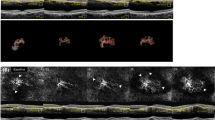
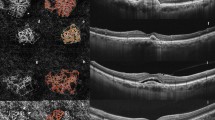
Abstract
Introduction
Macular neovascularization (MNV) secondary to age-related macular degeneration (AMD) is well managed by anti-vascular endothelial growth factor (anti-VEGF) intravitreal injections. However, outer retinal atrophy represents an unavoidable occurrence detected during follow-up. Several imaging metrics have been proposed as clinically relevant in stratifying the risk of onset of outer retinal atrophy. The main goal of this study is to evaluate the impact of noninvasive imaging metrics on the assessment of outer retinal atrophy onset in a large cohort of eyes with neovascular AMD managed in a real-world setting.
Methods
This study was a prospective, observational, case series. We included patients affected by newly diagnosed neovascular AMD, requiring anti-VEGF intravitreal injections. We collected clinical and imaging data, with a planned follow-up of 24 months. The multimodal imaging protocol included optical coherence tomography, optical coherence tomography angiography, and fundus autofluorescence. We collected noninvasive imaging metrics and we assessed the relationship with the morphological and functional outcome evaluated at 12-month and 24-month time points.
Results
We included 370 eyes of 370 patients with exudative AMD (210 male; mean age 79 ± 8 years). MNV were classified as follows: type 1, 198 (54%); type 2, 89 (24%); polypoidal choroidal vasculopathy, 29 (7%); and type 3, 54 (15%). A total of 120 out of 370 eyes (33%) showed complete outer retinal atrophy at the end of the 2-year follow-up. The presence of intraretinal fluid, thinning of the Sattler choroidal layer, late anti-VEGF switch, the overall number of anti-VEGF injections, and the perfusion characteristics of the MNV were found to be the most relevant factors associated with the onset of outer retinal atrophy. The other collected metrics were found to be less clinically relevant, also showing no cumulative effect in the multivariate analysis (p > 0.05).
Conclusions
We identified imaging metrics significantly associated with the 2-year risk onset of outer retinal atrophy. These metrics might pave the way for the development of future customized anti-VEGF treatment strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Many noninvasive imaging metrics have been proposed as associated with the course of neovascular age-related macular degeneration (AMD) and the risk of outer retinal atrophy. |
We conducted this study on a large cohort of macular neovascularization (MNV) eyes treated in a real-world setting to assess which imaging parameters are the most relevant to assess the morphological and functional outcome of neovascular AMD. |
What was learned from the study? |
The presence of intraretinal fluid, thinning of the Sattler choroidal layer, late anti-vascular endothelial growth factor (anti-VEGF) switch, the overall number of anti-VEGF injections, and the perfusion characteristics of the MNV were found to be the most relevant factors associated with the onset of outer retinal atrophy and with the morphological and functional outcome of neovascular AMD. These results will contribute to the development of personalized treatment strategies. |
Introduction
Macular neovascularization (MNV) and exudation are common complications occurring in advanced stages of age-related macular degeneration (AMD). Noninvasive multimodal imaging has identified several biomarkers of disease progression and outcomes, based on technique. The main prognostic optical coherence tomography (OCT)-based biomarkers in wet AMD include the different types of fluid [1], drusen and pseudodrusen [2], hyperreflective foci (HF) [3], and the status of the ellipsoid zone (EZ) [4]. These parameters have been found to be useful for outcome prediction and for setting treatment strategies [5,6,7,8]. More recently, quantitative optical coherence tomography angiography (OCTA) has also provided promising metrics for determining MNV severity and for stratifying patients in subgroups by different morpho-functional outcome [9,10,11,12]. All these parameters have been employed in different settings to assess the risk of AMD stage progression, MNV or geographic atrophy onset, and response to treatments, and also involving artificial intelligence-based approaches [6,7,8, 13,14,15,16]. However, a major unmet need remains the identification of the most relevant imaging metrics to determine the morphological and functional outcome of neovascular AMD. In the present study, we analyzed a large cohort of eyes treated in a real-life setting, focusing on the identification of structural OCT and angiographic biomarkers associated with the onset of outer retinal atrophy and visual outcome.
Methods
This study was designed as an observational, prospective case series. We recruited patients with AMD complicated by newly diagnosed MNV at the Department of Ophthalmology, IRCCS San Raffaele Scientific Institute, Milan, Italy. Exclusion criteria were other ocular disorders (glaucoma or other optic neuropathies, uveitis, retinal vascular disorders), any ophthalmic surgery performed in the 6 months before the inclusion, media opacities, and uncontrolled systemic disorders potentially affecting the interpretation of the study. The study was conducted in accordance with the Declaration of Helsinki and was approved by the ethical committee of IRCCS Scientific Institute San Raffaele Hospital (MIRD). The study used non-identifiable data, and all patients provided informed consent.
The ophthalmological examination included best-corrected visual acuity (BCVA) measured by ETDRS chart, anterior and posterior slit-lamp examination, tonometry, fundus autofluorescence (FAF), OCT (Heidelberg Spectralis HRA2+OCT, Heidelberg Engineering, Germany), and OCTA (DRI Triton Topcon, Topcon Inc., Japan). The diagnosis of MNV was confirmed by fluorescein angiography and the lesions were classified in accordance with the CONAN (Consensus on Neovascular AMD Nomenclature) classification. The standardized OCT acquisition protocol included radial (automated real time [ART] = 100, enhanced depth imaging [EDI], 1536 A-scans, 30° scan length, 6 sections), raster (ART = 30, EDI, 1024 A-scans, 20° × 15° volume length, 19 sections, inter-line space 240 µm), and dense (ART = 15, EDI, 15° × 5° volume length, 49 sections, 384 A-scans, inter-line space 30 µm) acquisitions with ART 25 and EDI. We included 4.5 × 4.5 mm OCTA acquisitions (Topcon quality index > 70).
All the patients underwent anti-vascular endothelial growth factor (anti-VEGF [either ranibizumab or aflibercept]) treatments considering a three-monthly loading dose followed by a treat-and-extend regimen carried out in a real-life clinical setting. Treatment switch was established at the discretion of the ophthalmologists.
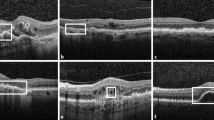
Central macular thickness (CMT) was obtained from the automatic measurement provided by Heidelberg Eye Explorer (HEYEX) software. Two expert graders (Andrea Saladino, Cecilia Mularoni) independently performed the manual measurements of choroidal thickness (CT), Haller layer thickness (HLT), and Sattler layer thickness (SLT) using Heidelberg segmentation tools on horizontal, foveal-centered, EDI radial scan. The lower margin of the retinal pigment epithelium (RPE) and the upper margin of the choroidal–scleral interface were considered as the extreme boundaries. CT and HLT were obtained from the mean value of five samples [subfoveal, 750 µm (right–left) and 1500 µm (right–left)], whereas SLT was considered as the result of CT-HLT, to address the challenging detection of SLT boundaries. The same graders analyzed structural OCT images in order to assess the presence of the following: drusen and pseudodrusen (at least 5 detected in three consecutive foveal scans), EZ status (preserved, disrupted, or absent), pigment epithelium detachment (PED) (at least 1 detected in three consecutive foveal scans), intraretinal fluid, subretinal fluid, subretinal hyperreflective material (SHRM), and retinal and choroidal HF, defined as well-circumscribed, discrete, dot-shaped, hyperreflective lesions (at least 10 detected in three consecutive foveal scans). From fluorescein angiography (FA) images, the two expert graders manually segmented the staining area of the MNV lesion on early FA (allowed range 15–30 s from the injection) and the leakage area on late FA (allowed range 6–9 min from the injection). In addition, the same graders assessed the presence of complete outer retinal atrophy on FAF images, considered as the presence of hyporeflective alterations with FAF intensity similar to retinal vessels. OCTA reconstructions of superficial capillary plexus (SCP), deep capillary plexus (DCP), and choriocapillaris (CC) were exported in .tiff format and loaded in ImageJ software for the calculation of vessel density (VD) [17]. We also used in-house scripts to calculate MNV vessel tortuosity (VT) as described in previous studies [18, 19].
For the statistical analyses, we considered baseline, 12-month, and 24-month measurements. All measurements were taken at least twice on the same scans to calculate repeatability and reproducibility. We also calculated the inter-class correlation coefficient (ICC) to assess inter-grader agreement through a two-way random-effects model (overall ICC 0.88; range 0.79–0.92).
The main outcome measure was the assessment of the relationship between the collected structural OCT, FA, and OCTA parameters and the onset of complete outer retinal atrophy. The secondary outcome was the correlation with visual outcome.
For the statistical analysis (SPSS, Chicago, IL, USA), age, gender, and clinical status were considered as fixed factors. The normality distribution of each variable was assessed by a linear mixed model, through frequency histograms and quantile–quantile plots. Descriptive statistics of continuous variables were reported as mean ± standard deviation, whereas frequency and percentages were described as categorical variables. A two-tailed t test was used to analyze continuous variables. Subgroup analyses were performed considering the presence of one or more structural OCT parameter detected at baseline (drusen, pseudodrusen, EZ status PED, intraretinal fluid, subretinal fluid, SHRM, retinal and choroidal HF). Pearson correlation analysis was adopted to assess the statistical relationship among all the considered metrics. In view of the multiple testing, the Bonferroni approach was chosen to address the question of multiple comparisons. Multivariate analyses were performed to assess the presence of possible cumulative effects of more parameters on the visual outcome. Each collected parameter was considered as dichotomous (present or absent) and used to establish a score discriminating more patient subgroups. One-way analysis of variance (ANOVA) and regression analysis were used to assess possible inter-subgroup differences in terms of morphological and functional outcome. The statistical significance was set to p < 0.05.
Results
We collected data from 370 eyes of 370 patients with exudative AMD (210 males; mean age 79 ± 8 years). MNV were classified as follows: type 1, 198 (54%); type 2, 89 (24%); polypoidal choroidal vasculopathy (PCV), 29 (7%); and type 3, 54 (15%). Baseline BCVA was 0.45 ± 0.34 LogMAR, which improved to 0.41 ± 0.38 LogMAR after 2 years of follow-up (p = 0.02), whereas baseline CMT was 384 ± 169 µm, and improved to 295 ± 137 µm at the end of the follow-up (p < 0.001), with a mean of 7 ± 2 anti-VEGF injections administered during the first year and 4 ± 2 administered during the second year. Eighty-four eyes (23%) underwent anti-VEGF switch over the 2-year follow-up. No significant differences were found regarding the clinical course of the disease, based on the different anti-VEGF molecules (p > 0.05). The complete clinical and imaging data are reported in Table 1. Six out of 370 eyes (2%) showed complete outer retinal atrophy already at baseline, whereas 120/370 eyes (33%) showed complete outer retinal atrophy at the end of the 2-year follow-up.
We included OCTA analysis for 145/370 (39%) eyes. The remaining OCTA data were discarded because of a Topcon quality index < 70, poorly detected MNV network, and high number of artifacts. The included eyes were classified as 92 type I MNV (63%) and 53 type II MNV (37%). The complete clinical and imaging data are reported in Table 2. After 2 years of follow-up, this cohort of eyes showed stable MNV VT and SCP VD (p > 0.05), whereas DCP VD and CC VD were significantly lower than baseline (p < 0.05).
The correlation analysis (Table 3) showed a statistically significant relationship between MNV type and the onset of outer retinal atrophy (Pearson coeff. 0.437; p < 0.001); in particular, PCV and type 3 lesions were associated with higher onset of outer retinal atrophy. The onset of retinal atrophy, other than being linked with significantly worse visual outcome (Pearson coeff. 0.723; p < 0.001), was significantly associated with the presence of intraretinal fluid (Pearson coeff. 0.444; p < 0.001), lower SLT (Pearson coeff. −0.331; p < 0.001), a lower number of intravitreal injections (Pearson coeff. −0.356; p < 0.001), higher MNV FA staining area (Pearson coeff. 0.361; p < 0.001), and lower MNV FA leakage area (Pearson coeff. −0.292; p < 0.001). In addition, the anti-VEGF switch was slightly significantly associated with lower incidence of outer retinal atrophy (Pearson coeff. −0.327; p < 0.001) and better visual outcome (Pearson coeff. −0.294; p < 0.001), although leading to a significantly higher number of intravitreal injections (Pearson coeff. 0.468; p < 0.001). Moreover, the presence of intraretinal fluid was the parameter most associated with EZ integrity detected at the end of the follow-up (Pearson coeff. 0.453; p < 0.001). Indeed, the other collected biomarkers showed lower significant values, as follows: drusen (Pearson coeff. 0.148; p = 0.004), pseudodrusen (Pearson coeff. 0.342; p = 0.002), intraretinal HF (Pearson coeff. 0.255; p = 0.002), SLT (Pearson coeff. −0.374; p = 0.001), MNV FA staining area (Pearson coeff. 0.384; p < 0.001), and MNV FA leakage area (Pearson coeff. -0.332; p < 0.001). Looking at the OCTA subgroup, high MNV VT values correlated negatively with DCP VD (Pearson coeff. −0.251; p = 0.002) and visual outcome (Pearson coeff. 0.243; p = 0.003) and were significantly associated with higher onset of atrophy (Pearson coeff. 0.361; p = 0.001). Intraretinal fluid was associated with lower VD values detected for SCP and DCP (Pearson coeff. −0.274; p = 0.001; Pearson coeff. −0.332; p = 0.001). In addition, CC VD correlated with visual outcome (Pearson coeff. −0.236; p = 0.001) and CT (Pearson coeff. 0.290; p = 0.001). Our multivariate analysis showed no cumulative effects of structural OCT biomarkers (drusen, pseudodrusen, PED, subretinal fluid, intraretinal fluid, and SHRM) on visual outcome (p > 0.05). Similarly, no significant differences were found for the cumulative OCTA and FA parameters (p > 0.05). Indeed, the subgroup analysis performed using a parameter-based score highlighted no significant difference in terms of visual outcome or onset of complete outer retinal atrophy (all p > 0.05).
Discussion
In the present paper, we described the 2-year course and outcome of a large cohort of eyes with AMD complicated by MNV. Visual outcome and number of anti-VEGF injections were in line with other real-world reports [20,21,22]. We confirmed that type 3 and aneurysmal type 1 MNV were associated with worse visual outcome and higher onset of complete outer retinal atrophy. Conversely, type 1 and type 2 MNV lesions were well managed by anti-VEGF injections. Drusen and pseudodrusen prevalence were found to be similar in our cohort of eyes (89% vs. 75% of cases). The most frequently found fluid type was subretinal fluid (88% of cases), followed by intraretinal fluid (61% of cases). Intraretinal fluid, which was found to be correlated with type 2 and type 3 MNV lesions, was significantly associated with worse visual outcome and higher onset of retinal atrophy, confirming its negative prognostic role in neovascular AMD [23]. Our findings further highlight the importance of different types of fluid in the pathogenesis of wet AMD, confirming the role of fluid interpretation as a fundamental step for the management of the disease. It is worth noting how the present real-life study agreed with the results previously obtained through more advanced artificial intelligence-based approaches [24]. Complete outer retinal atrophy was also significantly associated with a lower number of anti-VEGF injections, probably related to the fact that more atrophic lesions were also less characterized by exudation and blood retinal barrier impairment (higher FA staining and lower FA leakage). This finding confirmed what was recently found in a small cohort of eyes [18], further supporting the need for develo** customized anti-VEGF treatment regimens instead of fixed schemes (either pro re nata or treat-and extend). Indeed, the modification of fixed schemes guided by the recurrence of fluid is potentially prone to undertreatment of atrophic lesions. In addition, complete outer retinal atrophy was significantly associated with lower SLT values. This finding builds upon the results of a recent study showing that CC impairment and SLT thinning may be found in eyes with AMD undergoing neovascular changes [12]. Based on the present findings, SLT might also be considered a useful prognostic biomarker for later onset of MNV. Atrophy onset was negatively associated with anti-VEGF treatment switching, although this phenomenon may be easily related to the fact that the switch is associated with a higher number of anti-VEGF injections, rather then potentially better control of VEGF pathways [20]. OCTA investigation, performed in a limited number of eyes (145 out of 370), provided meaningful findings that included the association between retinal atrophy and higher MNV VT values, as already reported in previous studies [9, 10], further confirming the importance of quantitative OCTA-based assessment in the MNV clinical setting. Moreover, intraretinal fluid was associated with worse VD values for both SCP and DCP. Although we cannot exclude the possible displacement effect of intraretinal capillaries caused by the intraretinal cysts, the worse outcome associated with this specific type of fluid supports the hypothesis of a real intraretinal vascular impairment. Our post hoc analyses were not able to highlight cumulative effects in those eyes characterized by more structural OCT, FA, and OCTA parameters. This might mean that neovascular AMD pathogenesis is characterized by morphological findings with variable prognostic roles. In essence, we confirmed the negative prognostic role of intraretinal fluid, SLT thinning, high MNV VT, and low number of anti-VEGF injections. Pseudodrusen were significantly associated with a higher incidence of either neovascular AMD or geographic atrophy [25]. From this point of view, this OCT biomarker characterized the majority of our study cohort (75% of cases), confirming its association with MNV onset. However, we found no additional negative prognostic role regarding the course of the disease and the onset of outer retinal atrophy. PED and SHRM are known to be negative prognostic biomarkers in neovascular AMD [23]. The lack of significant a prognostic role of these two biomarkers in the present study might be related to the relatively low prevalence characterizing our cohort of eyes (26% and 28%, respectively). Retinal and choroidal HF can be associated with disease activity [26, 27]; however, we found no significant association with the onset of retinal atrophy in this case as well.
We are aware that our study has several limitations, first of all related to the relatively short follow-up and the real-life setting. The most evident limitation is the low number of eyes included for the OCTA analyses and characterized only by type 1 and type 2 MNV lesions. Unfortunately, OCTA is known to be inefficient in detecting aneurismal type 1 and type 3 MNV lesions [28]. In addition, depending on several variables, the proper detection of the MNV network in its entirety is often limited by the intensity of the signal and the quality of the images [29, 30]. For all these reasons, we were only able to include 145 out of 370 eyes in order to guarantee high quality data. We found no significant differences regarding the effect of each anti-VEGF molecule on the course of the disease. Although this finding is in line with previous evidence, we should highlight a relatively high variability regarding the treatment switch timing (early or late switch), as this was at the ophthalmologists’ discretion. Moreover, the lack of significance might also be related to the relatively short follow-up or the fact that all the patients received a proper number of injections, thus guaranteeing a good therapeutic cover during the entire follow-up, independently from the chosen anti-VEGF molecule. These are common factors affecting a real-life setting. From this point of view, future prospective studies should be focused on the assessment of possible differences among different anti-VEGF molecules. Similarly, dedicated analyses on the effect of early and late switch strategies on the course of the disease might also be clinically relevant. Furthermore, we are aware of the existence of detailed classification of different stages of EZ status and retinal atrophy [31]. However, to make the study easily reproducible in clinical settings, we preferred to adopt older EZ status classification (present, disrupted, and absent) and to include only complete outer retinal atrophy. Further studies are needed in order to assess in greater detail the course of neovascular AMD and the effect of each structural OCT and OCTA parameter on different stages of outer retinal atrophy.
Conclusions
In summary, the present study confirmed the role of intraretinal fluid, SLT thinning, high MNV VT, and low number of anti-VEGF injections as negative prognostic biomarkers in neovascular AMD. Other structural OCT and OCTA parameters may be associated with the onset of MNV, but were found not to be significantly associated with further worsening of the morpho-functional outcome. Future studies should be focused on the development of customized anti-VEGF treatment strategies, based on quantitative multimodal imaging, to optimize treatment effects and to improve clinical outcomes.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available due to internal rules and privacy issues.
References
Spaide RF, et al. Consensus nomenclature for reporting neovascular age-related macular degeneration data: consensus on neovascular age-related macular degeneration nomenclature study group. Ophthalmology. 2020;127(5):616–36.
Kovach JL, et al. The relationship between reticular pseudodrusen and severity of AMD. Ophthalmology. 2016;123(4):921–3.
Abri Aghdam K, Pielen A, Framme C, Junker B. Correlation between hyperreflective foci and clinical outcomes in neovascular age-related macular degeneration after switching to aflibercept. Invest Ophthalmol Vis Sci. 2015;56(11):6448–55.
Ehlers JP, et al. Longitudinal assessment of ellipsoid zone integrity, subretinal hyperreflective material, and subretinal pigment epithelium disease in neovascular age-related macular degeneration. Ophthalmol Retina. 2021;5(12):1204–13.
Pauleikhoff D, Kirchhof B. Retreatment criteria in anti-VEGF therapy of exudative AMD: critical analysis of present regimes and new morphological definition of “lesion activity.” Graefes Arch Clin Exp Ophthalmol. 2011;249(5):631–2.
Schmidt-Erfurth U, et al. Guidelines for the management of neovascular age-related macular degeneration by the European Society of Retina Specialists (EURETINA). Br J Ophthalmol. 2014;98(9):1144–67.
Amoaku WM, et al. Defining response to anti-VEGF therapies in neovascular AMD. Eye (Lond). 2015;29(6):721–31.
Schmidt-Erfurth U, Waldstein SM. A paradigm shift in imaging biomarkers in neovascular age-related macular degeneration. Prog Retin Eye Res. 2016;50:1–24.
Arrigo A, et al. Optical coherence tomography angiography can categorize different subgroups of choroidal neovascularization secondary to age-related macular degeneration. Retina. 2020;40(12):2263–9.
Arrigo A, Aragona E, Di Nunzio C, Bandello F, Parodi MB. Quantitative optical coherence tomography angiography parameters in type 1 macular neovascularization secondary to age-related macular degeneration. Transl Vis Sci Technol. 2020;9(9):48.
Arrigo A, et al. Morphological and functional relationship between OCTA and FA/ICGA quantitative features in AMD-related macular neovascularization. Front Med (Lausanne). 2021;8:758668.
Arrigo A, et al. Choroidal modifications preceding the onset of macular neovascularization in age-related macular degeneration. Ophthalmol Ther Ophthalmol Ther. 2022;11(1):377–86.
Schmidt-Erfurth U, et al. Machine learning to analyze the prognostic value of current imaging biomarkers in neovascular age-related macular degeneration. Ophthalmol Retina. 2018;2(1):24–30.
Ashraf M, Souka A, Adelman RA. Age-related macular degeneration: using morphological predictors to modify current treatment protocols. Acta Ophthalmol. 2018;96(2):120–33.
Phan LT, Broadhead GK, Hong TH, Chang AA. Predictors of visual acuity after treatment of neovascular age-related macular degeneration - current perspectives. Clin Ophthalmol. 2021;15:3351–67.
Ferrara D, Newton EM, Lee AY. Artificial intelligence-based predictions in neovascular age-related macular degeneration. Curr Opin Ophthalmol. 2021;32(5):389–96.
Arrigo A, et al. The impact of different thresholds on optical coherence tomography angiography images binarization and quantitative metrics. Sci Rep. 2021;11(1):14758.
Arrigo A, et al. Morphological and functional relationship between OCTA and FA/ICGA quantitative features in AMD-related macular neovascularization. Front Med (Lausanne). 2021;8: 758668.
Arrigo A, et al. Quantitative optical coherence tomography angiography parameter variations after treatment of macular neovascularization secondary to age-related macular degeneration. Retina. 2021;41(7):1463–9.
Arrigo A, et al. Different outcomes of anti-VEGF treatment for neovascular AMD according to neovascular sutypes and baseline features: 2-year real-life clinical outcomes. Biomed Res Int. 2021;2021:5516981.
Li E, Donati S, Lindsley KB, Krzystolik MG, Virgili G. Treatment regimens for administration of anti-vascular endothelial growth factor agents for neovascular age-related macular degeneration. Cochrane Database Syst Rev. 2020;5(5):CD012208.
Corazza P, et al. Long-term outcomes of intravitreal anti-VEGF therapies in patients affected by neovascular age-related macular degeneration: a real-life study. BMC Ophthalmol. 2021;21(1):300.
Arnold JJ, Markey CM, Kurstjens NP, Guymer RH. The role of sub-retinal fluid in determining treatment outcomes in patients with neovascular age-related macular degeneration–a phase IV randomised clinical trial with ranibizumab: the FLUID study. BMC Ophthalmol. 2016;16:31.
Schmidt-Erfurth U, et al. AI-based monitoring of retinal fluid in disease activity and under therapy. Prog Retin Eye Res. 2022;86: 100972.
Zhou Q, et al. Pseudodrusen and incidence of late age-related macular degeneration in fellow eyes in the comparison of age-related macular degeneration treatments trials. Ophthalmology. 2016;123(7):1530–40.
Parodi MB, et al. Hyperreflective foci number correlates with choroidal neovascularization activity in angioid streaks. Invest Ophthalmol Vis Sci. 2018;59(8):3314–9.
Arrigo A, et al. Hyperreflective foci precede macular neovascularization formation in angioid streaks. Retin Cases Brief Rep. 2023;17(4):392–6.
Spaide RF, Fujimoto JG, Waheed NK, Sadda SR, Staurenghi G. Optical coherence tomography angiography. Prog Retin Eye Res. 2018;64:1–55.
Arrigo A, et al. High reflectivity and low reflectivity properties on OCTA influence the detection of macular neovascularization in AMD. Front Phys. 2021;9:2021.
Arrigo A, Aragona E, Battaglia Parodi M, Bandello F. Quantitative approaches in multimodal fundus imaging: state of the art and future perspectives. Prog Retin Eye Res. 2023;92: 101111.
Sadda SR, et al. Consensus definition for atrophy associated with age-related macular degeneration on OCT: classification of atrophy report 3. Ophthalmology. 2018;125(4):537–48.
Acknowledgements
We thank the participants of the study.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Study design and manuscript drafting (Alessandro Arrigo MD, PhD, Andrea Saladino MD); data collection and analysis (Emanuela Aragona MD, PhD, Costanza Barresi MD, Cecilia Mularoni MD); critical revision of the manuscript, supervision of the entire study (Francesco Bandello MD, FEBO, Maurizio Battaglia Parodi MD).
Corresponding author
Ethics declarations
Conflict of Interest
Francesco Bandello consultant for: Alcon (Fort Worth, TX, USA), Alimera Sciences (Alpharetta, GA, USA), Allergan Inc (Irvine, CA, USA), Farmila-Thea (Clermont-Ferrand, France), Bayer Shering-Pharma (Berlin, Germany), Bausch & Lomb (Rochester, NY, USA), Genentech (San Francisco, CA, USA), Hoffmann-La-Roche (Basel, Switzerland), NovagaliPharma (Évry, France), Novartis (Basel, Switzerland), Sanofi-Aventis (Paris, France), Thrombogenics (Heverlee, Belgium), Zeiss (Dublin, CA, USA). Alessandro Arrigo, Andrea Saladino, Emanuela Aragona, Costanza Barresi, Cecilia Mularoni and Maurizio Battaglia Parodi have nothing to disclose.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki and was approved by the ethical committee of IRCCS Scientific Institute San Raffaele Hospital (MIRD). The study used non-identifiable data and all the patients provided informed consent.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Arrigo, A., Saladino, A., Aragona, E. et al. Clinical and Imaging Biomarkers Associated with Outer Retinal Atrophy Onset in Exudative Age-Related Macular Degeneration: A Real-Word Prospective Study. Ophthalmol Ther 13, 1185–1196 (2024). https://doi.org/10.1007/s40123-024-00903-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-024-00903-4