. [224].
6.2 Ru(II) Complex Chemosensors for Other Amino Acids
Ru(II) complexes have also been developed for the detection of other amino acids, such as methionine (Met) and histidine (His), through the response mechanism of amino acid-dominated binding of metal ions (e.g., Cu2+ and Ni2+) [56]. Therefore, development of the Ru(II) complex chemosensors with minimal quenching from the surrounding environments, particularly the levels of oxygen, is also demanded. With intense background autofluorescence in biological systems limiting the use of other probes, recent research has confirmed that indeed Ru(II) complexes with prolonged emission lifetime can be used for TGL bioassays and imaging [28, 31, 202]. Such a background-free bioassay and imaging approach allow the determination of target analytes in living cells with higher sensitivity and signal-to-noise (S/N) ratio, which can be further investigated for biomolecule detection in vivo in future studies.
In summary, taking together with the unique photo-physical/-chemical properties of Ru(II) complexes and the potential applications of chemosensors, ongoing research is expected to develop robust Ru(II) complex chemosensors for the determination and imaging of ions and biomolecules in the future. We hope that this review will provide a knowledge base for the Ru(II) complex chemosensor area and inspire the readers to contribute to this promising research field in the future.
Abbreviations
- Ru(II):
-
Ruthenium(II)
- HPLC:
-
High-performance liquid chromatography
- ICP-OES/MS:
-
Inductively coupled plasma-optical emission spectroscopy/mass spectrometry
- bpy:
-
2,2′-Bipyridine
- CT:
-
Computerized tomography
- MRI:
-
Magnetic resonance imaging
- PET:
-
Positron emission tomography
- CA:
-
Contrast agent
- LoD:
-
Limit of detection
- ϕ:
-
Quantum yields
- TGL:
-
Time-gated luminescence
- Ir(III):
-
Iridium(III)
- Pt(II):
-
Platinum(II)
- Au(I):
-
Gold(I)
- Re(I):
-
Rhenium(I)
- Os(II):
-
Osmium(II)
- MLCT:
-
Metal-to-ligand charge transfer
- ILCT:
-
Intraligand charge transfer
- LLCT:
-
Ligand-to-ligand charge transfer
- MMLCT:
-
Metal–metal-to-ligand charge transfer
- LMCT:
-
Ligand-to-metal charge transfer
- MLLCT:
-
Metal-to-ligand-ligand charge transfer
- LMMCT:
-
Ligand-to-metal–metal charge transfer
- MC:
-
Metal centered
- LC:
-
Ligand centered
- FRET:
-
Förster resonance energy transfer
- PeT:
-
Photo-induced electron transfer
- dppz:
-
Dipyridophenazine
- ROS:
-
Reactive oxygen species
- RNS:
-
Reactive nitrogen species
- RCS:
-
Reactive carbonyl species
- RSS:
-
Reactive sulfur species
- F− :
-
Fluoride
- CH3COO− :
-
Acetate
- CN− :
-
Cyanide
- H2PO4 − :
-
Phosphate
- Cl− :
-
Chloride
- Br− :
-
Bromide
- CH3CN:
-
Acetonitrile
- DMSO:
-
Dimethyl sulfoxide
- SCN− :
-
Thiocyanate
- Cys:
-
Cysteine
- Hcy:
-
Homocysteine
- GSH:
-
Glutathione
- HSO3 − :
-
Hydrogen sulfite
- S2 − :
-
Sulfide
- PPi:
-
Pyrophosphate
- DPA:
-
Di(2-picolyl)amine
- ATP:
-
Adenosine triphosphate
- UCNP:
-
Upconversion nanoparticle
- UCL:
-
Upconversion luminescence
- NO:
-
Nitric oxide
- ONOO− :
-
Peroxynitrite
- HNO:
-
Nitroxyl
- O2 • − :
-
Superoxide
- Cy5:
-
Cyanine 5
- •OH:
-
Hydroxyl radicals
- H2O2 :
-
Hydrogen peroxide
- 1O2 :
-
Singlet oxygen
- HOCl:
-
Hypochlorous acid
- DNP:
-
2,4-Dinitrophenyl
- BFO:
-
Benzofurazan-1-oxide
- GO:
-
Glyoxal
- CO:
-
Carbon monoxide
- MGO:
-
Methylglyoxal
- FA:
-
Formaldehyde
- CBS:
-
Cystathionine β-synthase
- CSE:
-
Cystathionine γ-lyase
- H2S:
-
Hydrogen sulfide
- NBD:
-
7-Nitro-2,1,3-benzoxadiazoles
- His:
-
Histidine
- S/N:
-
Signal-to-noise
References
Schreiber Stuart L, Kotz Joanne D, Li M, Aubé J, Austin Christopher P, Reed John C, Rosen H, White EL, Sklar Larry A, Lindsley Craig W, Alexander Benjamin R, Bittker Joshua A, Clemons Paul A, de Souza A, Foley Michael A, Palmer M, Shamji Alykhan F, Wawer Mathias J, McManus O, Wu M, Zou B, Yu H, Golden Jennifer E, Schoenen Frank J, Simeonov A, Jadhav A, Jackson Michael R, Pinkerton Anthony B, Chung Thomas DY, Griffin Patrick R, Cravatt Benjamin F, Hodder Peter S, Roush William R, Roberts E, Chung D-H, Jonsson Colleen B, Noah James W, Severson William E, Ananthan S, Edwards B, Oprea Tudor I, Conn PJ, Hopkins Corey R, Wood Michael R, Stauffer Shaun R, Emmitte Kyle A, Brady Linda S, Driscoll J, Li Ingrid Y, Loomis Carson R, Margolis Ronald N, Michelotti E, Perry Mary E, Pillai A, Yao Y (2015) Advancing biological understanding and therapeutics discovery with small-molecule probes. Cell 161:1252–1265
Park S-H, Kwon N, Lee J-H, Yoon J, Shin I (2020) Synthetic ratiometric fluorescent probes for detection of ions. Chem Soc Rev 49:143–179
Singh H, Tiwari K, Tiwari R, Pramanik SK, Das A (2019) Small molecule as fluorescent probes for monitoring intracellular enzymatic transformations. Chem Rev 119:11718–11760
Jiang X, Wang L, Carroll SL, Chen J, Wang MC, Wang J (2018) Challenges and opportunities for small-molecule fluorescent probes in redox biology applications. Antioxid Redox Signal 29:518–540
Ma S, Chen G, Xu J, Liu Y, Li G, Chen T, Li Y, James TD (2021) Current strategies for the development of fluorescence-based molecular probes for visualizing the enzymes and proteins associated with Alzheimer’s disease. Coord Chem Rev 427:213553
Rahimi F, Chatzimichail S, Saifuddin A, Surman AJ, Taylor-Robinson SD, Salehi-Reyhani A (2020) A review of portable high-performance liquid chromatography: the future of the field? Chromatographia 83:1165–1195
Balcaen L, Bolea-Fernandez E, Resano M, Vanhaecke F (2015) Inductively coupled plasma—Tandem mass spectrometry (ICP-MS/MS): a powerful and universal tool for the interference-free determination of (ultra)trace elements—a tutorial review. Anal Chim Acta 894:7–19
Theiner S, Loehr K, Koellensperger G, Mueller L, Jakubowski N (2020) Single-cell analysis by use of ICP-MS. J Anal At Spectrom 35:1784–1813
Park SY, Yoon SA, Cha Y, Lee MH (2021) Recent advances in fluorescent probes for cellular antioxidants: detection of NADH, hNQO1, H2S, and other redox biomolecules. Coord Chem Rev 428:213613
Withers PJ, Bouman C, Carmignato S, Cnudde V, Grimaldi D, Hagen CK, Maire E, Manley M, Du Plessis A, Stock SR (2021) X-ray computed tomography. Nat Rev Methods Primers 1:18
Avasthi A, Caro C, Pozo-Torres E, Leal MP, García-Martín ML (2020) Magnetic nanoparticles as MRI contrast agents. Top Curr Chem 378:40
Vaquero JJ, Kinahan P (2015) Positron emission tomography: current challenges and opportunities for technological advances in clinical and preclinical imaging systems. Annu Rev Biomed Eng 17:385–414
Willmann JK, van Bruggen N, Dinkelborg LM, Gambhir SS (2008) Molecular imaging in drug development. Nat Rev Drug Discov 7:591–607
Ametamey SM, Honer M, Schubiger PA (2008) Molecular imaging with PET. Chem Rev 108:1501–1516
Meng Q, Wu M, Shang Z, Zhang Z, Zhang R (2022) Responsive gadolinium(III) complex-based small molecule magnetic resonance imaging probes: design, mechanism and application. Coord Chem Rev 457:214398
Kobayashi H, Ogawa M, Alford R, Choyke PL, Urano Y (2010) New strategies for fluorescent probe design in medical diagnostic imaging. Chem Rev 110:2620–2640
Feng H, Meng Q, Ta HT, Zhang R (2020) Development of “dual-key-and-lock” responsive probes for biosensing and imaging. New J Chem 44:12890–12896
Wu L, Huang J, Pu K, James TD (2021) Dual-locked spectroscopic probes for sensing and therapy. Nat Rev Chem 5:406–421
Fernández-Suárez M, Ting AY (2008) Fluorescent probes for super-resolution imaging in living cells. Nat Rev Mol Cell Biol 9:929–943
Liu C, Gao X, Yuan J, Zhang R (2020) Advances in the development of fluorescence probes for cell plasma membrane imaging. Trends Analyt Chem 133:116092
Zhou X, Lee S, Xu Z, Yoon J (2015) Recent progress on the development of chemosensors for gases. Chem Rev 115:7944–8000
Wongkongkatep J, Ojida A, Hamachi I (2017) Fluorescence sensing of inorganic phosphate and pyrophosphate using small molecular sensors and their applications. Top Curr Chem 375:30
Gori M, Thakur A, Sharma A, Flora SJS (2021) Organic-molecule-based fluorescent chemosensor for nerve agents and organophosphorus pesticides. Top Curr Chem 379:33
Feng H, Liu J, Qaitoon A, Meng Q, Sultanbawa Y, Zhang Z, Xu ZP, Zhang R (2021) Responsive small-molecule luminescence probes for sulfite/bisulfite detection in food samples. Trends Anal Chem 136:116199
Zhang R, Song B, Yuan J (2018) Bioanalytical methods for hypochlorous acid detection: recent advances and challenges. Trends Anal Chem 99:1–33
Sedgwick AC, Brewster JT, Wu T, Feng X, Bull SD, Qian X, Sessler JL, James TD, Anslyn EV, Sun X (2021) Indicator displacement assays (IDAs): the past, present and future. Chem Soc Rev 50:9–38
Wu L, Sedgwick AC, Sun X, Bull SD, He X-P, James TD (2019) Reaction-based fluorescent probes for the detection and imaging of reactive oxygen, nitrogen, and sulfur species. Acc Chem Res 52:2582–2597
Zhang KY, Yu Q, Wei H, Liu S, Zhao Q, Huang W (2018) Long-Lived Emissive Probes for Time-Resolved Photoluminescence Bioimaging and Biosensing. Chem Rev 118:1770–1839
Aulsebrook ML, Graham B, Grace MR, Tuck KL (2018) Lanthanide complexes for luminescence-based sensing of low molecular weight analytes. Coord Chem Rev 375:191–220
Heffern MC, Matosziuk LM, Meade TJ (2014) Lanthanide probes for bioresponsive imaging. Chem Rev 114:4496–4539
Zhang R, Yuan J (2020) Responsive metal complex probes for time-gated luminescence biosensing and imaging. Acc Chem Res 53:1316–1329
Connell TU, Donnelly PS (2018) Labelling proteins and peptides with phosphorescent d6 transition metal complexes. Coord Chem Rev 375:267–284
Caporale C, Massi M (2018) Cyclometalated iridium(III) complexes for life science. Coord Chem Rev 363:71–91
Wing-Wah Yam V, Kam-Wing Lo K, Kit-Mai Fung W, Wang C-R (1998) Design of luminescent polynuclear copper(I) and silver(I) complexes with chalcogenides and acetylides as the bridging ligands. Coord Chem Rev 171:17–41
Lo KK-W (2015) Luminescent rhenium(I) and iridium(III) polypyridine complexes as biological probes, imaging reagents, and photocytotoxic agents. Acc Chem Res 48:2985–2995
Lo KK-W, Tsang KH-K, Sze K-S, Chung C-K, Lee TK-M, Zhang KY, Hui W-K, Li C-K, Lau JS-Y, Ng DC-M, Zhu N (2007) Non-covalent binding of luminescent transition metal polypyridine complexes to avidin, indole-binding proteins and estrogen receptors. Coord Chem Rev 251:2292–2310
Lo KK-W, Louie M-W, Zhang KY (2010) Design of luminescent iridium(III) and rhenium(I) polypyridine complexes as in vitro and in vivo ion, molecular and biological probes. Coord Chem Rev 254:2603–2622
Coogan MP, Fernández-Moreira V (2014) Progress with, and prospects for, metal complexes in cell imaging. Chem Commun 50:384–399
Liu Z, He W, Guo Z (2013) Metal coordination in photoluminescent sensing. Chem Soc Rev 42:1568–1600
Zhao Q, Huang C, Li F (2011) Phosphorescent heavy-metal complexes for bioimaging. Chem Soc Rev 40:2508–2524
Campagna S, Puntoriero F, Nastasi F, Bergamini G, Balzani V (2007) In: Balzani V, Campagna S (eds) Photochemistry and photophysics of coordination compounds I. Springer Berlin Heidelberg, Berlin, Heidelberg, pp 117–214
Ma D-L, Ma VP-Y, Chan DS-H, Leung K-H, He H-Z, Leung C-H (2012) Recent advances in luminescent heavy metal complexes for sensing. Coord Chem Rev 256:3087–3113
Zhao Q, Li F, Huang C (2010) Phosphorescent chemosensors based on heavy-metal complexes. Chem Soc Rev 39:3007–3030
Singh A, Barman P (2021) Recent advances in schiff base ruthenium metal complexes: synthesis and applications. Top Curr Chem 379:29
Balzani V, Ceroni P, Credi A, Venturi M (2021) Ruthenium tris(bipyridine) complexes: Interchange between photons and electrons in molecular-scale devices and machines. Coord Chem Rev 433:213758
McClenaghan ND, Leydet Y, Maubert B, Indelli MT, Campagna S (2005) Excited-state equilibration: a process leading to long-lived metal-to-ligand charge transfer luminescence in supramolecular systems. Coord Chem Rev 249:1336–1350
Lo KK-W (2020) Molecular design of bioorthogonal probes and imaging reagents derived from photofunctional transition metal complexes. Acc Chem Res 53:32–44
Demas JN, DeGraff BA (2001) Applications of luminescent transition platinum group metal complexes to sensor technology and molecular probes. Coord Chem Rev 211:317–351
Keefe MH, Benkstein KD, Hupp JT (2000) Luminescent sensor molecules based on coordinated metals: a review of recent developments. Coord Chem Rev 205:201–228
Tyson DS, Castellano FN (1999) Intramolecular singlet and triplet energy transfer in a ruthenium(II) diimine complex containing multiple pyrenyl chromophores. J Phys Chem A 103:10955–10960
Li M-J, Wong KM-C, Yi C, Yam VW-W (2012) New ruthenium(II) complexes functionalized with coumarin derivatives: synthesis, energy-transfer-based sensing of esterase, cytotoxicity, and imaging studies. Chem Eur J 18:8724–8730
Yang D, Guan S, Niu Y, **e Z, Zhou S, Qu X (2018) Construction of a hypoxia responsive upconversion nanosensor for tumor imaging by fluorescence resonance energy transfer from carbon dots to ruthenium complex. J Mater Chem B 6:2315–2322
Zhang R, Liang L, Meng Q, Zhao J, Ta HT, Li L, Zhang Z, Sultanbawa Y, Xu ZP (2019) Responsive upconversion nanoprobe for background-free hypochlorous acid detection and bioimaging. Small 15:1803712
Yip AM-H, Lo KK-W (2018) Luminescent rhenium(I), ruthenium(II), and iridium(III) polypyridine complexes containing a poly(ethylene glycol) pendant or bioorthogonal reaction group as biological probes and photocytotoxic agents. Coord Chem Rev 361:138–163
Lo KK-W, Choi AW-T, Law WH-T (2012) Applications of luminescent inorganic and organometallic transition metal complexes as biomolecular and cellular probes. Dalton Trans 41:6021–6047
Ji S, Wu W, Wu W, Song P, Han K, Wang Z, Liu S, Guo H, Zhao J (2010) Tuning the luminescence lifetimes of ruthenium(II) polypyridine complexes and its application in luminescent oxygen sensing. J Mater Chem 20:1953–1963
Payne SJ, Fiore GL, Fraser CL, Demas JN (2010) Luminescence oxygen sensor based on a ruthenium(II) star polymer complex. Anal Chem 82:917–921
Zhou C, Zhao W-x, You F-t, Geng Z-x, Peng H-s (2019) Highly stable and luminescent oxygen nanosensor based on ruthenium-containing metallopolymer for real-time imaging of intracellular oxygenation. ACS Sens 4:984–991
Ghosh A, Das P, Gill MR, Kar P, Walker MG, Thomas JA, Das A (2011) Photoactive RuII–polypyridyl complexes that display sequence selectivity and high-affinity binding to duplex DNA through groove binding. Chem Eur J 17:2089–2098
Nonat AM, Quinn SJ, Gunnlaugsson T (2009) Mixed f−d coordination complexes as dual visible- and near-infrared-emitting probes for targeting DNA. Inorg Chem 48:4646–4648
Olson EJC, Hu D, Hörmann A, Jonkman AM, Arkin MR, Stemp EDA, Barton JK, Barbara PF (1997) First observation of the key intermediate in the “light-switch” mechanism of [Ru(phen)2d ppz]2+. J Am Chem Soc 119:11458–11467
Gill MR, Garcia-Lara J, Foster SJ, Smythe C, Battaglia G, Thomas JA (2009) A ruthenium(II) polypyridyl complex for direct imaging of DNA structure in living cells. Nat Chem 1:662–667
Baggaley E, Gill MR, Green NH, Turton D, Sazanovich IV, Botchway SW, Smythe C, Haycock JW, Weinstein JA, Thomas JA (2014) Dinuclear ruthenium(II) complexes as two-photon, time-resolved emission microscopy probes for cellular DNA. Angew Chem Int Ed 53:3367–3371
Saadallah D, Bellakhal M, Amor S, Lefebvre J-F, Chavarot-Kerlidou M, Baussanne I, Moucheron C, Demeunynck M, Monchaud D (2017) Selective luminescent labeling of DNA and RNA quadruplexes by π-extended ruthenium light-up probes. Chem Eur J 23:4967–4972
Sheet SK, Sen B, Thounaojam R, Aguan K, Khatua S (2017) Ruthenium(II) complex-based luminescent bifunctional probe for Ag+ and phosphate ions: Ag+-assisted detection and imaging of rRNA. Inorg Chem 56:1249–1263
Wragg A, Gill MR, Turton D, Adams H, Roseveare TM, Smythe C, Su X, Thomas JA (2014) Tuning the cellular uptake properties of luminescent heterobimetallic iridium(III)–ruthenium(II) DNA imaging probes. Chem Eur J 20:14004–14011
Boynton AN, Marcélis L, Barton JK (2016) [Ru(Me4phen)2d ppz]2+, a light switch for DNA mismatches. J Am Chem Soc 138:5020–5023
Puckett CA, Barton JK (2008) Mechanism of cellular uptake of a ruthenium polypyridyl complex. Biochemistry 47:11711–11716
Puckett CA, Barton JK (2007) Methods to explore cellular uptake of ruthenium complexes. J Am Chem Soc 129:46–47
Berni E, Gosse I, Badocco D, Pastore P, Sojic N, Pinet S (2009) Differential photoluminescent and electrochemiluminescent detection of anions with a modified ruthenium(II)–bipyridyl complex. Chem Eur J 15:5145–5152
Aletti AB, Gillen DM, Gunnlaugsson T (2018) Luminescent/colorimetric probes and (chemo-) sensors for detecting anions based on transition and lanthanide ion receptor/binding complexes. Coord Chem Rev 354:98–120
Busschaert N, Caltagirone C, Van Rossom W, Gale PA (2015) Applications of supramolecular anion recognition. Chem Rev 115:8038–8155
Jose DA, Kar P, Koley D, Ganguly B, Thiel W, Ghosh HN, Das A (2007) Phenol- and catechol-based ruthenium(II) polypyridyl complexes as colorimetric sensors for fluoride ions. Inorg Chem 46:5576–5584
Mo H-J, Niu Y-L, Zhang M, Qiao Z-P, Ye B-H (2011) Photophysical, electrochemical and anion sensing properties of Ru(ii) bipyridine complexes with 2,2′-biimidazole-like ligand. Dalton Trans 40:8218–8225
Baggi G, Boiocchi M, Ciarrocchi C, Fabbrizzi L (2013) Enhancing the anion affinity of urea-based receptors with a Ru(terpy)22+ chromophore. Inorg Chem 52:5273–5283
Bhaumik C, Saha D, Das S, Baitalik S (2011) Synthesis, structural characterization, photophysical, electrochemical, and anion-sensing studies of luminescent homo- and heteroleptic ruthenium(II) and osmium(II) complexes based on terpyridyl-imidazole ligand. Inorg Chem 50:12586–12600
Das S, Karmakar S, Mardanya S, Baitalik S (2014) Synthesis, structural characterization, and multichannel anion and cation sensing studies of a bifunctional Ru(ii) polypyridyl–imidazole based receptor. Dalton Trans 43:3767–3782
Khatua S, Samanta D, Bats JW, Schmittel M (2012) Rapid and highly sensitive dual-channel detection of cyanide by bis-heteroleptic ruthenium(II) complexes. Inorg Chem 51:7075–7086
Chowdhury B, Sinha S, Ghosh P (2016) Selective sensing of phosphates by a new bis-heteroleptic RuII complex through halogen bonding: a superior sensor over its hydrogen-bonding analogue. Chem Eur J 22:18051–18059
Chowdhury B, Khatua S, Dutta R, Chakraborty S, Ghosh P (2014) Bis-heteroleptic ruthenium(II) complex of a triazole ligand as a selective probe for phosphates. Inorg Chem 53:8061–8070
Hao Y, Yang P, Li S, Huang X, Yang X-J, Wu B (2012) Selective anion sensing by a ruthenium(ii)–bipyridyl-functionalized tripodal tris(urea) receptor. Dalton Trans 41:7689–7694
Gale PA, Caltagirone C (2015) Anion sensing by small molecules and molecular ensembles. Chem Soc Rev 44:4212–4227
Saha D, Das S, Mardanya S, Baitalik S (2012) Structural characterization and spectroelectrochemical, anion sensing and solvent dependence photophysical studies of a bimetallic Ru(ii) complex derived from 1,3-di(1H-imidazo[4,5-f][1,10]phenanthroline-2-yl)benzene. Dalton Trans 41:8886–8898
Sun Y, Hudson ZM, Rao Y, Wang S (2011) Tuning and switching MLCT phosphorescence of [Ru(bpy)3]2+ complexes with triarylboranes and anions. Inorg Chem 50:3373–3378
Zhang W, ** X, Wang Y-L, Du Z, Liu C, Liu J, Song B, Yuan J, Zhang R (2020) Responsive ruthenium complex probe for phosphorescence and time-gated luminescence detection of bisulfite. Dalton Trans 49:5531–5538
Zhang R, Yu X, Yin Y, Ye Z, Wang G, Yuan J (2011) Development of a heterobimetallic Ru(II)–Cu(II) complex for highly selective and sensitive luminescence sensing of sulfide anions. Anal Chim Acta 691:83–88
Ghosh A, Ganguly B, Das A (2007) Urea-based ruthenium(II)−polypyridyl complex as an optical sensor for anions: synthesis, characterization, and binding studies. Inorg Chem 46:9912–9918
Bhaumik C, Das S, Saha D, Dutta S, Baitalik S (2010) Synthesis, characterization, photophysical, and anion-binding studies of luminescent heteroleptic bis-tridentate ruthenium(II) complexes based on 2,6-bis(benzimidazole-2-yl)pyridine and 4′-substituted 2,2′:6′,2′′ Terpyridine derivatives. Inorg Chem 49:5049–5062
Gale PA, Howe ENW, Wu X, Spooner MJ (2018) Anion receptor chemistry: highlights from 2016. Coord Chem Rev 375:333–372
Karmakar S, Maity D, Mardanya S, Baitalik S (2015) Pyrene and imidazole functionalized luminescent bimetallic Ru(ii) terpyridine complexes as efficient optical chemosensors for cyanide in aqueous, organic and solid media. Dalton Trans 44:18607–18623
Hancock LM, Marchi E, Ceroni P, Beer PD (2012) Anion sensing in aqueous media by photo-active transition-metal bipyridyl rotaxanes. Chem Eur J 18:11277–11283
Mondal D, Bar M, Mukherjee S, Baitalik S (2016) Design of Ru(II) complexes based on anthraimidazoledione-functionalized terpyridine ligand for improvement of room-temperature luminescence characteristics and recognition of selective anions: experimental and DFT/TD-DFT study. Inorg Chem 55:9707–9724
Maity D, Vyas G, Bhatt M, Paul P (2015) Detection of NaCN in aqueous media using a calixarene-based fluoroionophore containing ruthenium(II)-bipyridine as the fluorogenic unit. RSC Adv 5:6151–6159
Mardanya S, Karmakar S, Bar M, Baitalik S (2015) Pyrene-biimidazole based Ru(II) and Os(II) complexes as highly efficient probes for the visible and near-infrared detection of cyanide in aqueous media. Dalton Trans 44:21053–21072
Cheng F, He C, Yu S, Yin H (2017) Highly selective SCN—fluorescent sensing by a Ru(II) complex containing functionalized polypyridine. Inorganica Chim Acta 462:43–49
Lin Z-h, Ou S-j, Duan C-y, Zhang B-g, Bai Z-p (2006) Naked-eye detection of fluoride ion in water: a remarkably selective easy-to-prepare test paper. Chem Commun 624–626
Lin Z-h, Zhao Y-g, Duan C-y, Zhang B-g, Bai Z-p (2006) A highly selective chromo- and fluorogenic dual responding fluoride sensor: naked-eye detection of F− ion in natural water via a test paper. Dalton Trans 3678–3684
Su X, Hu R, Li X, Zhu J, Luo F, Niu X, Li M, Zhao Q (2016) Hydrophilic indolium cycloruthenated complex system for visual detection of bisulfite with a large red shift in absorption. Inorg Chem 55:745–754
Chan J, Dodani SC, Chang CJ (2012) Reaction-based small-molecule fluorescent probes for chemoselective bioimaging. Nat Chem 4:973–984
Zhang R, Yu X, Ye Z, Wang G, Zhang W, Yuan J (2010) Turn-on luminescent probe for cysteine/homocysteine based on a ruthenium(II) complex. Inorg Chem 49:7898–7903
Zhang W, Liu H, Zhang F, Wang Y-L, Song B, Zhang R, Yuan J (2018) Development of a ruthenium(II) complex-based luminescence probe for detection of hydrogen sulfite in food samples. Microchem J 141:181–187
Zhu J-W, Ou H-D, Xu N, Deng W, Yao Z-J (2020) Ruthenium-based phosphorescent probe for selective and naked-eye detection of cyanide in aqueous media. Dyes Pigm 176:108196
Li M, Liang Q, Zheng M, Fang C, Peng S, Zhao M (2013) An efficient ruthenium tris(bipyridine)-based luminescent chemosensor for recognition of Cu(ii) and sulfide anion in water. Dalton Trans 42:13509–13515
Zheng Z-B, Huang Q-Y, Han Y-F, Zuo J, Ma Y-N (2017) Ruthenium(II) complex-based chemosensors for highly sensitive and selective sequential recognition of copper ion and cyanide. Sens Actuators B Chem 253:203–212
Zhang S-T, Li P, Kou X, **ao D (2018) Highly selective and sensitive luminescent turn-on probe for pyrophosphate detection in aqueous solution. ChemistrySelect 3:10057–10063
Cheng F, Chen J, Wang F, Tang N, Hou N, Chen G (2012) Synthesis, characterization, and pH-induced luminescence switching of two trinuclear Ru(II) polypyridyl complexes containing imidazole. Transit Met Chem 37:415–421
Yin H, Liu Z, Yu S, Yang Y, Dong J, Yang X, Wang F, He C, Cheng F (2021) A binuclear ruthenium polypyridyl complex: synthesis, characterization, pH luminescence sensor and electrochemical properties. Transit Met Chem 46:49–56
**g B, Wu T, Tian C, Zhang M, Shen T (2000) pH-dependent luminescence of ruthenium(II) polypyridine complexes. Bull Chem Soc Jpn 73:1749–1755
Bai G-Y, Wang K-Z, Duan Z-M, Gao L-H (2004) Luminescent pH sensing and DNA binding properties of a novel ruthenium(II) complex. J Inorg Biochem 98:1017–1022
Yu F, Shen C, Zheng T, Chu W-K, **ang J, Luo Y, Ko C-C, Guo Z-Q, Lau T-C (2016) Acid-base behaviour in the absorption and emission spectra of ruthenium(II) complexes with hydroxy-substituted bipyridine and phenanthroline ligands. Eur J Inorg Chem 2016:3641–3648
Gonçalves HMR, Maule CD, Jorge PAS, Esteves da Silva JCG (2008) Fiber optic lifetime pH sensing based on ruthenium(II) complexes with dicarboxybipyridine. Anal Chim Acta 626:62–70
Zheng Z-B, Kang S-Y, Zhao Y, Zhang N, Yi X, Wang K-Z (2015) pH and copper ion luminescence on/off sensing by a dipyrazinylpyridine-appended ruthenium complex. Sens Actuators B Chem 221:614–624
Zheng Z-B, Wu Y-Q, Wang K-Z, Li F (2014) pH luminescence switching, dihydrogen phosphate sensing, and cellular uptake of a heterobimetallic ruthenium(ii)–rhenium(i) complex. Dalton Trans 43:3273–3284
San Tan S, Yanagisawa S, Inagaki K, Morikawa Y, Kassim MB (2017) Augmented pH-sensitivity absorbance of a ruthenium(II) bis(bipyridine) complex with elongation of the conjugated ligands: an experimental and theoretical investigation. Phys Chem Chem Phys 19:25734–25745
Tormo L, Bustamante N, Colmenarejo G, Orellana G (2010) Can luminescent Ru(II) polypyridyl dyes measure pH directly? Anal Chem 82:5195–5204
Yu H-j, Hao Z-f, Peng H-l, Rao R-h, Sun M, Alana WR, Ran C, Chao H, Yu L (2017) Near-infrared lysosome pH tracker and naked-eye colorimetric nucleic acids sensor based on ruthenium complexes [Ru(bim)2(dppz)]2+ and [Ru(bim)2(pip)]2+. Sens Actuators B Chem 252:313–321
Meng T-T, Wang H, Zheng Z-B, Wang K-Z (2017) pH-switchable “Off–On–Off” near-infrared luminescence based on a dinuclear ruthenium(II) complex. Inorg Chem 56:4775–4779
Liu H-Y, Zhang S-Q, Cui M-C, Gao L-H, Zhao H, Wang K-Z (2020) pH-sensitive near-IR emitting dinuclear ruthenium complex for recognition, two-photon luminescent imaging, and subcellular localization of cancer cells. ACS Appl Bio Mater 3:5420–5427
Carter KP, Young AM, Palmer AE (2014) Fluorescent sensors for measuring metal ions in living systems. Chem Rev 114:4564–4601
Cheng F, Ren M, He C, Yin H (2016) Luminescent chemosensor for Hg2+ ion based on a dinuclear Ru(II) complex containing open chain azacrown ether. Inorg Chim Acta 450:170–175
Li M, Sheth S, Xu Y, Song Q (2020) Ru(II)-bipyridine complex as a highly sensitive luminescent probe for Cu2+ detection and cell imaging. Microchem J 156:104848
**a XL, Zhang DB, Zhang JL, Pu SZ (2020) Highly sensitive ruthenium complex-based fluorescent probe for copper ion detection. Tetrahedron 76:131526
Kumar P, Kumar S (2020) Copper ion luminescence on/off sensing by a quinoline-appended ruthenium(II)-polypyridyl complex in aqueous media. J Mol Struct 1202:127242
Zhang Y, Liu Z, Zhang Y, Xu Y, Li H, Wang C, Lu A, Sun S (2015) A reversible and selective luminescent probe for Cu2+ detection based on a ruthenium(II) complex in aqueous solution. Sens Actuators B Chem 211:449–455
Lin Q-T, Pei L-M, Xu W-C, Chao H, Ji L-N (2012) [Ru(bpy)2(pipdpa)]2+ as a highly sensitive and selective luminescent chemosensor for Cu2+ in aqueous solution. Inorg Chem Commun 16:104–106
Li X, Chen Y, Meng J, Lü K, Zhang A, Zhang B (2011) A new luminescent ruthenium(II) polypyridine-derived dipicolylamine complex as a sensor for Cu2+ ions. Chin J Chem 29:1947–1950
Liu X-W, **ao Y, Zhang S-B, Lu J-L (2017) A selective luminescent sensor for the detection of copper (II) ions based on a ruthenium complex containing DPA moiety. Inorg Chem Commun 84:56–58
Geißer B, Alsfasser R (2003) Probing the aqueous copper(II) coordination chemistry of bifunctional chelating amino acid ligands with a luminescent ruthenium chromophore. Dalton Trans 612–618
He C, Yu S, Ma S, Liu Z, Yao L, Cheng F, Liu P (2019) A novel ruthenium(II) polypyridyl complex bearing 1,8-naphthyridine as a high selectivity and sensitivity fluorescent chemosensor for Cu2+ and Fe3+ ions. Molecules 24
Ramachandran M, Anandan S (2020) Triazole appending ruthenium(II) polypyridine complex for selective sensing of phosphate anions through C–H–anion interaction and copper(II) ions via cancer cells. New J Chem 44:6186–6196
He C-L, Ren F-L, Zhang X-B, Dong Y-Y, Zhao Y (2006) A fluorescent chemosensor for copper(II) based on a carboxylic acid-functionalized tris(2,2′-bipyridine)-ruthenium(II) complex. Anal Sci 22:1547–1551
Comba P, Krämer R, Mokhir A, Naing K, Schatz E (2006) Synthesis of new phenanthroline-based heteroditopic ligands—highly efficient and selective fluorescence sensors for copper(II) ions. Eur J Inorg Chem 2006:4442–4448
Cheng F, Tang N, Miao K, Wang F (2014) A dinuclear ruthenium(II) polypyridyl complex containing a terpy-like fragment for Cu2+ probing. Z Anorg Allg Chem 640:1816–1821
Zhang P, Pei L, Chen Y, Xu W, Lin Q, Wang J, Wu J, Shen Y, Ji L, Chao H (2013) A dinuclear ruthenium(II) complex as a one- and two-photon luminescent probe for biological Cu2+ detection. Chem Eur J 19:15494–15503
Ajayakumar G, Sreenath K, Gopidas KR (2009) Phenothiazine attached Ru(bpy)32+ derivative as highly selective “turn-ON” luminescence chemodosimeter for Cu2+. Dalton Trans 1180–1186
Zhang Y, Liu Z, Yang K, Zhang Y, Xu Y, Li H, Wang C, Lu A, Sun S (2015) A ruthenium(II) complex as turn-on Cu(II) luminescent sensor based on oxidative cyclization mechanism and its application in vivo. Sci Rep 5:8172
Ye Z, An X, Song B, Zhang W, Dai Z, Yuan J (2014) A novel dinuclear ruthenium(ii)–copper(ii) complex-based luminescent probe for hydrogen sulfide. Dalton Trans 43:13055–13060
Padilla-Tosta Miguel E, Lloris José M, Martínez-Máñez R, Pardo T, Sancenón F, Soto J, Marcos MD (2001) ATP recognition through a fluorescence change in a multicomponent dinuclear system containing a Ru(Tpy)22+ fluorescent core and a cyclam−Cu2+ complex. Eur J Inorg Chem 2001:1221–1226
Zhang P, Wang Y, Huang W, Zhao Z, Li H, Wang H, He C, Liu J, Zhang Q (2018) “Turn off-on” phosphorescent sensor for biothiols based on a Ru–Cu ensemble. Sens Actuators B Chem 255:283–289
Zhang S-T, Li P, Liao C, Luo T, Kou X, **ao D (2018) A highly sensitive luminescent probe based on Ru(II)-bipyridine complex for Cu2+, l-Histidine detection and cellular imaging. Spectrochim Acta A 201:161–169
Wang J-N, Qi Q, Zhang L, Li S-H (2012) Turn-On luminescent sensing of metal cations via quencher displacement: rational design of a highly selective chemosensor for chromium(III). Inorg Chem 51:13103–13107
Liu Q, Peng J, Sun L, Li F (2011) High-efficiency upconversion luminescent sensing and bioimaging of Hg(II) by chromophoric ruthenium complex-assembled nanophosphors. ACS Nano 5:8040–8048
Duan C, Liang L, Li L, Zhang R, Xu ZP (2018) Recent progress in upconversion luminescence nanomaterials for biomedical applications. J Mater Chem B 6:192–209
Fan S-H, Shen J, Wu H, Wang K-Z, Zhang A-G (2015) A highly selective turn-on colorimetric and luminescence sensor based on a triphenylamine-appended ruthenium(II) dye for detecting mercury ion. Chin Chem Lett 26:580–584
Li X, Wu Y, Liu Y, Zou X, Yao L, Li F, Feng W (2014) Cyclometallated ruthenium complex-modified upconversion nanophosphors for selective detection of Hg2+ ions in water. Nanoscale 6:1020–1028
Li X, Du K, **e C, Wu Y, Zhang B, Tang D (2020) A highly sensitive and selective colorimetric probe based on a cycloruthenated complex: an Hg2+-promoted switch of thiophene coordination. Dalton Trans 49:2024–2032
Wu Y, Cheng X, **e C, Du K, Li X, Tang D (2020) A polymer membrane tethered with a cycloruthenated complex for colorimetric detection of Hg2+ ions. Spectrochim Acta A 228:117541
Odhiambo RA, Aluoch AO, Njenga LW, Kagwanja SM, Wandiga SO, Wendt OF (2018) Synthesis, characterisation and ion-binding properties of oxathiacrown ethers appended to [Ru(bpy)2]2+. Selectivity towards Hg2+, Cd2+ and Pb2+. RSC Adv 8:3663–3672
Khatua S, Schmittel M (2013) A single molecular light-up sensor for quantification of Hg2+ and Ag+ in aqueous medium: high selectivity toward Hg2+ over Ag+ in a mixture. Org Lett 15:4422–4425
Ru J, Tang X, Ju Z, Zhang G, Dou W, Mi X, Wang C, Liu W (2015) Exploitation and application of a highly sensitive Ru(II) complex-based phosphorescent chemodosimeter for Hg2+ in aqueous solutions and living cells. ACS Appl Mater Interfaces 7:4247–4256
Ru J, Mi X, Guan L, Tang X, Ju Z, Zhang G, Wang C, Liu W (2015) Design and application of a water-soluble phosphorescent Ru(ii) complex as turn-on sensing material for Hg2+. J Mater Chem B 3:6205–6212
Cheng F, Tang N, Yue X (2009) A new family of Ru(II) polypyridyl complexes containing open-chain crown ether for Mg2+ and Ca2+ probing. Spectrochim Acta A 71:1944–1951
He C, Yu S, Ma S, Liu Z, Yao L, Cheng F, Liu P (2019) A novel ruthenium(II) polypyridyl complex bearing 1,8-naphthyridine as a high selectivity and sensitivity fluorescent chemosensor for Cu2+ and Fe3+ ions. Molecules 24:4032
Cheng F, Tang N (2008) A novel family of Ru(II) polypyridyl complexes containing benzocrown ether for Na+ and Li+ probing. Inorg Chem Commun 11:400–403
Li C-Y, Zhang X-B, ** Z, Han R, Shen G-L, Yu R-Q (2006) A fluorescent chemosensor for cobalt ions based on a multi-substituted phenol-ruthenium(II) tris(bipyridine) complex. Anal Chim Acta 580:143–148
Lin H, Cinar ME, Schmittel M (2010) Comparison of ruthenium(II) and cyclometalated iridium(III) azacrown ether phenanthroline hybrids for the detection of metal cations by electrochemiluminescence. Dalton Trans 39:5130–5138
Barigelletti F, Flamigni L, Calogero G, Hammarström L, Sauvage J-P, Collin J-P (1998) A functionalized ruthenium(II)-bis-terpyridine complex as a rod-like luminescent sensor of zinc(II). Chem Commun 2333–2334
Zheng Z-B, Duan Z-M, Ma Y-Y, Wang K-Z (2013) Highly sensitive and selective difunctional ruthenium(II) complex-based chemosensor for dihydrogen phosphate anion and ferrous cation. Inorg Chem 52:2306–2316
Kumar A, Chhatwal M, Singh AK, Singh V, Trivedi M (2014) A fast and selective probe for monitoring Pd2+ in aqueous medium via the dual-optical readout. Chem Commun 50:8488–8490
**e Z, Wen J, Sun S, Zhang J, Deng X, Han S, Wang L, Zhang B, Hong C, Sun S (2021) In-vitro and in-vivo monitoring of gold(III) ions from intermediate metabolite of sodium aurothiomalate through water-soluble ruthenium (II) complex-based luminescent probe. Bioorg Chem 110:104749
Zhang R, Yong J, Yuan J, ** XuZ (2020) Recent advances in the development of responsive probes for selective detection of cysteine. Coord Chem Rev 408:213182
Liu J, Wu M, Zhang R, Xu ZP (2021) Oxygen-derived free radicals: Production, biological importance, bioimaging, and analytical detection with responsive luminescent nanoprobes. View 20200139
Wu Y, Vazquez-Prada KX, Liu Y, Whittaker AK, Zhang R, Ta HT (2021) Recent advances in the development of theranostic nanoparticles for cardiovascular diseases. Nanotheranostics 5:499–514
Winterbourn CC (2008) Reconciling the chemistry and biology of reactive oxygen species. Nat Chem Biol 4:278–286
Kwon N, Kim D, Swamy KMK, Yoon J (2021) Metal-coordinated fluorescent and luminescent probes for reactive oxygen species (ROS) and reactive nitrogen species (RNS). Coord Chem Rev 427:213581
Chen X, Wang F, Hyun JY, Wei T, Qiang J, Ren X, Shin I, Yoon J (2016) Recent progress in the development of fluorescent, luminescent and colorimetric probes for detection of reactive oxygen and nitrogen species. Chem Soc Rev 45:2976–3016
Patel RP, McAndrew J, Sellak H, White CR, Jo H, Freeman BA, Darley-Usmar VM (1999) Biological aspects of reactive nitrogen species. Biochim Biophys Acta Bioenerg 1411:385–400
Vidanapathirana AK, Psaltis PJ, Bursill CA, Abell AD, Nicholls SJ (2021) Cardiovascular bioimaging of nitric oxide: achievements, challenges, and the future. Med Res Rev 41:435–463
Wang S, Chen L, Jangili P, Sharma A, Li W, Hou J-T, Qin C, Yoon J, Kim JS (2018) Design and applications of fluorescent detectors for peroxynitrite. Coord Chem Rev 374:36–54
Zhang R, Ye Z, Wang G, Zhang W, Yuan J (2010) Development of a ruthenium(II) complex based luminescent probe for imaging nitric oxide production in living cells. Chem Eur J 16:6884–6891
Zhang W, Zhao D, Zhang R, Ye Z, Wang G, Yuan J, Yang M (2011) A ruthenium(ii) complex based turn-on electrochemiluminescence probe for the detection of nitric oxide. Analyst 136:1867–1872
Vidanapathirana AK, Pullen BJ, Zhang R, Duong M, Goyne JM, Zhang X, Bonder CS, Abell AD, Bursill CA, Nicholls SJ, Psaltis PJ (2019) A novel ruthenium-based molecular sensor to detect endothelial nitric oxide. Sci Rep 9:1720
Zhang W, Zhang J, Zhang H, Cao L, Zhang R, Ye Z, Yuan J (2013) Development and application of a ruthenium(II) complex-based photoluminescent and electrochemiluminescent dual-signaling probe for nitric oxide. Talanta 116:354–360
Yu X, Zhang R, Ye Z, Song B, Yuan J (2013) Design and synthesis of a ruthenium(II) complex-based luminescent probe for highly selective and sensitive luminescence detection of nitric oxide. J Fluoresc 23:1113–1120
Ghosh A, Das P, Saha S, Banerjee T, Bhatt HB, Das A (2011) Diamine derivative of a ruthenium(II)-polypyridyl complex for chemodosimetric detection of nitrite ion in aqueous solution. Inorg Chim Acta 372:115–119
Ma J, Wu J, Liu W, Wang P, Fan Z (2012) Ruthenium(II) complex-based fluorescent sensor for peroxynitrite. Spectrochim Acta A 94:340–345
Zhang W, Liu Y, Gao Q, Liu C, Song B, Zhang R, Yuan J (2018) A ruthenium(II) complex–cyanine energy transfer scaffold based luminescence probe for ratiometric detection and imaging of mitochondrial peroxynitrite. Chem Commun 54:13698–13701
Jia X, Chen Q, Yang Y, Tang Y, Wang R, Xu Y, Zhu W, Qian X (2016) FRET-based mito-specific fluorescent probe for ratiometric detection and imaging of endogenous peroxynitrite: Dyad of Cy3 and Cy5. J Am Chem Soc 138:10778–10781
Gomes A, Fernandes E, Lima JLFC (2006) Use of fluorescence probes for detection of reactive nitrogen species: a review. J Fluoresc 16:119–139
Wang H, Zhang R, Bridle KR, Jayachandran A, Thomas JA, Zhang W, Yuan J, Xu ZP, Crawford DHG, Liang X, Liu X, Roberts MS (2017) Two-photon dual imaging platform for in vivo monitoring cellular oxidative stress in liver injury. Sci Rep 7:45374
Wilson GJ, Launikonis A, Sasse WHF, Mau AWH (1997) Excited-state processes in ruthenium(II) bipyridine complexes containing covalently bound arenes. J Phys Chem A 101:4860–4866
Tyson DS, Bialecki J, Castellano FN (2000) Ruthenium(II) complex with a notably long excited state lifetime. Chem Commun 2355–2356
Tyson DS, Luman CR, Zhou X, Castellano FN (2001) New Ru(II) chromophores with extended excited-state lifetimes. Inorg Chem 40:4063–4071
Tyson DS, Henbest KB, Bialecki J, Castellano FN (2001) Excited state processes in ruthenium(II)/pyrenyl complexes displaying extended lifetimes. J Phys Chem A 105:8154–8161
Wilson GJ, Sasse WHF, Mau AWH (1996) Singlet and triplet energy transfer processes in ruthenium(II) bipyridine complexes containing covalently bound arenes. Chem Phys Lett 250:583–588
Yin H-J, Liu Y-J, Gao J, Wang K-Z (2017) A highly sensitive and selective visible-light excitable luminescent probe for singlet oxygen based on a dinuclear ruthenium complex. Dalton Trans 46:3325–3331
Ye Z, Song B, Yin Y, Zhang R, Yuan J (2013) Development of singlet oxygen-responsive phosphorescent ruthenium(II) complexes. Dalton Trans 42:14380–14383
Zhang R, Ye Z, Song B, Dai Z, An X, Yuan J (2013) Development of a ruthenium(II) complex-based luminescent probe for hypochlorous acid in living cells. Inorg Chem 52:10325–10331
Zhang J, Zhang D, **ao L, Pu S (2021) Development of an ultrasensitive Ru(II) complex-based fluorescent probe with phenothiazine unit for selective detection HOCl and its application in water samples. Dyes Pigm 188:109179
Yu X, Zhang W, Ye Z, Song B, Yuan J (2015) Development of a functional ruthenium(II) complex that can act as a photoluminescent and electrochemiluminescent dual-signaling probe for hypochlorous acid. J Fluoresc 25:997–1004
Cao L, Zhang R, Zhang W, Du Z, Liu C, Ye Z, Song B, Yuan J (2015) A ruthenium(II) complex-based lysosome-targetable multisignal chemosensor for in vivo detection of hypochlorous acid. Biomaterials 68:21–31
Sen B, Sheet SK, Patra SK, Koner D, Saha N, Khatua S (2019) Highly selective detection of hypochlorous acid by a bis-heteroleptic Ru(II) complex of pyridyl-1,2,3-triazole ligand via C(sp2)–H hydroxylation. Inorg Chem 58:9982–9991
Li M, Wang Y, Yang Y, Gao Y, Zhao M, Zheng M, Peng S (2015) Oximated ruthenium tris-bipyridyl complex: synthesis and luminescent response specifically for ClO− in water containing multiple ions. Dalton Trans 44:14071–14076
Liu F, Gao Y, Wang J, Sun S (2014) Reversible and selective luminescent determination of ClO−/H2S redox cycle in vitro and in vivo based on a ruthenium trisbipyridyl probe. Analyst 139:3324–3329
Zhang R, Song B, Dai Z, Ye Z, **ao Y, Liu Y, Yuan J (2013) Highly sensitive and selective phosphorescent chemosensors for hypochlorous acid based on ruthenium(II) complexes. Biosens Bioelectron 50:1–7
Ye Z, Zhang R, Song B, Dai Z, ** D, Goldys EM, Yuan J (2014) Development of a functional ruthenium(II) complex for probing hypochlorous acid in living cells. Dalton Trans 43:8414–8420
Shi W, Song B, Liu Z, Zhang W, Tan M, Song F, Yuan J (2020) Smart bimodal imaging of hypochlorous acid in vivo using a heterobimetallic ruthenium(II)–gadolinium(III) complex probe. Anal Chem 92:11145–11154
Liu Z, Gao K, Wang B, Yan H, **ng P, Zhong C, Xu Y, Li H, Chen J, Wang W, Sun S (2016) A dinuclear ruthenium(II) complex as turn-on luminescent probe for hypochlorous acid and its application for in vivo imaging. Sci Rep 6:29065
Ohata J, Bruemmer KJ, Chang CJ (2019) Activity-based sensing methods for monitoring the reactive carbon species carbon monoxide and formaldehyde in living systems. Acc Chem Res 52:2841–2848
Kele P, Li X, Duerkop A (2013) New luminescent ruthenium probes for detection of diacetyl. Microchem J 108:156–160
Zhang W, Zhang F, Wang Y-L, Song B, Zhang R, Yuan J (2017) Red-emitting ruthenium(II) and iridium(III) complexes as phosphorescent probes for methylglyoxal in vitro and in vivo. Inorg Chem 56:1309–1318
Liu C, Zhang R, Zhang W, Liu J, Wang Y-L, Du Z, Song B, Xu ZP, Yuan J (2019) “Dual-key-and-lock” ruthenium complex probe for lysosomal formaldehyde in cancer cells and tumors. J Am Chem Soc 141:8462–8472
Kolluru GK, Shen X, Kevil CG (2020) Reactive sulfur species. Arterioscler Thromb Vasc Biol 40:874–884
Gruhlke MCH, Slusarenko AJ (2012) The biology of reactive sulfur species (RSS). Plant Physiol Biochem 59:98–107
Pluth MD, Zhao Y, Cerda MM (2020) In: Chenoweth DM (ed) methods in enzymology. Academic Press, pp 149–164
Yuan S, Shen X, Kevil CG (2017) Beyond a gasotransmitter: hydrogen sulfide and polysulfide in cardiovascular health and immune response. Antioxid Redox Signal 27:634–653
Jiang C, Huang H, Kang X, Yang L, ** Z, Sun H, Pluth MD, Yi L (2021) NBD-based synthetic probes for sensing small molecules and proteins: design, sensing mechanisms and biological applications. Chem Soc Rev 50:7436–7495
Liu C, Liu J, Zhang W, Wang Y-L, Gao X, Song B, Yuan J, Zhang R (2021) A Ruthenium(II) complex-based probe for colorimetric and luminescent detection and imaging of hydrogen sulfide in living cells and organisms. Anal Chim Acta 1145:114–123
Du Z, Song B, Zhang W, Duan C, Wang Y-L, Liu C, Zhang R, Yuan J (2018) Quantitative monitoring and visualization of hydrogen sulfide in vivo using a luminescent probe based on a ruthenium(II) complex. Angew Chem Int Ed 57:3999–4004
Cheng Z, Kuru E, Sachdeva A, Vendrell M (2020) Fluorescent amino acids as versatile building blocks for chemical biology. Nat Rev Chem 4:275–290
Niu L-Y, Chen Y-Z, Zheng H-R, Wu L-Z, Tung C-H, Yang Q-Z (2015) Design strategies of fluorescent probes for selective detection among biothiols. Chem Soc Rev 44:6143–6160
Zheng Z-B, Cui J-C, Han Y-F, Ge Y-Q, Zuo J, Hao W-X (2019) Development of fast-response turn-on phosphorescent probes for biothiols based on ruthenium(II) complexes. Anal Methods 11:2341–2350
Zheng Z-B, Han Y-F, Ge Y-Q, Cui J-C, Zuo J, Nie K (2019) Rapid and selective detection of biothiols by novel ruthenium(II) complex-based phosphorescence probes. Spectrochim Acta A 216:328–334
Zhang W, Zhang R, Zhang J, Ye Z, ** D, Yuan J (2012) Photoluminescent and electrochemiluminescent dual-signaling probe for bio-thiols based on a ruthenium(II) complex. Anal Chim Acta 740:80–87
Li M-J, Zhan C-Q, Nie M-J, Chen G-N, Chen X (2011) Selective recognition of homocysteine and cysteine based on new ruthenium(II) complexes. J Inorg Biochem 105:420–425
Chen H, Li X, Wu Y, Gao W, Bai R (2012) A ruthenium(II) complex with environment-responsive dual emission and its application in the detection of cysteine/homocysteine. Dalton Trans 41:13292–13297
Ye Z, Gao Q, An X, Song B, Yuan J (2015) A functional ruthenium(II) complex for imaging biothiols in living bodies. Dalton Trans 44:8278–8283
Li G-Y, Liu J-P, Huang H-Y, Wen Y, Chao H, Ji L-N (2013) Colorimetric and luminescent dual-signaling responsive probing of thiols by a ruthenium(II)-azo complex. J Inorg Biochem 121:108–113
Li Y, Shi N, Li M (2019) An efficient ruthenium(II) tris(bipyridyl)-based chemosensor for the specific detection of cysteine and its luminescence imaging in living zebrafish. New J Chem 43:18517–18524
Chow C-F, Chiu BKW, Lam MHW, Wong W-Y (2003) A trinuclear heterobimetallic Ru(II)/Pt(II) complex as a chemodosimeter selective for sulfhydryl-containing amino acids and peptides. J Am Chem Soc 125:7802–7803
Ji S, Guo H, Yuan X, Li X, Ding H, Gao P, Zhao C, Wu W, Wu W, Zhao J (2010) A highly selective OFF-ON red-emitting phosphorescent thiol probe with large stokes shift and long luminescent lifetime. Org Lett 12:2876–2879
Zhang R, Ye Z, Yin Y, Wang G, ** D, Yuan J, Piper JA (2012) Develo** red-emissive ruthenium(II) complex-based luminescent probes for cellular imaging. Bioconjug Chem 23:725–733
Gao Q, Zhang W, Song B, Zhang R, Guo W, Yuan J (2017) Development of a novel lysosome-targeted ruthenium(II) complex for phosphorescence/time-gated luminescence assay of biothiols. Anal Chem 89:4517–4524
Liu C, Liu J, Zhang W, Wang Y-L, Liu Q, Song B, Yuan J, Zhang R (2020) “Two birds with one stone” ruthenium(II) complex probe for biothiols discrimination and detection in vitro and in vivo. Adv Sci 7:2000458
Yang Y, Li Y, Zhi X, Xu Y, Li M (2020) A red-emitting luminescent probe for sequentially detecting Cu2+ and cysteine/histidine in aqueous solution and its imaging application in living zebrafish. Dyes Pigm 183:108690
Gao Q, Song B, Ye Z, Yang L, Liu R, Yuan J (2015) A highly selective phosphorescence probe for histidine in living bodies. Dalton Trans 44:18671–18676
Li X, Gorle AK, Sundaraneedi MK, Keene FR, Collins JG (2018) Kinetically-inert polypyridylruthenium(II) complexes as therapeutic agents. Coord Chem Rev 375:134–147
Ko C-N, Li G, Leung C-H, Ma D-L (2019) Dual function luminescent transition metal complexes for cancer theranostics: the combination of diagnosis and therapy. Coord Chem Rev 381:79–103
Liu J, Zhang C, Rees TW, Ke L, Ji L, Chao H (2018) Harnessing ruthenium(II) as photodynamic agents: encouraging advances in cancer therapy. Coord Chem Rev 363:17–28
Knoll JD, Turro C (2015) Control and utilization of ruthenium and rhodium metal complex excited states for photoactivated cancer therapy. Coord Chem Rev 282–283:110–126
Acknowledgements
The authors gratefully acknowledge the financial support from Australian Research Council (DE170100092) and National Health and Medical Research Council (APP1175808).
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection “Metal Legand Chromophores for Bioassays,” edited by Kenneth Kam-Wing Lo and Peter Kam-Keung Leung.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, M., Zhang, Z., Yong, J. et al. Determination and Imaging of Small Biomolecules and Ions Using Ruthenium(II) Complex-Based Chemosensors. Top Curr Chem (Z) 380, 29 (2022). https://doi.org/10.1007/s41061-022-00392-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41061-022-00392-8